
Synopsis
Synopsis
0
API Suppliers
0
USDMF
0
CEP/COS
0
JDMF
0
EU WC
0
KDMF
0
NDC API
0
VMF
0
Listed Suppliers
0
EDQM
0
USP
0
JP
0
Others
0
Canada
0
South Africa
0
Listed Dossiers
DRUG PRODUCT COMPOSITIONS
0
US Patents
0
US Exclusivities
0
Health Canada Patents
Regulatory FDF Prices
NA
0
API
0
FDF
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
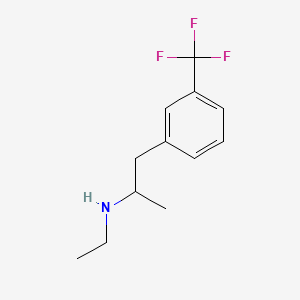

1. Fenfluramine Hydrochloride
2. Fenfluramine Hydrochloride, (+-)-isomer
3. Fenfluramine Hydrochloride, R Isomer
4. Fenfluramine Hydrochloride, R-isomer
5. Fenfluramine, (+-)-isomer
6. Fenfluramine, R Isomer
7. Fenfluramine, R-isomer
8. Fintepla
9. Hydrochloride, Fenfluramine
10. Isomeride
11. Pondimin
1. Rotondin
2. Obedrex
3. Pesos
4. Ponderax Pa
5. 458-24-2
6. Acino
7. Dl-fenfluramine
8. Fenfluramina
9. Fenfluraminum
10. Pondimin
11. (+-)-fenfluramine
12. Fenfluramin
13. N-ethyl-1-[3-(trifluoromethyl)phenyl]propan-2-amine
14. Phenfluramine
15. Brabafen
16. 2-ethylamino-1-(3-trifluoromethylphenyl)propane
17. Zx008
18. Fintepla
19. Ponderax
20. Fenfluramine (inn)
21. 1-(meta-trifluoromethyl-phenyl)-2 Ethylaminopropane
22. N-ethyl-alpha-methyl-3-trifluoromethylphenethylamine
23. 3-(trifluoromethyl)-n-ethyl-alpha-methylphenethylamine
24. 2ds058h2cf
25. Chebi:5000
26. Benzeneethanamine, N-ethyl-.alpha.-methyl-3-(trifluoromethyl)-
27. Fenfluramina [dcit]
28. Zx-008
29. J5.760f
30. Ncgc00159473-02
31. Z008
32. Fenfluraminum [inn-latin]
33. S-768
34. Z-008
35. Fenfluramine [inn]
36. Fenfluramine [inn:ban]
37. Dea No. 1670
38. Fenfluraminehydrochloride
39. Phenethylamine, N-ethyl-.alpha.-methyl-m-(trifluoromethyl)-
40. Ponderax (tn)
41. Hsdb 3080
42. S 768
43. Einecs 207-276-3
44. N-ethyl-1-(3-(trifluoromethyl)phenyl)propan-2-amine
45. Brn 4783711
46. Unii-2ds058h2cf
47. N-ethyl-alpha-methyl-m-(trifluoromethyl)phenethylamine
48. N-ethyl-alpha-methyl-3-(trifluoromethyl)benzeneethanamine
49. Benzeneethanamine, N-ethyl-alpha-methyl-3-(trifluoromethyl)-
50. Fenfluramine Hydrochloride 100 Microg/ml In Acetonitrile
51. Fenfluramine (+)
52. Fenfluramine (-)
53. Prestwick_868
54. Adipomin (salt/mix)
55. Ponderex (salt/mix)
56. Pondimin (salt/mix)
57. Phenethylamine, N-ethyl-alpha-methyl-m-(trifluoromethyl)-
58. Fenfluramine [mi]
59. Ncgc00159473-05
60. Dsstox_cid_3044
61. Fenfluramine [hsdb]
62. Dsstox_rid_76850
63. Fenfluramine [vandf]
64. 1-(m-trifluoromethyl-phenyl)-2-ethylaminopropane
65. Dsstox_gsid_23044
66. Schembl16215
67. Chembl87493
68. Divk1c_000980
69. Fenfluramine [who-dd]
70. Gtpl4613
71. Dtxsid4023044
72. Bdbm84738
73. Kbio1_000980
74. Ninds_000980
75. Nsc_3337
76. Tox21_111697
77. Fenfluramine Hydrochloride (salt/mix)
78. Akos011892098
79. Db00574
80. Idi1_000980
81. Ncgc00159473-03
82. Ncgc00159473-04
83. Ac-16024
84. Cas-458-24-2
85. Cas_16105-77-4
86. Ft-0770401
87. C06996
88. D07945
89. L000742
90. Q418928
91. N-ethyl-.alpha.-methyl-3-trifluoromethylphenethylamine
92. 3-(trifluoromethyl)-n-ethyl-.alpha.-methylphenethylamine
93. Ethyl({1-[3-(trifluoromethyl)phenyl]propan-2-yl})amine
94. N-ethyl-1-[3-(trifluoromethyl)phenyl]-2-propanamine #
95. Ethyl-[1-methyl-2-(3-trifluoromethyl-phenyl)-ethyl]-amine
96. N-ethyl-.alpha.-methyl-m-(trifluoromethyl)phenethylamine
97. N-ethyl-.alpha.-methyl-3-(trifluoromethyl)benzeneethanamine
98. N-ethyl-.alpha.-methyl-m-(trifluoromethyl)phenethylamine Hydrochloride (salt/mix)
| Molecular Weight | 231.26 g/mol |
|---|---|
| Molecular Formula | C12H16F3N |
| XLogP3 | 3.4 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 4 |
| Rotatable Bond Count | 4 |
