
Synopsis
Synopsis
0
CEP/COS
0
JDMF
0
EU WC
0
KDMF
0
NDC API
0
VMF
0
US Patents
0
US Exclusivities
0
Health Canada Patents
Annual Reports
NA
0
FDF
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
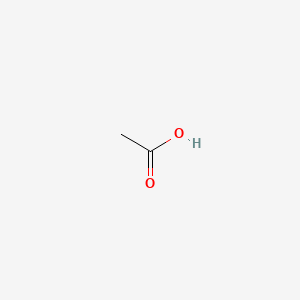

1. Acetic Acid Glacial
2. Acetic Acid, Glacial
3. Glacial Acetic Acid
4. Glacial, Acetic Acid
5. Vinegar
1. Ethanoic Acid
2. 64-19-7
3. Glacial Acetic Acid
4. Ethylic Acid
5. Vinegar Acid
6. Acetic Acid, Glacial
7. Acetic Acid Glacial
8. Methanecarboxylic Acid
9. Acetasol
10. Essigsaeure
11. Acide Acetique
12. Vinegar
13. Aci-jel
14. Azijnzuur
15. Aceticum Acidum
16. Acido Acetico
17. Kyselina Octova
18. Octowy Kwas
19. Pyroligneous Acid
20. Hoac
21. Azijnzuur [dutch]
22. Ethanoic Acid Monomer
23. Acetyl Alcohol
24. Essigsaeure [german]
25. Ethoic Acid
26. Caswell No. 003
27. Otic Tridesilon
28. Octowy Kwas [polish]
29. Otic Domeboro
30. Acetic Acid (natural)
31. Acide Acetique [french]
32. Acido Acetico [italian]
33. Fema No. 2006
34. Kyselina Octova [czech]
35. Acoh
36. Acetic Acid, Water Solutions
37. Acetic Acid-
38. Carboxylic Acids, C2-3
39. Ethanoate
40. Un2789
41. Un2790
42. Mecooh
43. Epa Pesticide Chemical Code 044001
44. Nsc 132953
45. Brn 0506007
46. Acetic Acid, Diluted
47. Acetic Acid [jan]
48. Ai3-02394
49. Ch3cooh
50. Acidum Aceticum Glaciale
51. Ch3-cooh
52. Ch3co2h
53. 10.methanecarboxylic Acid
54. Chembl539
55. Nsc-132953
56. Nsc-406306
57. Ins No.260
58. E-260
59. Chebi:15366
60. Ins-260
61. Q40q9n063p
62. Ethanoat
63. Shotgun
64. Mfcd00036152
65. Acetic Acid, Of A Concentration Of More Than 10 Per Cent, By Weight, Of Acetic Acid
66. Nsc-111201
67. Nsc-112209
68. Nsc-115870
69. Nsc-127175
70. 68475-71-8
71. C2:0
72. Perchloric Acid Solution
73. Orlex
74. Vosol
75. Wln: Qv1
76. Acetic Acid Solution, Not Less Than 50% But More Than 80% Acid, By Mass [un2790] [corrosive]
77. Acetic Acid Solution, With More Than 10% And Less Than 50% Acid, By Mass [un2790] [corrosive]
78. Acetic Acid, Glacial Or Acetic Acid Solution, >80% Acid, By Mass [un2789] [corrosive]
79. Acetic Acid, >=99.7%
80. Acetic Acid, Aqueous Solution
81. Fema Number 2006
82. Acetic Acid, Acs Reagent, >=99.7%
83. Acy
84. Hsdb 40
85. Ccris 5952
86. Methane Carboxylic Acid
87. Einecs 200-580-7
88. Acetic Acid 0.25% In Plastic Container
89. Ethylate
90. Acetic Aicd
91. Acetic-acid
92. Acidum Aceticum
93. Glacial Acetate
94. Acetic Cid
95. Actic Acid
96. Unii-q40q9n063p
97. Acetic -acid
98. Distilled Vinegar
99. Methanecarboxylate
100. Acetic Acid, Glacial [usp:jan]
101. Nat. Acetic Acid
102. Acetasol (tn)
103. Acetic Acid,glacial
104. Acetic Acid Natural
105. Vinegar (salt/mix)
106. Acetic Acid, Propionic Acid Distillate
107. Meco2h
108. Undiluted Acetic Acid
109. 63459-47-2
110. Oxytocin Identification
111. 3,3'-(1,4-phenylene)dipropiolic Acid
112. Hoocch3
113. Acetic Acid (recovered)
114. 546-67-8
115. Acetic Acid Lc/ms Grade
116. E 260
117. Acetic Acid [ii]
118. Acetic Acid [mi]
119. Acetic Acid, Acs Reagent
120. Dsstox_cid_4394
121. Acetic Acid Solution, 1n
122. Acetic Acid-[13c,d3]
123. Bmse000191
124. Bmse000817
