
Synopsis
Synopsis
0
USDMF
0
CEP/COS
0
JDMF
0
KDMF
0
VMF
0
Europe
0
Canada
0
Australia
0
South Africa
0
Listed Dossiers
DRUG PRODUCT COMPOSITIONS
0
US Patents
0
US Exclusivities
0
Health Canada Patents
US Medicaid
NA
Annual Reports
NA
Regulatory FDF Prices
NA
0
API
0
FDF
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
0
News #PharmaBuzz
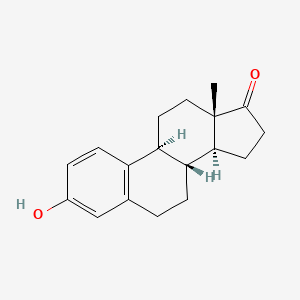

1. Estrone, (+-)-isomer
2. Estrone, (8 Alpha)-isomer
3. Estrone, (9 Beta)-isomer
4. Estrovarin
5. Folliculin (hormone)
6. Kestrone
7. Unigen
8. Wehgen
1. 53-16-7
2. Folliculin
3. Oestrone
4. Theelin
5. Follicular Hormone
6. Thelykinin
7. Ketohydroxyestrin
8. Estrovarin
9. Estrugenone
10. Femidyn
11. Kestrone
12. Tokokin
13. Estron
14. Follicunodis
15. Aquacrine
16. Disynformon
17. Folliculine
18. Glandubolin
19. Hiestrone
20. Hormestrin
21. Oestroform
22. Oestronum
23. Oestroperos
24. Folipex
25. Folisan
26. Kolpon
27. Menagen
28. Unden
29. Estrone-a
30. Ketohydroxyoestrin
31. 3-hydroxyestra-1,3,5(10)-trien-17-one
32. 1,3,5(10)-estratrien-3-ol-17-one
33. Estrona [spanish]
34. Folliculine Benzoate
35. Estrogenic Substance
36. Ovex (tablets)
37. Estronum [inn-latin]
38. Estrol
39. Estrona [inn-spanish]
40. Destrone
41. Oestrin
42. Endofolliculina
43. Estra-1,3,5(10)-trien-17-one, 3-hydroxy-
44. Crinovaryl
45. Cristallovar
46. Crystogen
47. Estrusol
48. Folikrin
49. Follestrine
50. Follestrol
51. Hormofollin
52. Hormovarine
53. Ketodestrin
54. Menformon
55. Mestronaq
56. Ovifollin
57. Perlatan
58. Solliculin
59. Thelestrin
60. Thynestron
61. Wynestron
62. (+)-estrone
63. Ketohydroxy-estratriene
64. 3-hydroxy-17-keto-estra-1,3,5-triene
65. Femestrone Injection
66. 3-hydroxyestra-1,3,5(10)-triene-17-one
67. 3-hydroxy-oestra-1,3,5(10)-trien-17-one
68. 1,3,5(10)-oestratrien-3-ol-17-one
69. 3-hydroxy-17-keto-oestra-1,3,5-triene
70. Delta-1,3,5-estratrien-3beta-ol-17-one
71. Fem-o-gen
72. (+/-)-oestrone
73. Delta-1,3,5-estratrien-3-beta-ol-17-one
74. Delta-1,3,5-oestratrien-3beta-ol-17-one
75. Delta-1,3,5-oestratrien-3-beta-ol-17-one
76. Folliculinum
77. (+/-)-estrone
78. Estrone Dl-form [mi]
79. Estrone, (+/-)-
80. Follicular-hormone
81. 3-hydroxy-1,3,5(10)-estratrien-17-one
82. (8r,9s,13s,14s)-3-hydroxy-13-methyl-7,8,9,11,12,14,15,16-octahydro-6h-cyclopenta[a]phenanthren-17-one
83. 19973-76-3
84. E(sub 1)
85. Estradiol Metabolite E1
86. 1,3,5-estratrien-3-ol-17-one
87. 3-hydroxy-estra-1,3,5(10)-trien-17-one
88. Chembl1405
89. Mls000028475
90. 2di9ha706a
91. X9xka379t9
92. Chebi:17263
93. Nsc-9699
94. Way 164397
95. Estra-1,3,5(10)-trien-17-one, 3-hydroxy-, (+/-)-
96. Estronum
97. Smr000058338
98. Dsstox_cid_2367
99. Dsstox_rid_76560
100. Dsstox_gsid_22367
101. (13s)-3-hydroxy-13-methyl-7,8,9,11,12,13,15,16-octahydro-6h-cyclopenta[a]phenanthren-17(14h)-one
102. [2,4,6,7-3h]-e1
103. Estrone (e1)
104. Hydroxyestrones
105. Unden (pharmaceutical) (van)
106. (9beta,13alpha)-3-hydroxyestra-1,3,5(10)-trien-17-one