| Exact Mass | 231.12348400 g/mol |
| Monoisotopic Mass | 231.12348400 g/mol |
| Topological Polar Surface Area | 12 Ų |
| Heavy Atom Count | 16 |
| Formal Charge | 0 |
| Complexity | 203 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 1 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
The Food and Drug Administration, acting on ... evidence about significant side-effects associated with fenfluramine and dexfenfluramine, has asked the manufacturers to voluntarily withdraw both treatments for obesity from the market. ... Both companies have agreed to voluntarily withdraw their drugs. The FDA is not requesting the withdrawal of phentermine, the third widely used medication for obesity. The action is based on ... findings from doctors who have evaluated patients taking these two drugs with echocardiograms, a special procedure that can test the functioning of heart valves. These findings indicate that approximately 30 percent of patients who were evaluated had abnormal echocardiograms, even though they had no symptoms. This is a much higher than expected percentage of abnormal test results.
US FDA; Center for Drug Evaluation and Research; FDA Announces Withdrawal Fenfluramine and Dexfenfluramine. For Immediate Release - September 15, 1997. Washington, DC: Food Drug Admin. Available from, as of October 12, 2005: https://www.fda.gov/cder/news/phen/fenphenpr81597.htm
Appetite Depressants; Serotonin Agents; Serotonin Uptake Inhibitors
National Library of Medicine's Medical Subject Headings online file (MeSH, 1999)
Adjunct to caloric restriction in the short term treatment (a few weeks) of exogenous obesity. /Use is included in the labeling approved by the US Food and Drug Administration. /Fenfluramine hydrochloride/
McEvoy, G.K. (ed.). American Hospital Formulary Service - Drug Information 97. Bethesda, MD: American Society of Health-System Pharmacists, Inc. 1997 (Plus Supplements)., p. 1770
Temporal association between use of fenfluramine (Pondimin) or dexfenfluramine (Redux) and the development of unusual mitral, aortic, tricuspid, and/or pulmonary valvular (usually multivalvular) and echocardiographic abnormalities (that sometimes occurred concomitantly with pulmonary hypertension, occasionally required open heart surgery, and rarely were fatal) resulted in the withdrawal of /this/ anorexigenic agents from the US market in 1997.
McEvoy, G.K. (ed.). American Hospital Formulary Service- Drug Information 2005. Bethesda, MD: American Society of Health-System Pharmacists, Inc. 2005 (Plus Supplements)., p. 2357
Fenfluramine is contraindicated in patients with severe hypertension, glaucoma, or symptomatic cardiovascular disease including arrhythmias, and in those with known hypersensitivity to fenfluramine or other sympathomimetic amines. Fenfluramine is contraindicated during or within 14 days of administration of monoamine oxidase inhibitors. The drug is also contraindicated in patients with a history of drug abuse. Fenfluramine should not be administered to patients with alcoholism, since adverse psychiatric effects (e.g., psychosis) may occur.
McEvoy, G.K. (ed.). American Hospital Formulary Service - Drug Information 97. Bethesda, MD: American Society of Health-System Pharmacists, Inc. 1997 (Plus Supplements)., p. 1770
Although some clinical studies have reported the use of fenfluramine in obese children, its safety and efficacy in pediatric patients have not been established and fenfluramine is not recommended for use in children younger than 12 years of age.