125. Bmse000857
126. Otic Domeboro (salt/mix)
127. Ec 200-580-7
128. Acetic Acid (jp17/nf)
129. Acetic Acid [fhfi]
130. Acetic Acid [inci]
131. Acetic Acid [for Lc-ms]
132. Acetic Acid [vandf]
133. Dsstox_rid_77386
134. Nciopen2_000659
135. Nciopen2_000682
136. Dsstox_gsid_24394
137. Acetic Acid [mart.]
138. Acetic Acid, Glacial (usp)
139. Buffer Solution, Ph 4.64
140. 4-02-00-00094 (beilstein Handbook Reference)
141. Glacial Acetic Acid (jp17)
142. Un 2790 (salt/mix)
143. Acetic Acid [who-dd]
144. Acetic Acid [who-ip]
145. Aceticum Acidum [hpus]
146. Ins No. 260
147. Gtpl1058
148. Acetic Acid Glacial Hplc Grade
149. Acetic Acid Solution, For Hplc
150. Acetic Acid, Analytical Standard
151. Acetic Acid, Glacial Usp Grade
152. Dtxsid5024394
153. Acetic Acid, Puriss., >=80%
154. Acetic Acid, 99.8%, Anhydrous
155. Acetic Acid, Ar, >=99.8%
156. Acetic Acid, Lr, >=99.5%
157. Acetic Acid, Glacial Reagent Acs
158. Acetic Acid Solution, 1 N, 1 M
159. Acetic Acid, Extra Pure, 99.8%
160. Acetic Acid, 99.5-100.0%
161. Acetic Acid, Glacial, Acs Reagent
162. Str00276
163. Zinc5224164
164. Acetic Acid, Puriss., 99-100%
165. Tox21_301453
166. Acetic Acid, Glacial, >=99.85%
167. Bdbm50074329
168. Lmfa01010002
169. Nsc132953
170. Nsc406306
171. Stl264240
172. Tclp Extraction Fluid 2 (salt/mix)
173. Acetic Acid, 1% V/v Aqueous Solution
174. Acetic Acid, 4% V/v Aqueous Solution
175. Acetic Acid, Environmental Grade Plus
176. Acetic Acid, For Hplc, >=99.8%
177. Akos000268789
178. Acidum Aceticum [who-ip Latin]
179. Buffer Solution (acetate), Ph 4.01
180. Db03166
181. Un 2789
182. Acetic Acid, >=99.5%, Fcc, Fg
183. Acetic Acid, Natural, >=99.5%, Fg
184. Acetic Acid, Reagentplus(r), >=99%
185. Cas-64-19-7
186. Acetic Acid, Usp, 99.5-100.5%
187. Ncgc00255303-01
188. Acetic Acid 1000 Microg/ml In Methanol
189. Acetic Acid Solution, 25% W/w, Aqueous
190. Acetic Acid Solution, 56% W/w, Aqueous
191. Acetic Acid Solution, 60% W/w, Aqueous
192. Acetic Acid Solution, 84% W/w, Aqueous
193. Acetic Acid, 0.1n Standardized Solution
194. Acetic Acid, 1.0n Standardized Solution
195. Acetic Acid, Saj First Grade, >=99.0%
196. Buffer Solution (acetate), Ph 4.0-4.6
197. Db-085748
198. Acetic Acid 1000 Microg/ml In Acetonitrile
199. Acetic Acid, >=99.99% Trace Metals Basis
200. Acetic Acid, Jis Special Grade, >=99.7%
201. Acetic Acid, Purified By Double-distillation
202. Ft-0621735
203. Ft-0621743
204. Ft-0621764
205. Ft-0661109
206. Ft-0661110
207. Acetic Acid, Uv Hplc Spectroscopic, 99.9%
208. Acetic Acid, Vetec(tm) Reagent Grade, >=99%
209. Bifido Selective Supplement B, For Microbiology
210. C00033
211. D00010
212. Orlex Hc Component Acetic Acid, Glacial
213. Q47512
214. Vosol Hc Component Acetic Acid, Glacial
215. Acetic Acid, Glacial, Electronic Grade, 99.7%
216. Tridesilon Component Acetic Acid, Glacial
217. A834671
218. Acetasol Hc Component Acetic Acid, Glacial
219. Acetic Acid, >=99.7%, Saj Super Special Grade
220. Acetic Acid, Glacial Component Of Borofair
221. Acetic Acid, Glacial Component Of Orlex Hc
222. Acetic Acid, Glacial Component Of Vosol Hc
223. Sr-01000944354