107. Oestrone [steroidal Oestrogens]
108. (1s,10r,11s,15s)-5-hydroxy-15-methyltetracyclo[8.7.0.0^{2,7}.0^{11,15}]heptadeca-2(7),3,5-trien-14-one
109. (8r,9s,13s,14s)-3-hydroxy-13-methyl-6,7,8,9,11,12,13,14,15,16-decahydro-17h-cyclopenta[a]phenanthren-17-one
110. Ccris 285
111. Estrone (tn)
112. Hsdb 3324
113. Natural Estrogenic Substance-estrone
114. Nsc 9699
115. Einecs 200-164-5
116. Estrone (jan/usp/inn)
117. Estrone [usp:inn:ban]
118. Brn 1915077
119. Unii-2di9ha706a
120. Unii-x9xka379t9
121. Aquest
122. Cas-53-16-7
123. Ncgc00015423-03
124. (8r,9s,13s,14s)-3-hydroxy-13-methyl-6,7,8,9,11,12,13,14,15,16-decahydro-17h-cyclopenta(a)phenanthren-17-one
125. J3z
126. Cmc_13458
127. Estrone [vandf]
128. Opera_id_330
129. Estrone [hsdb]
130. Estrone, >=99%
131. Estrone [inn]
132. Estrone [jan]
133. Estrone [mi]
134. Estrone [mart.]
135. Prestwick0_000914
136. Prestwick1_000914
137. Prestwick2_000914
138. Prestwick3_000914
139. Spectrum5_002047
140. E0026
141. Estrone [usp-rs]
142. Estrone [who-dd]
143. Bmse000549
144. E 9750
145. Ec 200-164-5
146. Folliculinum [hpus]
147. Lopac0_000513
148. Schembl21702
149. Bspbio_000788
150. 3-08-00-01171 (beilstein Handbook Reference)
151. Mls001077340
152. Mls002695951
153. Mls006011890
154. Bidd:er0145
155. Estrone [orange Book]
156. Spbio_002977
157. Bpbio1_000868
158. Gtpl2818
159. Megxm0_000444
160. Sgcut00128
161. Estrone [usp Monograph]
162. Dtxsid4022367
163. Estrone 1.0 Mg/ml In Methanol
164. Acon0_000083
165. Acon1_000122
166. Bdbm17289
167. [2,4,6,7-3h]-estrone
168. Hms1570h10
169. Hms2090e22
170. Hms2097h10
171. Hms2232o15
172. Hms3261h07
173. Hms3714h10
174. Act02603
175. Bcp13336
176. Hy-b0234
177. To_000049
178. Estrone 100 Microg/ml In Methanol
179. Tox21_113567
180. Tox21_201375
181. Tox21_303651
182. Tox21_500513
183. Bbl033470
184. Bl-090
185. Lmst02010004
186. S1665
187. Stk801833
188. Zinc13509425
189. Estrone 1000 Microg/ml In Methanol
190. Akos005622512
191. Akos007930641
192. Tox21_113567_1
193. Ac-1395
194. Ccg-204604
195. Db00655
196. Ds-6316
197. Estrone 100 Microg/ml In Acetonitrile
198. Lp00513
199. Sdccgsbi-0050497.p002
200. Smp1_000123
201. Ncgc00023643-03
202. Ncgc00023643-04
203. Ncgc00023643-05
204. Ncgc00023643-06
205. Ncgc00023643-07
206. Ncgc00023643-08
207. Ncgc00023643-09
208. Ncgc00023643-10
209. Ncgc00023643-11
210. Ncgc00023643-12
211. Ncgc00023643-13
212. Ncgc00023643-15
213. Ncgc00023643-16
214. Ncgc00023643-22
215. Ncgc00179433-01
216. Ncgc00179433-02
217. Ncgc00179433-03
218. Ncgc00257402-01
219. Ncgc00258926-01
220. Ncgc00261198-01
221. Estrone, Meets Usp Testing Specifications
222. D1,3,5(10)-estratrien-3-ol-17-one
223. 3-hydroxyl-1,3,5(10)-estratien-17-one
224. 3-hydroxyoestra-1,3,5(10)-trien-17-one
225. Ab00382990
226. Estrone, Vetranal(tm), Analytical Standard
227. Eu-0100513
228. 3-hydroxy-estra-1,3,5(10)-triene-17-one
229. C00468
230. D00067
231. Ab00382990-16
232. Ab00382990-17
