
Synopsis
Synopsis
0
API Suppliers
0
USDMF
0
CEP/COS
0
JDMF
0
EU WC
0
KDMF
0
NDC API
0
VMF
0
Listed Suppliers
0
EDQM
0
USP
0
JP
0
Others
0
Europe
0
Canada
0
Australia
0
South Africa
0
Listed Dossiers
DRUG PRODUCT COMPOSITIONS
0
US Patents
0
US Exclusivities
0
Health Canada Patents
US Medicaid
NA
Annual Reports
NA
Regulatory FDF Prices
NA
0
API
0
FDF
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
0
News #PharmaBuzz
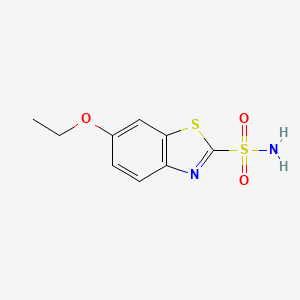

1. Ethamide
2. Ethoxazolamide
3. Ethoxyzolamide
1. 452-35-7
2. 6-ethoxy-2-benzothiazolesulfonamide
3. Ethoxyzolamide
4. Ethoxazolamide
5. Ethamide
6. Cardrase
7. Etoxzolamide
8. Glaucotensil
9. 6-ethoxy-1,3-benzothiazole-2-sulfonamide
10. Diuretic C
11. Redupresin
12. 2-benzothiazolesulfonamide, 6-ethoxy-
13. 6-ethoxybenzo[d]thiazole-2-sulfonamide
14. 6-ethoxyzolamide
15. 6-ethoxybenzothiazole-2-sulfonamide
16. U-4191
17. Nsc 10679
18. Chembl18
19. Nsc-10679
20. Redupresin;l-643786;pnu-4191
21. Mls000028637
22. 6-ethoxy-benzothiazole-2-sulfonic Acid Amide
23. Mingoral
24. Chebi:101096
25. Z52h4811wx
26. 6-ethoxy-2-benzothiazole8ulfonamide
27. Ncgc00018249-04
28. Smr000059148
29. Dsstox_cid_3021
30. 6-ethoxybenzothiazole-2-sulphonamide
31. Dsstox_rid_76834
32. Dsstox_gsid_23021
33. Cardrase (tn)
34. Cas-452-35-7
35. Ezl
36. Ethoxzolamide [usp]
37. Hsdb 3268
38. Einecs 207-199-5
39. Brn 0212240
40. Athamid
41. Unii-z52h4811wx
42. Ai3-50805
43. 3caj
44. 3dcw
45. 3mdz
46. Ethoxzolamide, Ezm
47. Mfcd00057089
48. 3dd0
49. Opera_id_1207
50. Ethoxzolamide [mi]
51. 6-(ethyloxy)-1,3-benzothiazole-2-sulfonamide
52. Ethoxzolamide [hsdb]
53. Schembl63941
54. 4-27-00-04404 (beilstein Handbook Reference)
55. Mls001077357
56. Ethoxzolamide [who-dd]
57. Gtpl6814
58. Dtxsid1023021
59. Bdbm10882
60. Zinc56721
61. Hms2093j03
62. Hms2094m03
63. Hms2233n09
64. Hms3373k15
65. Hms3715f20
66. Hms3746m15
67. Pharmakon1600-01505426
68. Ethoxzolamide [orange Book]
69. Bcp24088
70. Hy-b1480
71. Nsc10679
72. Pnu-4191
73. Tox21_110848
74. 6-ethoxy-2-benzothiazole Sulfonamide
75. Nsc759129
76. Wln: T56 Bn Dsj Cszw Go2
77. Akos015915628
78. Tox21_110848_1
79. Ccg-213432
80. Cs-7836
81. Db00311
82. Nsc-759129
83. Vs-0126
84. Benzothiazole, 6-ethoxy-2-sulfonamide-
85. Ncgc00018249-01
86. Ncgc00018249-02
87. Ncgc00018249-03
88. Ncgc00018249-05
89. Ncgc00022533-04
90. Ncgc00186657-01
91. 6-ethoxy-2-benzothiazolesulfonamide, 97%
92. Sbi-0206851.p001
93. 6-ethoxy-1,3-benzothiazole-2-sulfonamide #
94. Ft-0707866
95. Sw220095-1
96. D02441
97. Ab00383049_12
98. Ab00383049_13
99. Q265352
100. Sr-01000000236
101. L-643786
102. Sr-01000000236-2
103. Brd-k18131774-001-10-1
104. Ethoxzolamide, United States Pharmacopeia (usp) Reference Standard
| Molecular Weight | 258.3 g/mol |
|---|---|
| Molecular Formula | C9H10N2O3S2 |
| XLogP3 | 2 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 6 |
| Rotatable Bond Count | 3 |
| Exact Mass | 258.01328453 g/mol |
| Monoisotopic Mass | 258.01328453 g/mol |
| Topological Polar Surface Area | 119 Ų |
| Heavy Atom Count | 16 |
| Formal Charge | 0 |
| Complexity | 341 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
Carbonic Anhydrase Inhibitors; Diuretics
National Library of Medicine's Medical Subject Headings online file (MeSH, 1999)
.../USED FOR/ ADJUNCTIVE TREATMENT OF CHRONIC SIMPLE (OPEN-ANGLE) GLAUCOMA, SECONDARY GLAUCOMA, & PREOPERATIVELY IN ACUTE ANGLE-CLOSURE GLAUCOMA TO LOWER INTRAOCULAR PRESSURE PRIOR TO SURGERY.