McEvoy, G.K. (ed.). American Hospital Formulary Service - Drug Information 97. Bethesda, MD: American Society of Health-System Pharmacists, Inc. 1997 (Plus Supplements)., p. 1770
General anesthetics should be administered with caution to patients receiving fenfluramine prior to surgery, since the drug may have catecholamine depleting effects following prolonged administration. If general anesthesia cannot be avoided, cardiac monitoring and facilities for cardiac resuscitation are essential during surgery in these patients.
McEvoy, G.K. (ed.). American Hospital Formulary Service - Drug Information 97. Bethesda, MD: American Society of Health-System Pharmacists, Inc. 1997 (Plus Supplements)., p. 1771
For more Drug Warnings (Complete) data for FENFLURAMINE (14 total), please visit the HSDB record page.
The lowest reported fatal dose of fenfluramine hydrochloride was 400 mg in a small child and the highest reported nonfatal dose was 1.8 g in an adult.
McEvoy, G.K. (ed.). American Hospital Formulary Service - Drug Information 97. Bethesda, MD: American Society of Health-System Pharmacists, Inc. 1997 (Plus Supplements)., p. 1771
Fenfluramine is indicated for the treatment of seizures in Dravet syndrome patients aged two years and older.
Treatment of seizures associated with Dravet syndrome as an add-on therapy to other antiepileptic medicines for patients 2 years of age and older.
Fenfluramine increases extracellular serotonin levels, and also acts as both a serotonergic 5-HT2 receptor agonist and 1 receptor antagonist. These activities, through an incompletely understood mechanism, lead to anti-epileptiform activity and therapeutic benefit. This modulation has other effects such as decreased appetite, weight loss, sedation, lethargy, increased blood pressure, and mood alteration including possible suicidal ideation. There is a risk of glaucoma and potentially fatal serotonin syndrome. Fenfluramine should be gradually withdrawn following treatment alteration or cessation.
Serotonin Agents
Drugs used for their effects on serotonergic systems. Among these are drugs that affect serotonin receptors, the life cycle of serotonin, and the survival of serotonergic neurons. (See all compounds classified as Serotonin Agents.)
Selective Serotonin Reuptake Inhibitors
Compounds that specifically inhibit the reuptake of serotonin in the brain. (See all compounds classified as Selective Serotonin Reuptake Inhibitors.)
N03
A - Alimentary tract and metabolism
A08 - Antiobesity preparations, excl. diet products
A08A - Antiobesity preparations, excl. diet products
A08AA - Centrally acting antiobesity products
A08AA02 - Fenfluramine
N - Nervous system
N03 - Antiepileptics
N03A - Antiepileptics
N03AX - Other antiepileptics
N03AX26 - Fenfluramine
Absorption
Fenfluramine has a steady-state Tmax of between four and five hours and an absolute bioavailability of approximately 68-74%. Fenfluramine administered to pediatric patients at 0.7 mg/kg/day up to 26 mg resulted in a mean Cmax of 68.0 ng/mL with a coefficient of variation of 41%; similarly the AUC0-24 was 1390 (44%) ng\*h/mL.
Route of Elimination
Over 90% of fenfluramine is excreted in urine and less than 5% in feces; unchanged fenfluramine and the major active metabolite norfenfluramine account for less than 25% of the recovered amount.
Volume of Distribution
Fenfluramine has an apparent volume of distribution of 11.9 L/kg with a coefficient of variation of 16.5% following oral administration in healthy subjects.
Clearance
Fenfluramine has a mean clearance of 24.8 L/h with a coefficient of variation of 29% in healthy subjects.
Postmortem blood concentrations in one adult and three children ranged from 6.5 to 16 mg/L. A fenfluramine hair level of 14.1 ng/mg was demonstrated in an overdose fatality.
Dart, R.C. (ed). Medical Toxicology. Third Edition, Lippincott Williams & Wilkins. Philadelphia, PA. 2004., p. 875
Fenfluramine is widely distributed into tissues with a Vd of 12 to 16 L/kg. ... Excretion of the parent compound is enhanced in acidic urine.
Dart, R.C. (ed). Medical Toxicology. Third Edition, Lippincott Williams & Wilkins. Philadelphia, PA. 2004., p. 874
Fenfluramine is widely distributed in almost all body tissues. Autoradiographic studies in rats showed highest concentrations of the drug in stomach and intestine; lower concentrations were found in lungs, liver, brain and spinal cord, and bone marrow. In monkeys, fenfluramine and its de-ethylated metabolite cross the placental barrier. It is not known whether fenfluramine is distributed into milk.