233. Ab00382990_18
234. Estra-1(10),2,4-trien-17-one, 3-hydroxy-
235. Ethinylestradiol Impurity C [ep Impurity]
236. 003e620
237. Q414986
238. Sr-01000000085
239. Sr-01000075867
240. Q-201073
241. Sr-01000000085-3
242. Sr-01000075867-1
243. Brd-k81839095-001-04-6
244. Brd-k81839095-001-30-1
245. Estradiol Hemihydrate Impurity A [ep Impurity]
246. 86c77018-146d-4603-acea-ca0d8c4f1e2c
247. Estrone, European Pharmacopoeia (ep) Reference Standard
248. Z1695906713
249. Estrone, United States Pharmacopeia (usp) Reference Standard
250. Estrone, Pharmaceutical Secondary Standard; Certified Reference Material
251. Estrone Solution, 1.0 Mg/ml In Methanol, Ampule Of 1 Ml, Certified Reference Material
252. (1s,10r,11s,15s)-5-hydroxy-15-methyltetracyclo[8.7.0.0;{2,7}.0;{11,15}]heptadeca-2,4,6-trien-14-one
253. (8r,13s)-3-hydroxy-13-methyl-6,7,8,9,11,12,13,14,15,16-decahydro-cyclopenta[a]phenanthren-17-one
| Molecular Weight | 270.4 g/mol |
|---|---|
| Molecular Formula | C18H22O2 |
| XLogP3 | 3.1 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 2 |
| Rotatable Bond Count | 0 |
| Exact Mass | 270.161979940 g/mol |
| Monoisotopic Mass | 270.161979940 g/mol |
| Topological Polar Surface Area | 37.3 Ų |
| Heavy Atom Count | 20 |
| Formal Charge | 0 |
| Complexity | 418 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 4 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
Estrogens
National Library of Medicine's Medical Subject Headings online file (MeSH, 2009)
Esterified Estrogens and Methyltestosterone Tablets H.S. and Esterified Estrogens and Methyltestosterone Tablets D.S. are indicated in the: Treatment of moderate to severe vasomotor symptoms associated with the menopause in those patients not improved by estrogens alone. (There is no evidence that estrogens are effective for nervous symptoms or depression without associated vasomotor symptoms, and they should not be used to treat such conditions.) /Included in US product label/
US Natl Inst Health; DailyMed. Current Medication Information EEMT H.S. (estrone sodium sulfate and methyltestosterone) tablet, coated (May 2010). Available from, as of February 28, 2011: https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?id=23251
/Estrone is indicated as/ Hormone replacement therapy (HRT) for estrogen deficiency symptoms in peri- and post-menopausal women. Prevention of osteoporosis in post-menopausal women at high risk of future fractures who are intolerant of, or contraindicated for, other medicinal products approved for the prevention of osteoporosis.
Datapharm Communications Ltd; Electronic Medicines Compendium (eMC), Summary of Product Characteristics (SPC) for Hormonin Tablets (Last updated December 2007). Available from, as of March 8, 2011: https://www.medicines.org.uk/EMC/medicine/20688/SPC/Hormonin+Tablets/#PHARMACOLOGICAL_PROPS
Esterified estrogens /is indicated/ for replacement in female hypogonadism...