Osol, A. and J.E. Hoover, et al. (eds.). Remington's Pharmaceutical Sciences. 15th ed. Easton, Pennsylvania: Mack Publishing Co., 1975., p. 867
EFFECTIVE DOSE APPEARS TO VARY FROM 125 TO 1000 MG/DAY, GIVEN ORALLY IN DIVIDED DOSES.
Goodman, L.S., and A. Gilman. (eds.) The Pharmacological Basis of Therapeutics. 5th ed. New York: Macmillan Publishing Co., Inc., 1975., p. 828
MEDICATION (VET): AS DIURETIC IN MILD CONGESTIVE CARDIAC EDEMAS.
Rossoff, I.S. Handbook of Veterinary Drugs. New York: Springer Publishing Company, 1974., p. 207
For more Therapeutic Uses (Complete) data for ETHOXZOLAMIDE (6 total), please visit the HSDB record page.
... CARBONIC ANHYDRASE INHIBITORS SHOULD BE USED CAUTIOUSLY IN PATIENTS WITH OBSTRUCTIVE PULMONARY DISEASE BECAUSE THEY MAY PRECIPITATE ACUTE RESP FAILURE. /CARBONIC ANHYDRASE INHIBITORS/
American Medical Association, Council on Drugs. AMA Drug Evaluations Annual 1994. Chicago, IL: American Medical Association, 1994., p. 2117
SINCE CARBONIC ANHYDRASE INHIBITORS MAY HAVE TERATOGENIC EFFECTS, THESE DRUGS SHOULD BE AVOIDED DURING EARLY PREGNANCY. /CARBONIC ANHYDRASE INHIBITORS/
American Medical Association, Council on Drugs. AMA Drug Evaluations Annual 1994. Chicago, IL: American Medical Association, 1994., p. 2117
CARBONIC ANHYDRASE INHIBITORS ALSO ARE ADMINISTERED FOR PREOPERATIVE TREATMENT OF ACUTE ANGLE-CLOSURE AND CONGENITAL GLAUCOMAS. ... mORE THAN 50% OF PATIENTS ... MUST DISCONTINUE THERAPY BECAUSE OF ADVERSE REACTIONS. ... OSMOTIC AGENTS ARE USED TO REDUCE INTRAOCULAR PRESSURE AND VITREOUS VOLUME RAPIDLY PRIOR TO IRIDECTOMY AND OTHER OCULAR SURGICAL PROCEDURES. /CARBONIC ANHYDRASE INHIBITORS/
American Medical Association, Council on Drugs. AMA Drug Evaluations Annual 1994. Chicago, IL: American Medical Association, 1994., p. 808
RENAL COLIC, HEMATURIA & OLIGURIA OR ANURIA MAY OCCUR DURING PROLONGED THERAPY... CARBONIC ANHYDRASE INHIBITORS REDUCE URIC ACID EXCRETION & INCR BLOOD URIC ACID LEVEL. ... HYPERURICEMIA IS USUALLY ASYMPTOMATIC BUT RARELY HAS LED TO AN EXACERBATION OF GOUT. /CARBONIC ANHYDRASE INHIBITORS/
American Medical Association, Council on Drugs. AMA Drug Evaluations Annual 1994. Chicago, IL: American Medical Association, 1994., p. 2116
For more Drug Warnings (Complete) data for ETHOXZOLAMIDE (7 total), please visit the HSDB record page.
For use in the treatment of duodenal ulcers, as a diuretic, and in the treatment of glaucoma, and may also be useful in the treatment of seizures associated with epilepsy.
