
Synopsis
Synopsis
0
API Suppliers
0
USDMF
0
CEP/COS
0
JDMF
0
EU WC
0
KDMF
0
NDC API
0
VMF
0
Listed Suppliers
0
EDQM
0
USP
0
JP
0
Others
0
Europe
0
Canada
0
Australia
0
South Africa
0
Listed Dossiers
DRUG PRODUCT COMPOSITIONS
0
US Patents
0
US Exclusivities
0
Health Canada Patents
US Medicaid
NA
Annual Reports
NA
Regulatory FDF Prices
NA
0
API
0
FDF
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
0
News #PharmaBuzz
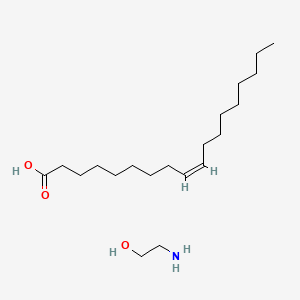

1. Ethamolin
2. Oleic Acid, 2-aminoethyl Ester
3. Monoethanolamine Oleate
1. Ethamolin
2. Monoethanolamine Oleate
3. Oldamin
4. 2272-11-9
5. Beta-hydroxyethylammonium Oleate
6. Oleic Acid (ethanolamine)
7. U4ry8mrx7c
8. 2-aminoethan-1-ol Oleate
9. Ethanolamine Oleate [usan]
10. Monoethanolamine Oleate [inn]
11. Nsc-760382
12. 2-aminoethanol;(z)-octadec-9-enoic Acid
13. Ethanolamine Oleate (usan)
14. Oleic Acid Compound With 2-aminoethanol (1:1)
15. Monoethanolamine Oleate (jan)
16. Neosclerol
17. Monolate
18. Moramin
19. Varicetin
20. Varex
21. 9-octadecenoic Acid (z)-, Compound With 2-aminoethanol (1:1)
22. Monoethanolamine Oleate [jan]
23. Ethamolin (tn)
24. Esclerosina
25. Antivariz
26. Phlebocid
27. Thanomin
28. Tindanol
29. Neo-varicane
30. Ethanolamine-oleate
31. Oldamin (tn)
32. Monoethanolamini Oleas
33. Monoaethanolamini Oleas
34. Monoetanolammina Oleato
35. Cis-9-octadecenoic Acid, Ethanolamine Salt
36. Unii-u4ry8mrx7c
37. Oleate De Monoethanolamine
38. Oleato De Monoetanolaminio
39. Oleic Acid Monoethanolamine
40. Oleic Acid Ethanolamine Salt
41. Oleic Acid, Ethanolamine Salt
42. Schembl146090
43. Ccris 3324
44. Monoetanolammina Oleato [dcit]
45. Oleic Acid Monoethanolamine Salt
46. Chembl3989529
47. Dtxsid8023008
48. Ethanolamine Oleate [mi]
49. Oleic Acid, 2-aminoethanol Salt
50. Oleic Acid, Monoethanolamine Salt
51. Chebi:178699
52. 9-octadecenoic Acid (z)-, Compd. With 2-aminoethanol (1:1)