American Society of Health System Pharmacists; AHFS Drug Information 2010. Bethesda, MD. (2010), p. 3139
For more Therapeutic Uses (Complete) data for ESTRONE (9 total), please visit the HSDB record page.
ESTROGENS INCREASE THE RISK OF ENDOMETRIAL CANCER: Close clinical surveillance of all women taking estrogens is important. Adequate diagnostic measures, including endometrial sampling when indicated, should be undertaken to rule out malignancy in all cases of undiagnosed persistent or recurring abnormal vaginal bleeding. There is no evidence that the use of "natural" estrogens results in a different endometrial risk profile than synthetic estrogens at equivalent estrogen doses.
US Natl Inst Health; DailyMed. Current Medication Information ENJUVIA (synthetic conjugated estrogens, b) tablet (March 2010). Available from, as of February 22, 2011: https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?id=22407
CARDIOVASCULAR AND OTHER RISKS: Estrogens with or without progestins should not be used for the prevention of cardiovascular disease or dementia. The estrogen alone substudy of the Women's Health Initiative (WHI) reported increased risks of stroke and deep vein thrombosis (DVT) in postmenopausal women (50 to 79 years of age) during 6.8 years and 7.1 years, respectively, of treatment with oral conjugated estrogens (CE 0.625 mg) alone per day, relative to placebo.
US Natl Inst Health; DailyMed. Current Medication Information ENJUVIA (synthetic conjugated estrogens, b) tablet (March 2010). Available from, as of February 22, 2011: https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?id=22407
The Women's Health Initiative Memory Study (WHIMS), a substudy of WHI, reported increased risk of developing probable dementia in postmenopausal women 65 years of age or older during 4 years of treatment with oral conjugated estrogens plus medroxyprogesterone acetate relative to placebo. It is unknown whether this finding applies to younger postmenopausal women or to women taking estrogen alone therapy. Other doses of oral conjugated estrogens with medroxyprogesterone acetate, and other combinations and dosage forms of estrogens and progestins were not studied in the WHI clinical trials and, in the absence of comparable data, these risks should be assumed to be similar. Because of these risks, estrogens with or without progestins should be prescribed at the lowest effective doses and for the shortest duration consistent with treatment goals and risks for the individual woman.
US Natl Inst Health; DailyMed. Current Medication Information EEMT H.S. (estrone sodium sulfate and methyltestosterone) tablet, coated (May 2010). Available from, as of February 28, 2011: https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?id=23251
Other doses of oral conjugated estrogens with medroxyprogesterone acetate, and other combinations and dosage forms of estrogens and progestins were not studied in the WHI clinical trials and, in the absence of comparable data, these risks should be assumed to be similar. Because of these risks, estrogens with or without progestins should be prescribed at the lowest effective doses and for the shortest duration consistent with treatment goals and risks for the individual woman.
US Natl Inst Health; DailyMed. Current Medication Information EEMT H.S. (estrone sodium sulfate and methyltestosterone) tablet, coated (May 2010). Available from, as of February 28, 2011: https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?id=23251
For more Drug Warnings (Complete) data for ESTRONE (45 total), please visit the HSDB record page.
For management of perimenopausal and postmenopausal symptoms.
Estrone, a synthetically prepared or naturally occurring steroidal estrogen obtained from pregnant equine urine, is the primary circulating estrogen after menopause. Estrone is naturally derived from the peripheral conversion of androstenedione by an aromatase enzyme found in adipose tissues and is converted to estradiol in peripheral tissues. The estrogenic potency of estrone is one third that of estradiol. Estropipate is piperazine-stabilized estrone sulfate. Estrone, and estropipate are used to treat abnormalities related to gonadotropin hormone dysfunction, vasomotor symptoms, atrophic vaginitis, and vulvar atrophy associated with menopause, and for the prevention of osteoporosis due to estrogen deficiency.
Estrogens
Compounds that interact with ESTROGEN RECEPTORS in target tissues to bring about the effects similar to those of ESTRADIOL. Estrogens stimulate the female reproductive organs, and the development of secondary female SEX CHARACTERISTICS. Estrogenic chemicals include natural, synthetic, steroidal, or non-steroidal compounds. (See all compounds classified as Estrogens.)