McEvoy, G.K. (ed.). American Hospital Formulary Service - Drug Information 97. Bethesda, MD: American Society of Health-System Pharmacists, Inc. 1997 (Plus Supplements)., p. 1770
Following oral administration, fenfluramine hydrochloride is readily absorbed from the GI tract. Correlation of blood concentrations with clinical effects has not been established. ... The rate of urinary excretion depends on urinary flow rate and pH ... Fenfluramine is also excreted in saliva and sweat to a small extent. /Fenfluramine hydrochloride/
McEvoy, G.K. (ed.). American Hospital Formulary Service - Drug Information 97. Bethesda, MD: American Society of Health-System Pharmacists, Inc. 1997 (Plus Supplements)., p. 1770
Fenfluramine is metabolized primarily in the liver by CYP1A2, CYP2B6, CYP2D6, CYP2C9, CYP2C19, and CYP3A4/5 to yield the major active metabolite norfenfluramine and several other minor inactive metabolites.
Fenfluramine hydrochloride is metabolized to norfenfluramine by de-ethylation; this metabolite is further deaminated and oxidized to m-trifluoromethylbenzoic acid. The drug is excreted principally in the urine as m-trifluoromethylhippuric acid, a glycine conjugate of m-trifluoromethylbenzoic acid, and smaller quantities of norfenfluramine and unchanged drug. There are wide interindividual variations in rates of biotransformation and elimination of fenfluramine and its metabolites... /Fenfluramine hydrochloride/
McEvoy, G.K. (ed.). American Hospital Formulary Service - Drug Information 97. Bethesda, MD: American Society of Health-System Pharmacists, Inc. 1997 (Plus Supplements)., p. 1770
/Fenfluramine/ is metabolized in the liver by N-dealkylation to the active metabolite norfenfluramine. Less than 15% of a therapeutic dose is excreted as parent compound or active metabolite; the remainder is nonactive benzoic acid and alcohol derivatives. ...
Dart, R.C. (ed). Medical Toxicology. Third Edition, Lippincott Williams & Wilkins. Philadelphia, PA. 2004., p. 874
99% of cerebral fenfluramine was dealkylated to norfenfluramine. N-acetylnorfenfluramine & m-trifluoromethyl hippuric acid were identified as cerebral metabolites.
Sherman AD, Gal EM; Cerebral Metabolism of Intraventricular (3)H-Fenfluramine; Neuropharmacology 16 (5): 309-15 (1977)
Fenfluramine has an elimination half-life of 20 hours in healthy subjects.
In one study, the mean elimination half-life of fenfluramine in patient with uncontrolled pH was about 20 hr while elimination half-life was about 11 hr when an acidic urinary pH was maintained. /Fenfluramine hydrochloride/
McEvoy, G.K. (ed.). American Hospital Formulary Service - Drug Information 97. Bethesda, MD: American Society of Health-System Pharmacists, Inc. 1997 (Plus Supplements)., p. 1770
Considerable first-pass effect due to rapid n-dealkylation of fenfluramine is apparent after per os doses. Rapid metabolism of derivative n-(2-benzoyloxyethyl)norfenfluramine has also been reported in man with apparent biological t/2 for total drug material of about 2 hr.
The Chemical Society. Foreign Compound Metabolism in Mammals Volume 3. London: The Chemical Society, 1975., p. 158
The half life of fenfluramine is 13 to 30 hours and is urine pH dependent. ...
Dart, R.C. (ed). Medical Toxicology. Third Edition, Lippincott Williams & Wilkins. Philadelphia, PA. 2004., p. 875
Dravet syndrome is a complex pediatric encephalopathy characterized by recurrent pharmacoresistant seizures of variable type, delayed development, and in many cases, impairment in speech, language, gait, and other neurocognitive functions. Despite substantial variation in presentation and severity, roughly 80% of patients with Dravet syndrome have mutations in the _SCN1A_ gene, which encodes the alpha subunit of a voltage-gated sodium channel (Nav1.1). This channel is predominantly localized in inhibitory GABAergic interneurons as well as in excitatory pyramidal neurons; it is thought that dysfunction of neurotransmission regulation results in the seizures and other corresponding symptoms of Dravet syndrome. Various _in vitro_ and _in vivo_ studies have demonstrated that fenfluramine is capable of acting as an agonist of multiple serotonin receptors including 5-HT1A, 5-HT1D, 5-HT2A, 5-HT2B, and 5-HT2C, as well as a 1 receptor antagonist. This is at least partly because fenfluramine, as well as its active metabolite norfenfluramine, can act on sodium-dependent serotonin transporters (SERTs) to reverse transport direction and thereby increase extracellular serotonin levels. However, work in animal models of Dravet syndrome suggest that only the modulation of 5-HT1D, 5-HT2C, 1, and possibly 5-HT2A receptors of fenfluramine result in the anti-epileptiform activity. Interestingly, 5-HT2B receptor agonism, which had previously been associated with cardiac valvulopathy, is not anticipated to have any therapeutic value in Dravet syndrome. Although the exact mechanism by which stimulation/inhibition of various receptors leads to the observed therapeutic benefit is unclear, it is hypothesized to be two-fold. Stimulation of 5-HT1D and 5-HT2C may result in increased GABAergic neurotransmission, while 1 receptor antagonism may help to modulate responses to _N_-methyl-D-aspartate (NMDA).