53. Ethanolamine Oleate [vandf]
54. Hy-b2096
55. Monoethanolamini Oleas [inn-latin]
56. Einecs 218-878-0
57. Fo-611
58. Db06689
59. Monoethanolamine Oleate [mart.]
60. Nsc 760382
61. Monoethanolamine Oleate [who-dd]
62. Ethanolamine Oleate [orange Book]
63. Oleate De Monoethanolamine [inn-french]
64. Oleato De Monoetanolaminio [inn-spanish]
65. Cs-0017525
66. D02276
67. Ec 218-878-0
68. Oleic Acid, Compd. With 2-aminoethanol (1:1)
69. W-109765
70. (9z)-octadec-9-enoic Acid - 2-aminoethanol (1:1)
71. 9-octadecenoic Acid (9z)-, Compd. With 2-aminoethanol (1:1)
72. 9-octadecenoic Acid (z)-, Compd With 2-aminoethanol (1:1)
| Molecular Weight | 343.5 g/mol |
|---|---|
| Molecular Formula | C20H41NO3 |
| Hydrogen Bond Donor Count | 3 |
| Hydrogen Bond Acceptor Count | 4 |
| Rotatable Bond Count | 16 |
| Exact Mass | 343.30864417 g/mol |
| Monoisotopic Mass | 343.30864417 g/mol |
| Topological Polar Surface Area | 83.6 Ų |
| Heavy Atom Count | 24 |
| Formal Charge | 0 |
| Complexity | 244 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 1 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 2 |
| 1 of 2 | |
|---|---|
| Drug Name | Ethamolin |
| PubMed Health | Ethanolamine (Injection) |
| Drug Classes | Sclerosing Agent |
| Drug Label | ETHAMOLIN (Ethanolamine Oleate) Injection is a mild sclerosing agent. Chemically it is C17H33COOHNH2CH2CH2OH. It has the following structure:The empirical formula is C20H41NO3, representing a molecular weight of 343.55.ETHAMOLIN Injection consis... |
| Active Ingredient | Ethanolamine oleate |
| Dosage Form | Injectable |
| Route | Injection |
| Strength | 50mg/ml |
| Market Status | Prescription |
| Company | Qol Medcl |
| 2 of 2 | |
|---|---|
| Drug Name | Ethamolin |
| PubMed Health | Ethanolamine (Injection) |
| Drug Classes | Sclerosing Agent |
| Drug Label | ETHAMOLIN (Ethanolamine Oleate) Injection is a mild sclerosing agent. Chemically it is C17H33COOHNH2CH2CH2OH. It has the following structure:The empirical formula is C20H41NO3, representing a molecular weight of 343.55.ETHAMOLIN Injection consis... |
| Active Ingredient | Ethanolamine oleate |
| Dosage Form | Injectable |
| Route | Injection |
| Strength | 50mg/ml |
| Market Status | Prescription |
| Company | Qol Medcl |
For the treatment of patients with esophageal varices that have recently bled, to prevent rebleeding.
When injected intravenously, ethanolamine oleate acts primarily by irritation of the intimal endothelium of the vein and produces a sterile dose-related inflammatory response. This results in fibrosis and possible occlusion of the vein. Ethanolamine oleate also rapidly diffuses through the venous wall and produces a dose-related extravascular inflammatory reaction.
Sclerosing Solutions
Chemical agents injected into blood vessels and lymphatic sinuses to shrink or cause localized THROMBOSIS; FIBROSIS, and obliteration of the vessels. This treatment is applied in a number of conditions such as VARICOSE VEINS; HEMORRHOIDS; GASTRIC VARICES; ESOPHAGEAL VARICES; PEPTIC ULCER HEMORRHAGE. (See all compounds classified as Sclerosing Solutions.)
C - Cardiovascular system
C05 - Vasoprotectives
C05B - Antivaricose therapy
C05BB - Sclerosing agents for local injection
C05BB01 - Monoethanolamine oleate
Absorption
After injection into an esophageal varix, ethanolamine oleate is cleared from the injection site within five minutes via the portal vein. Some of the medication also flows into the azygos vein through the periesophageal vein if more than 20 mL is injected.
The oleic acid component of ethanolamine oleate is responsible for the inflammatory response, and may also activate coagulation in vivo by release of tissue factor and activation of Hageman factor. The ethanolamine component, however, may inhibit fibrin clot formation by chelating calcium, so that a procoagulant action of ethanolamine oleate has not been demonstrated.
Related Excipient Companies

Excipients by Applications
ABOUT THIS PAGE
50
PharmaCompass offers a list of Ethanolamine Oleate API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Ethanolamine Oleate manufacturer or Ethanolamine Oleate supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Ethanolamine Oleate manufacturer or Ethanolamine Oleate supplier.
A Ethanolamine Oleate manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Ethanolamine Oleate, including repackagers and relabelers. The FDA regulates Ethanolamine Oleate manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Ethanolamine Oleate API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
A Ethanolamine Oleate supplier is an individual or a company that provides Ethanolamine Oleate active pharmaceutical ingredient (API) or Ethanolamine Oleate finished formulations upon request. The Ethanolamine Oleate suppliers may include Ethanolamine Oleate API manufacturers, exporters, distributors and traders.
Ethanolamine Oleate Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Ethanolamine Oleate GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Ethanolamine Oleate GMP manufacturer or Ethanolamine Oleate GMP API supplier for your needs.
A Ethanolamine Oleate CoA (Certificate of Analysis) is a formal document that attests to Ethanolamine Oleate's compliance with Ethanolamine Oleate specifications and serves as a tool for batch-level quality control.
Ethanolamine Oleate CoA mostly includes findings from lab analyses of a specific batch. For each Ethanolamine Oleate CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Ethanolamine Oleate may be tested according to a variety of international standards, such as European Pharmacopoeia (Ethanolamine Oleate EP), Ethanolamine Oleate JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Ethanolamine Oleate USP).