G - Genito urinary system and sex hormones
G03 - Sex hormones and modulators of the genital system
G03C - Estrogens
G03CA - Natural and semisynthetic estrogens, plain
G03CA07 - Estrone
G - Genito urinary system and sex hormones
G03 - Sex hormones and modulators of the genital system
G03C - Estrogens
G03CC - Estrogens, combinations with other drugs
G03CC04 - Estrone
Absorption
43%
The natural estrogens are generally quickly and well absorbed from the gastrointestinal tract, there being little difference between estrone, estradiol and estriol. Estrogens are inactivated in the liver. A proportion of absorbed estrogen is excreted in the bile and then reabsorbed from the intestine.
Datapharm Communications Ltd; Electronic Medicines Compendium (eMC), Summary of Product Characteristics (SPC) for Hormonin Tablets (Last updated December 2007). Available from, as of March 8, 2011: https://www.medicines.org.uk/EMC/medicine/20688/SPC/Hormonin+Tablets/#PHARMACOLOGICAL_PROPS
The distribution of exogenous estrogens is similar to that of endogenous estrogens. Estrogens are widely distributed in the body and are generally found in higher concentrations in the sex hormone target organs. Estrogens circulate in the blood largely bound to sex hormone binding globulin (SHBG) and albumin.
US Natl Inst Health; DailyMed. Current Medication Information EEMT H.S. (estrone sodium sulfate and methyltestosterone) tablet, coated (May 2010). Available from, as of February 22, 2011: https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?id=23251
Following administration of Hormonin, physiological estrogen concentrations are achieved at different rates. The maximum plasma levels and the time to reach the peak in the plasma level varied between subjects and was; estrone 750-2116 pmol/litre, 0.5-6.0 hours; estradiol 246-813 pmol/litre, 1-8 hours; estriol 173-241 pmol/litre, 5-12 hours. Following steady state conditions after cessation of Hormonin therapy, estrogen levels remained in the pre-menopausal range for approximately 48 hours.
Datapharm Communications Ltd; Electronic Medicines Compendium (eMC), Summary of Product Characteristics (SPC) for Hormonin Tablets (Last updated December 2007). Available from, as of March 8, 2011: https://www.medicines.org.uk/EMC/medicine/20688/SPC/Hormonin+Tablets/#PHARMACOLOGICAL_PROPS
Estrogens are available for oral, parenteral, transdermal, or topical administration ... absorption is generally good with the appropriate preparation. /Estrogens/
Hardman, J.G., L.E. Limbird, P.B. Molinoff, R.W. Ruddon, A.G. Goodman (eds.). Goodman and Gilman's The Pharmacological Basis of Therapeutics. 9th ed. New York, NY: McGraw-Hill, 1996., p. 1419
For more Absorption, Distribution and Excretion (Complete) data for ESTRONE (11 total), please visit the HSDB record page.
Hepatic.
Exogenous estrogens are metabolized in the same manner as endogenous estrogens. Circulating estrogens exist in a dynamic equilibrium of metabolic interconversions. These transformations take place mainly in the liver. Estradiol is converted reversibly to estrone, and both can be converted to estriol, which is the major urinary metabolite. Estrogens also undergo enterohepatic recirculation via sulfate and glucuronide conjugation in the liver, biliary secretion of conjugates into the intestine, and hydrolysis in the gut followed by reabsorption. In postmenopausal women, a significant proportion of the circulating estrogens exist as sulfate conjugates, especially estrone sulfate, which serves as a circulating reservoir for the formation of more active estrogens.
US Natl Inst Health; DailyMed. Current Medication Information EEMT H.S. (estrone sodium sulfate and methyltestosterone) tablet, coated (May 2010). Available from, as of February 22, 2011: https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?id=23251
In the liver estradiol is readily oxidised to estrone and both estradiol and estrone are converted by hydration to estriol. Metabolites of estrogens are mainly excreted in the urine as conjugates of glucuronic and sulphuric acid.