224. Acetic Acid, Glacial Component Of Tridesilon
225. Sr-01000944354-1
226. Acetic Acid Solution, Saj First Grade, 27.0-33.0%
227. Acetic Acid, Glacial Component Of Acetasol Hc
228. Glacial Acetic Acid, Meets Usp Testing Specifications
229. Acetic Acid, >=99.7%, Suitable For Amino Acid Analysis
230. Acetic Acid, >=99.7%, For Titration In Non-aqueous Medium
231. Acetic Acid, For Luminescence, Bioultra, >=99.5% (gc)
232. Acetic Acid, Glacial Or Acetic Acid Solution, >80% Acid, By Mass
233. Acetic Acid Solution, Not Less Than 50% But More Than 80% Acid, By Mass
234. Acetic Acid Solution, With More Than 10% And Less Than 50% Acid, By Mass
235. Acetic Acid, P.a., Acs Reagent, Reag. Iso, Reag. Ph. Eur., 99.8%
236. Acetic Acid, Semiconductor Grade Mos Puranal(tm) (honeywell 17926)
237. Glacial Acetic Acid, United States Pharmacopeia (usp) Reference Standard
238. Acetic Acid, Acculute Standard Volumetric Solution, Final Concentration 1.0n
239. Acetic Acid, Puriss. P.a., Acs Reagent, Reag. Iso, Reag. Ph. Eur., >=99.8%
240. Glacial Acetic Acid, Pharmaceutical Secondary Standard; Certified Reference Material
241. 158461-04-2
242. Acetic Acid, Glacial, Pharmagrade, Usp, Jp, Ph Eur, Manufactured Under Appropriate Gmp Controls For Pharma Or Biopharmaceutical Production.
243. Acetic Acid, Puriss., Meets Analytical Specification Of Ph. Eur., Bp, Usp, Fcc, 99.8-100.5%
| Molecular Weight | 60.05 g/mol |
|---|---|
| Molecular Formula | C2H4O2 |
| XLogP3 | -0.2 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 2 |
| Rotatable Bond Count | 0 |
| Exact Mass | 60.021129366 g/mol |
| Monoisotopic Mass | 60.021129366 g/mol |
| Topological Polar Surface Area | 37.3 Ų |
| Heavy Atom Count | 4 |
| Formal Charge | 0 |
| Complexity | 31 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
| 1 of 6 | |
|---|---|
| Drug Name | Acetic acid |
| PubMed Health | Acetic Acid (Inside the bladder) |
| Drug Classes | Bladder Irrigant |
| Drug Label | Each 100 mL contains:Glacial Acetic Acid USP 0.25 gWater for Injection USP qspH: 3.1 (2.83.4)Calculated Osmolarity: 42 mOsmol/literThe formula of the active ingredient is:IngredientMolecular FormulaMolecular WeightGlacial Acetic Acid USPCH3COO... |
| Active Ingredient | Acetic acid, glacial |
| Dosage Form | Solution/drops |
| Route | Otic |
| Strength | 2% |
| Market Status | Prescription |
| Company | Wockhardt; Vintage; Taro |
| 2 of 6 | |
|---|---|
| Drug Name | Chirhostim |
| PubMed Health | Secretin (Injection) |
| Drug Classes | Diagnostic Agent, Pancreatic Function, Endocrine-Metabolic Agent |
| Active Ingredient | Secretin synthetic human |
| Dosage Form | For solution |
| Route | Intravenous |
| Strength | 40mcg/vial; 16mcg/vial |
| Market Status | Prescription |
| Company | Chirhoclin |
| 3 of 6 | |
|---|---|
| Drug Name | Vosol |
| PubMed Health | Acetic Acid/Hydrocortisone (Into the ear) |
| Drug Classes | Anti-Infective/Anti-Inflammatory Combination |
| Active Ingredient | Acetic acid, glacial |
| Dosage Form | Solution/drops |
| Route | Otic |
| Strength | 2% |
| Market Status | Prescription |
| Company | Hi Tech Pharma |