The exact mechanism of action of fenfluramine has not been clearly defined. Results of animal studies indicate that its appetite-inhibiting may result from stimulation of the ventromedial nucleus of the hypothalamus. The mechanism by which this stimulation is mediated has not yet been determined. Although fenfluramine is used in the treatment of obesity as an anorexigenic, it has not been firmly established that the pharmacologic action is principally one of appetite suppression; other CNS actions and/or metabolic effects may be involved. ... Cardiovascular and autonomic effects produced by fenfluramine in animals appear to be qualitatively similar to those of amphetamine, but as a pressor agent it is 10-20 times less potent than dextroamphetamine. Some clinical studies have shown fenfluramine to have hypotensive effects in obese hypertensive patients. EEG studies, both awake and during sleep, show fenfluramine to be qualitatively different from amphetamine and other amphetamine congeners and suggest that fenfluramine may be more similar to sedative psychotherapeutic drugs rather than CNS or cerebral stimulants. There is some evidence that fenfluramine interferes with CNS pathway which regulate the release of human growth hormone.
McEvoy, G.K. (ed.). American Hospital Formulary Service - Drug Information 97. Bethesda, MD: American Society of Health-System Pharmacists, Inc. 1997 (Plus Supplements)., p. 1770
The neurochemical mechanisms by which drugs acting on central serotoninergic system modify feeding were reviewed. Fenfluramine, a clinically effective appetite suppressant, releases serotonin from nerve terminals and inhibits its reuptake, and considerable evidence suggests that these effects mediate its anorectic activity. The D isomer of fenfluramine is particularly specific in affecting serotonin mechanisms and causing anorexia. Transmitters other than serotonin such as acetylcholine, catecholamines and GABA are also affected by systemic administration of fenfluramine, but some of these effects are secondary to fenfluramine's action on serotoninergic mechanisms. Moreover, there is no evidence that these brain substances are involved in fenfluramine's ability to cause anorexia. Several studies with drugs affecting different serotonin mechanisms such as release and uptake or mimicking the action of serotonin at post-synaptic receptors suggest that increase serotonin release and direct stimulation of postsynaptic receptors are the most effective mechanisms for causing depression of food intake, although inhibition of serotonin uptake may also contribute in appropriate conditions. Development of serotonin receptor hyposensitivity and, in some instances, decreased serotonin levels may lead to tolerance to the anorectic activity of drugs enhancing serotonin transmission, the degree of this depending critically on the type of effect on serotonin mechanisms and intensity and duration of serotonin receptor activation. Recent evidence suggests that a decrease in serotonin function causes stimulation of feeding. This may lead to development of new strategies for the treatment of clinical anorexias.
PMID:2427023 Garattini S et al; Appetite 7 Suppl: 15-38 (1986)
Global Sales Information

ABOUT THIS PAGE
51
PharmaCompass offers a list of Fenfluramine API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Fenfluramine manufacturer or Fenfluramine supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Fenfluramine manufacturer or Fenfluramine supplier.
A Fenfluramine manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Fenfluramine, including repackagers and relabelers. The FDA regulates Fenfluramine manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Fenfluramine API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
A Fenfluramine supplier is an individual or a company that provides Fenfluramine active pharmaceutical ingredient (API) or Fenfluramine finished formulations upon request. The Fenfluramine suppliers may include Fenfluramine API manufacturers, exporters, distributors and traders.
Fenfluramine Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Fenfluramine GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Fenfluramine GMP manufacturer or Fenfluramine GMP API supplier for your needs.
A Fenfluramine CoA (Certificate of Analysis) is a formal document that attests to Fenfluramine's compliance with Fenfluramine specifications and serves as a tool for batch-level quality control.
Fenfluramine CoA mostly includes findings from lab analyses of a specific batch. For each Fenfluramine CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Fenfluramine may be tested according to a variety of international standards, such as European Pharmacopoeia (Fenfluramine EP), Fenfluramine JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Fenfluramine USP).