Ethoxzolamide is an inhibitor of the carbonic anhydrase enzyme in proximal renal tubules that works by decreasing the reabsorption of water, sodium, potassium, bicarbonate. It also decreases the activity of carbonic anhydrase expressed in the CNS, which leads to increased seizure threshold. Inhibition of carbonic anhydrase in the eye contributes to its effect of reducing intraocular pressure and decreasing aqueous humor.
Diuretics
Agents that promote the excretion of urine through their effects on kidney function. (See all compounds classified as Diuretics.)
Carbonic Anhydrase Inhibitors
A class of compounds that reduces the secretion of H+ ions by the proximal kidney tubule through inhibition of CARBONIC ANHYDRASES. (See all compounds classified as Carbonic Anhydrase Inhibitors.)
Absorption
Rapidly absorbed with 65% bioavailability
ABSORBED FROM GI TRACT...DISTRIBUTED IN PLASMA, MUSCLES, BRAIN, KIDNEYS, LIVER, LUNGS, ERYTHROCYTES, AQUEOUS HUMOR, & CSF. APPROX 40%...EXCRETED UNCHANGED BY KIDNEYS /HUMAN, ORAL/
American Society of Hospital Pharmacists. Data supplied on contract from American Hospital Formulary Service and other current ASHP sources., p. 1976
2.5-5.5 hours
Ethoxzolamide binds to and inhibits carbonic anhydrase I, which plays an essential role in facilitating the transport of CO2 and H+ in the intracellular space, across biological membranes, and in the layers of the extracellular space. Through inhibition of the enzyme, the balance of applicable membrane equilibrium systems are affected.
MAJOR PHARMACOLOGICAL ACTION...IS INHIBITION OF...CARBONIC ANHYDRASE. STUDIES WITH PURIFIED ENZYME HAVE SHOWN THAT INHIBITION IS NONCOMPETITIVE. NONCATALYZED HYDRATION OR DEHYDRATION REACTION CAN TAKE PLACE, OF COURSE, IN ABSENCE OF ENZYME.
Goodman, L.S., and A. Gilman. (eds.) The Pharmacological Basis of Therapeutics. 5th ed. New York: Macmillan Publishing Co., Inc., 1975., p. 826
CARBONIC ANHYDRASE INHIBITORS...DECR CONCN OF HYDROGEN IONS AVAIL FOR EXCHANGE WITH SODIUM & POTASSIUM & TO COMBINE WITH BICARBONATE TO FORM CARBONIC ACID. /CARBONIC ANHYDRASE INHIBITORS/
Osol, A. and J.E. Hoover, et al. (eds.). Remington's Pharmaceutical Sciences. 15th ed. Easton, Pennsylvania: Mack Publishing Co., 1975., p. 862
...EFFECT /OF CARBONIC ANHYDRASE INHIBITORS/ ON INTRAOCULAR PRESSURE DOES NOT DEPEND UPON DIURESIS. THEY REDUCE AQUEOUS PRODN ... & THUS LOWER PRESSURE. /CARBONIC ANHYDRASE INHIBITORS/
American Medical Association, Council on Drugs. AMA Drug Evaluations Annual 1994. Chicago, IL: American Medical Association, 1994., p. 2116
ABOUT THIS PAGE
56
PharmaCompass offers a list of Ethoxzolamide API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Ethoxzolamide manufacturer or Ethoxzolamide supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Ethoxzolamide manufacturer or Ethoxzolamide supplier.
A Ethoxzolamide manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Ethoxzolamide, including repackagers and relabelers. The FDA regulates Ethoxzolamide manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Ethoxzolamide API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
A Ethoxzolamide supplier is an individual or a company that provides Ethoxzolamide active pharmaceutical ingredient (API) or Ethoxzolamide finished formulations upon request. The Ethoxzolamide suppliers may include Ethoxzolamide API manufacturers, exporters, distributors and traders.
Ethoxzolamide Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Ethoxzolamide GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Ethoxzolamide GMP manufacturer or Ethoxzolamide GMP API supplier for your needs.
A Ethoxzolamide CoA (Certificate of Analysis) is a formal document that attests to Ethoxzolamide's compliance with Ethoxzolamide specifications and serves as a tool for batch-level quality control.
Ethoxzolamide CoA mostly includes findings from lab analyses of a specific batch. For each Ethoxzolamide CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Ethoxzolamide may be tested according to a variety of international standards, such as European Pharmacopoeia (Ethoxzolamide EP), Ethoxzolamide JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Ethoxzolamide USP).