| 4 of 6 | |
|---|---|
| Drug Name | Acetic acid |
| PubMed Health | Acetic Acid (Inside the bladder) |
| Drug Classes | Bladder Irrigant |
| Drug Label | Each 100 mL contains:Glacial Acetic Acid USP 0.25 gWater for Injection USP qspH: 3.1 (2.83.4)Calculated Osmolarity: 42 mOsmol/literThe formula of the active ingredient is:IngredientMolecular FormulaMolecular WeightGlacial Acetic Acid USPCH3COO... |
| Active Ingredient | Acetic acid, glacial |
| Dosage Form | Solution/drops |
| Route | Otic |
| Strength | 2% |
| Market Status | Prescription |
| Company | Wockhardt; Vintage; Taro |
| 5 of 6 | |
|---|---|
| Drug Name | Chirhostim |
| PubMed Health | Secretin (Injection) |
| Drug Classes | Diagnostic Agent, Pancreatic Function, Endocrine-Metabolic Agent |
| Active Ingredient | Secretin synthetic human |
| Dosage Form | For solution |
| Route | Intravenous |
| Strength | 40mcg/vial; 16mcg/vial |
| Market Status | Prescription |
| Company | Chirhoclin |
| 6 of 6 | |
|---|---|
| Drug Name | Vosol |
| PubMed Health | Acetic Acid/Hydrocortisone (Into the ear) |
| Drug Classes | Anti-Infective/Anti-Inflammatory Combination |
| Active Ingredient | Acetic acid, glacial |
| Dosage Form | Solution/drops |
| Route | Otic |
| Strength | 2% |
| Market Status | Prescription |
| Company | Hi Tech Pharma |
BACKGROUND: Ultrasound (US)-guided percutaneous acetic acid injection therapy (PAIT) is effective for patients with hepatocellular carcinoma (HCC). This study aimed to determine the occurrence and predictive value of persistent intra-tumoral retention of acetic acid after PAIT. METHODS: /The trial/ prospectively studied 60 (52 M, mean age 68 +/- 10 years) patients with 72 HCC nodules (45 < or = 3 cm) treated with PAIT. The presence of post-treatment persistent retention of acetic acid, defined as a homogeneous and highly hyperechoid mass in US appearance 3 days after completion of the treatment, was correlated with the treatment response. RESULTS: The mean size of the treated tumor was 2.9 +/- 1.0 cm (range 1.5-5 cm). Thirty (42%) HCC nodules showed complete tumor necrosis demonstrated by contrast-enhanced dynamic CT. Complete response was found in 22 (69%) of 32 nodules showing persistent intra-tumoral retention of acetic acid (P < 0.001). Small (< or = 3 cm) tumor size was also significantly associated with complete tumor necrosis (P = 0.001). There were no significant differences of the injection volume and treatment sessions between those with and without complete tumor necrosis in either small or large (> 3 cm) HCC (P > 0.1). Multivariate logistic regression analysis showed that persistent retention of acetic acid (odds ratio (OR) 10.4, 95% confidence interval (CI) 3.1-34.7; P < 0.001) and tumor size < or = 3 cm (OR 6.8, 95%, CI 1.8-25.8; P = 0.002) were independent factors predicting complete tumor necrosis. CONCLUSIONS: The presence of persistent retention of acetic acid is associated with a favorable response and may predict complete tumor necrosis after PAIT.
PMID:15000280 Huo TI et al; Scand J Gastroenterol. 39(2):168-73 (2004).
EXPL THER BACKGROUND AND AIM: Application of acetic acid topically to the mucosal or serosal side of the stomach has been well used to create a chronic gastric ulcer model. The aim of the present study was to apply it as a new cytoreductive approach in a mouse model of gastric cancer. METHODS: A total of 43 genetically engineered mice, the so-called (INS-GAS) mice that develop spontaneously gastric cancer at 10-14 months of age, were included. Acetic acid-induced ulcer method was applied to mice under isofluran anesthesia. The ulcer at the cancer side was made by exposing either the anterior serosal or posterior mucosal side of gastric wall to 0.1 mL of 60% or 100% acetic acid for 30 or 60 s with a cylindrical metal mold (4 mm ID). Route to the serosal side was intra-abdominal and one to the mucosal side was through a small hole made in the forestomach. The opposite side of gastric wall (no treatment with acetic acid) was used as the corresponding control. After the mice were sacrificed, the stomachs were collected 1, 3, 6 hrs or 1, 3 and 7 days, postoperatively, and evaluated by visual inspection and histology. RESULTS: Gastric cancer was found in both the anterior and posterior walls of the corpus in all 43 mice. Intraluminal pH value was between 11 and 13. Severe necrosis in the cancer was observed in the side exposing to acetic acid, but not in the control side, shortly after the treatment (i.e. within 30 or 60 min). The muscularis mucosa and muscle layers were less damaged, regardless of the side of the treatment. Ulcer formation in the cancer took place 1, 3 or 7 days later. The ulcer depth was sometimes at the muscularis mucosa and muscle layers. At 3 and 7 days, regeneration of epithelial cells was clearly observed in the ulcer margin in the stomach of mice. CONCLUSIONS: Topical application of acetic acid either from mucosal or serosal surface promptly caused the necrosis of tumor, suggesting the potential approach of this simple and reliable method as a cytoreductive treatment of gastric cancer in patients through endoscopy or laparoscopy.
PMID:22486870 Okabe S et al; J Gastroenterol Hepatol. 27 Suppl 3:40-8 (2012).
MEDICATION (Vet): Vesicant, caustic, destructive of warts.
O'Neil, M.J. (ed.). The Merck Index - An Encyclopedia of Chemicals, Drugs, and Biologicals. Cambridge, UK: Royal Society of Chemistry, 2013., p. 11
EXPL THER OBJECTIVE: To investigate whether enhancing vaginal acidity improves the success of medical abortions in the midtrimester. METHODS: A double-blind, randomized, placebo-controlled trial was conducted with 48 women with missed midtrimester abortions. Twice daily, the study participants (n=24) were treated with a 3% acetic acid gel and the controls (n=24) with a placebo gel, starting 2 days prior to initiating the misoprostol treatment. The primary outcome measures were the rates of successful abortion within 24 and 48 hours. Secondary measures included gel tolerability and adverse effects of the misoprostol treatment. RESULTS: The success rates were higher in the study group, within both 24 hours (11/23 vs 3/24; P=0.011) and 48 hours (18/23 vs 6/24; P<0.001). Among the women with a vaginal pH of 5 or higher at baseline, acidic gel was also associated with higher success rates within 24 hours (8/13 vs 2/15; P<0.01) and 48 hours (13/13 vs 3/15; P<0,001). The vaginal gels were well tolerated and the misoprostol treatment produced no serious adverse effects. CONCLUSION: A 3% acetic acid gel appears to be an effective and safe preparatory adjuvant to vaginal misoprostol treatment for midtrimester medical abortions, especially in women with a vaginal pH of 5 or higher...
PMID:22980429 Abd-El-Maeboud KH et al; Int J Gynaecol Obstet. 119(3):248-52 (2012).
... Thirty-five patients with histologically confirmed high-grade squamous intraepithelial lesions of the cervix entered the study. ...Telomerase activity was detectable in 27 of 35 (77.1%) fresh tissue samples, 15 of 35 (42.9%) tissue samples swabbed with 5% acetic acid, and 0 of 10 (0%) normal cervical tissue samples, respectively. Twelve samples became telomerase negative after 5% acetic acid applied. Among the 15 telomerase-positive tissue samples swabbed with 5% acetic acid, 12 had relative weak telomerase activity compared to corresponding fresh tissue samples, the other 3 remained the same.
PMID:9784327 ChangChien CC, et al; Gynecol Oncol 71 (1): 99-103 (1998)
... Glacial acetic acid is widely used as a substitute for chemical peeling because it is readily ... available and affordable. However, its use can result in a number of serious complications. A 28-year-old female patient was admitted to /the/ hospital with deep second-degree chemical burns on her face caused by the application of a mixture of glacial acetic acid and flour for chemical peeling. During a 6-month follow-up, hypertrophic scarring developed on the both nasolabial folds despite scar management. Glacial acetic acid is a concentrated form of the organic acid, which gives vinegar its sour taste and pungent smell, and it is also an important reagent during the production of organic compounds. Unfortunately, misleading information regarding the use of glacial acetic acid for chemical peeling is causing serious chemical burns. Furthermore, there is high possibility of a poor prognosis, which includes inflammation, hypertrophic scar formation and pigmentation associated with its misuse. ...
PMID:20708991 Yoo JH et al; . J Plast Reconstr Aesthet Surg. 63(12):e829-31 (2010).
The enhanced toxicity of acid instilled directly into the rectum, without benefit of dilution and neutralization in the upper intestine, is evident in a case of acetic acid intoxication by accidental rectal administration of 50 mL of 9% acetic acid to a 5-yr-old boy. The complications included necrosis of the colon, acute renal failure, acute liver dysfunction, disseminated intravascular coagulopathy (DIC) and sepsis.
PMID:8007044 Kawamata M et al; J Toxicol Clin Toxicol 32 (3): 333-36 (1994)
In two patients, accidental application of acetic acid to the eyes followed very quickly by irrigation with water resulted in immediate corneal opacification. ... Regeneration of the epithelium took many months, but corneal anesthesia and opacity were permanent.
Bingham, E.; Cohrssen, B.; Powell, C.H.; Patty's Toxicology Volumes 1-9 5th ed. John Wiley & Sons. New York, N.Y. (2001)., p. V5 703
Used to treat infections in the ear canal.
Anti-Bacterial Agents
Substances that inhibit the growth or reproduction of BACTERIA. (See all compounds classified as Anti-Bacterial Agents.)
Indicators and Reagents
Substances used for the detection, identification, analysis, etc. of chemical, biological, or pathologic processes or conditions. Indicators are substances that change in physical appearance, e.g., color, at or approaching the endpoint of a chemical titration, e.g., on the passage between acidity and alkalinity. Reagents are substances used for the detection or determination of another substance by chemical or microscopical means, especially analysis. Types of reagents are precipitants, solvents, oxidizers, reducers, fluxes, and colorimetric reagents. (From Grant and Hackh's Chemical Dictionary, 5th ed, p301, p499) (See all compounds classified as Indicators and Reagents.)
G - Genito urinary system and sex hormones
G01 - Gynecological antiinfectives and antiseptics
G01A - Antiinfectives and antiseptics, excl. combinations with corticosteroids
G01AD - Organic acids
G01AD02 - Acetic acid
S - Sensory organs
S02 - Otologicals
S02A - Antiinfectives
S02AA - Antiinfectives
S02AA10 - Acetic acid
Acetic acid is absorbed from the GI tract and through the lung.
Bingham, E.; Cohrssen, B.; Powell, C.H.; Patty's Toxicology Volumes 1-9 5th ed. John Wiley & Sons. New York, N.Y. (2001)., p. V5 701
Acetic acid ... is readily metabolized by most tissues and may give rise to the production of ketone bodies as intermediates. In vitro, acetate is incorporated into phospholipids, neutral lipids, steroids, sterols, and saturated and unsaturated fatty acids in a variety of human and animal tissue preparations. ...Metabolism of 14(C) acetate in mice results in radioactivity associated with the protein fractions of plasma and most major tissues.
Bingham, E.; Cohrssen, B.; Powell, C.H.; Patty's Toxicology Volumes 1-9 5th ed. John Wiley & Sons. New York, N.Y. (2001)., p. V5 701
In the body, acetic acid is partially converted into formic acid.
Sheftel, V.O.; Indirect Food Additives and Polymers. Migration and Toxicology. Lewis Publishers, Boca Raton, FL. 2000., p. 650
When dogs were administered large doses (1-2 g/kg ip or sc) of sodium acetate, only small amounts appeared in the urine, which is evidence of the rapid utilization of acetic acid.
Bingham, E.; Cohrssen, B.; Powell, C.H.; Patty's Toxicology Volumes 1-9 5th ed. John Wiley & Sons. New York, N.Y. (2001)., p. V5 701
Acetic Acid is a known human metabolite of acetaldehyde.
S73 | METXBIODB | Metabolite Reaction Database from BioTransformer | DOI:10.5281/zenodo.4056560
Although acetic acid has been shown to induce apoptosis in yeast, the exact apoptotic mechanisms remain unknown. Here, /the study examined/ the effects of acetic acid treatment on yeast cells by 2-DE, revealing alterations in the levels of proteins directly or indirectly linked with the target of rapamycin (TOR) pathway: amino-acid biosynthesis, transcription/translation machinery, carbohydrate metabolism, nucleotide biosynthesis, stress response, protein turnover and cell cycle. The increased levels of proteins involved in amino-acid biosynthesis presented a counteracting response to a severe intracellular amino-acid starvation induced by acetic acid. Deletion of GCN4 and GCN2 encoding key players of general amino-acid control (GAAC) system caused a higher resistance to acetic acid indicating an involvement of Gcn4p/Gcn2p in the apoptotic signaling. Involvement of the TOR pathway in acetic acid-induced apoptosis was also reflected by the higher survival rates associated to a terminal deoxynucleotidyl transferase-mediated dUTP nick end labeling (TUNEL)-negative phenotype and lower reactive oxygen species levels of Deltator1 cells. In addition, deletion mutants for several downstream mediators of the TOR pathway revealed that apoptotic signaling involves the phosphatases Pph21p and Pph22p but not Sit4p. Altogether, /these/ results indicate that GAAC and TOR pathways (Tor1p) are involved in the signaling of acetic acid-induced apoptosis.
PMID:19137548 Almeida B et al; Proteomics. 9(3):720-32 (2009).
Acetic acid was found to have actions on urinary bladder smooth muscle in /the/ routine ion channel screening assays. Numerous studies have examined the mechanisms of bladder irritation by acetic acid; however, the direct effect of acetic acid on ion channels in detrusor smooth muscle cells has not been evaluated. /The study/ used whole-cell patch-clamp techniques to examine the effect of acetic acid on large-conductance Ca2+-activated K+ channels (BKCa) from guinea pig detrusor smooth muscle cells and CHO cells expressing recombinant human BKCaalphabeta1 (CHO BKCaalphabeta1) and human BKCaalpha (CHO BKCaalpha). Acetic acid activated BKCa currents in a concentration-dependent (0.01% to 0.05% v/v) manner in all the cell systems studied. Acetic acid (0.05%) increased BKCa current at +30 mV by 2764 +/- 918% (n=8) in guinea pig detrusor smooth muscle cells. Acetic acid (0.03%) shifted the V1/2 of conductance-voltage curve by 64 +/- 14 (n=5), 128 +/- 14 (n=5), and 126 +/- 12 mV (n=4) in CHO BKCaalpha, CHO BKCaalphabeta1 and detrusor smooth muscle cells, respectively. This effect of acetic acid was found to be independent of pH and was also not produced by its salt form, sodium acetate. Automated patch-clamp experiments also showed similar activation of CHO BKCaalphabeta1 by acetic acid. In conclusion, acetic acid directly activates BKCa channels in detrusor smooth muscle cells. This novel study necessitates caution while interpreting the results from acetic acid bladder irritation model.
PMID:17382925 Ghatta S et al; Eur J Pharmacol. 563(1-3):203-8 (2007).
/It was/ previously shown that acetic acid activates a mitochondria-dependent death process in Saccharomyces cerevisiae and that the ADP/ATP carrier (AAC) is required for mitochondrial outer membrane permeabilization and cytochrome c release. Mitochondrial fragmentation and degradation have also been shown in response to this death stimulus. Herein, /the study/ show that autophagy is not active in cells undergoing acetic acid-induced apoptosis and is therefore not responsible for mitochondrial degradation. Furthermore, /the study/ found that the vacuolar protease Pep4p and the AAC proteins have a role in mitochondrial degradation using yeast genetic approaches. Depletion and overexpression of Pep4p, an orthologue of human cathepsin D, delays and enhances mitochondrial degradation respectively. Moreover, Pep4p is released from the vacuole into the cytosol in response to acetic acid treatment. AAC-deleted cells also show a decrease in mitochondrial degradation in response to acetic acid and are not defective in Pep4p release. Therefore, AAC proteins seem to affect mitochondrial degradation at a step subsequent to Pep4p release, possibly triggering degradation through their involvement in mitochondrial permeabilization. The finding that both mitochondrial AAC proteins and the vacuolar Pep4p interfere with mitochondrial degradation suggests a complex regulation and interplay between mitochondria and the vacuole in yeast programmed cell death.
PMID:20345665 Pereira C et al ; Mol Microbiol. 76(6):1398-410 (2010).
 TAPI, a leading global supplier of APIs, provides over 350 products and customized CDMO solutions for every stage of development.
TAPI, a leading global supplier of APIs, provides over 350 products and customized CDMO solutions for every stage of development.
About the Company : Founded in 1935, TAPI Technology & API Services has a long legacy of advancing health through innovation. Today, we offer one of the industry’s most comprehensive API portfolios ...
About the Company : Brenntag is the global market leader in providing a full range of chemicals and ingredients and value-added services. By connecting chemical manufacturers (our suppliers) and chemi...

About the Company : Harman Finochem Limited is a leading India-based Pharmaceutical Company which specializes in the manufacture and export of more than 45 Active Pharmaceutical Ingredients (APls) of ...

About the Company : Nandu Group consisting of Nandu Chemical Industries, Nandu Chemicals Private Limited and Nandu Pharma Private Limited has a diversified product range to serve multiple applications...

About the Company : We are working in this business since last 14 years, supplying chemicals of all Grades & Quality to our valued customers in major Industries, entirely to their satisfaction and at ...

About the Company : QUALITY CHEMICALS, SL is a fine chemicals manufacturer of pure salts (mineral, metallic, organic, etc.), solvents, acids, basis and some organics. Our products are destined to a wi...

About the Company : ReAgent is a Leading UK Chemical Supplier. We specialise in services such as Chemical Blending, Contract Packing, Contract Manufacturing, Sachet Filling and more. We are based in C...

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]DRUG PRODUCT COMPOSITIONS

Related Excipient Companies

Excipients by Applications
Global Sales Information

Market Place

REF. STANDARDS & IMPURITIES

ANALYTICAL

ABOUT THIS PAGE
87
PharmaCompass offers a list of Acetic Acid API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Acetic Acid manufacturer or Acetic Acid supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Acetic Acid manufacturer or Acetic Acid supplier.
A Glacial Acetic Acid manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Glacial Acetic Acid, including repackagers and relabelers. The FDA regulates Glacial Acetic Acid manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Glacial Acetic Acid API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
click here to find a list of Glacial Acetic Acid manufacturers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PhamaCompass.
A Glacial Acetic Acid supplier is an individual or a company that provides Glacial Acetic Acid active pharmaceutical ingredient (API) or Glacial Acetic Acid finished formulations upon request. The Glacial Acetic Acid suppliers may include Glacial Acetic Acid API manufacturers, exporters, distributors and traders.
click here to find a list of Glacial Acetic Acid suppliers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PharmaCompass.
A Glacial Acetic Acid DMF (Drug Master File) is a document detailing the whole manufacturing process of Glacial Acetic Acid active pharmaceutical ingredient (API) in detail. Different forms of Glacial Acetic Acid DMFs exist exist since differing nations have different regulations, such as Glacial Acetic Acid USDMF, ASMF (EDMF), JDMF, CDMF, etc.
A Glacial Acetic Acid DMF submitted to regulatory agencies in the US is known as a USDMF. Glacial Acetic Acid USDMF includes data on Glacial Acetic Acid's chemical properties, information on the facilities and procedures used, and details about packaging and storage. The Glacial Acetic Acid USDMF is kept confidential to protect the manufacturer’s intellectual property.
click here to find a list of Glacial Acetic Acid suppliers with USDMF on PharmaCompass.
Glacial Acetic Acid Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Glacial Acetic Acid GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Glacial Acetic Acid GMP manufacturer or Glacial Acetic Acid GMP API supplier for your needs.
A Glacial Acetic Acid CoA (Certificate of Analysis) is a formal document that attests to Glacial Acetic Acid's compliance with Glacial Acetic Acid specifications and serves as a tool for batch-level quality control.
Glacial Acetic Acid CoA mostly includes findings from lab analyses of a specific batch. For each Glacial Acetic Acid CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Glacial Acetic Acid may be tested according to a variety of international standards, such as European Pharmacopoeia (Glacial Acetic Acid EP), Glacial Acetic Acid JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Glacial Acetic Acid USP).