Datapharm Communications Ltd; Electronic Medicines Compendium (eMC), Summary of Product Characteristics (SPC) for Hormonin Tablets (Last updated December 2007). Available from, as of March 8, 2011: https://www.medicines.org.uk/EMC/medicine/20688/SPC/Hormonin+Tablets/#PHARMACOLOGICAL_PROPS
The 17beta-hydroxy steroid dehydrogenase transforms estrone to estradiol reversibly. This enzyme occurred in all tissues of all species examined and is linked to either the cytosolic or microsomal subcellular compartment. In human liver, a NAD-linked 17beta-hydroxy steroid 3-hydrogenase occurs in cytosol and in microsomes, and a further NADP-linked enzyme has been found in cytosol. Hence, estrone and estradiol are largely biologically equivalent; they are also metabolized via the same pathways.
IARC. Monographs on the Evaluation of the Carcinogenic Risk of Chemicals to Humans. Geneva: World Health Organization, International Agency for Research on Cancer, 1972-PRESENT. (Multivolume work). Available at: https://monographs.iarc.fr/ENG/Classification/index.php, p. V21 356 (1979)
The steroidal estrogens are metabolized principally in the liver, although the kidneys, gonads, and muscle tissues may be involved to some extent. The steroids and their metabolites are conjugated at the hydroxyl group of the C 3 position with sulfuric or glucuronic acid; these conjugates may undergo further metabolic change. Conjugation increases water solubility and facilitates excretion in urine. Large amounts of free estrogens are also distributed into the bile, reabsorbed from the GI tract, and recirculated through the liver where further degradation occurs. /Estrogen General Statement/
American Society of Health System Pharmacists; AHFS Drug Information 2010. Bethesda, MD. (2010), p. 3131
For more Metabolism/Metabolites (Complete) data for ESTRONE (13 total), please visit the HSDB record page.
Estrone has known human metabolites that include (2S,3S,4S,5R)-3,4,5-Trihydroxy-6-[[(8R,9S,13S,14S)-13-methyl-17-oxo-7,8,9,11,12,14,15,16-octahydro-6H-cyclopenta[a]phenanthren-3-yl]oxy]oxane-2-carboxylic acid.
S73 | METXBIODB | Metabolite Reaction Database from BioTransformer | DOI:10.5281/zenodo.4056560
19 hours
Estrogens enter the cells of responsive tissues (e.g. female organs, breasts, hypothalamus, pituitary) where they interact with estrogen receptors. Hormone-bound estrogen receptors dimerize, translocate to the nucleus of cells and bind to estrogen response elements (ERE) of genes. Binding to ERE alters the transcription rate of affected genes. Estrogens increase the hepatic synthesis of sex hormone binding globulin (SHBG), thyroid-binding globulin (TBG), and other serum proteins and suppress follicle-stimulating hormone (FSH) release from the anterior pituitary.
Endogenous estrogens are largely responsible for the development and maintenance of the female reproductive system and secondary sexual characteristics. Although circulating estrogens exist in a dynamic equilibrium of metabolic interconversions, estradiol is the principal intracellular human estrogen and is substantially more potent than its metabolites, estrone and estriol at the receptor level. ... Estrogens act through binding to nuclear receptors in estrogen-responsive tissues. To date, two estrogen receptors have been identified. These vary in proportion from tissue to tissue. Circulating estrogens modulate the pituitary secretion of the gonadotropins, luteinizing hormone (LH) and follicle stimulating hormone (FSH), through a negative feedback mechanism. Estrogens act to reduce the elevated levels of these hormones seen in postmenopausal women.
US Natl Inst Health; DailyMed. Current Medication Information EEMT H.S. (estrone sodium sulfate and methyltestosterone) tablet, coated (May 2010). Available from, as of February 28, 2011: https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?id=23251
Estrogens have an important role in the reproductive, skeletal, cardiovascular, and central nervous systems in women, and act principally by regulating gene expression. Biologic response is initiated when estrogen binds to a ligand-binding domain of the estrogen receptor resulting in a conformational change that leads to gene transcription through specific estrogen response elements (ERE) of target gene promoters; subsequent activation or repression of the target gene is mediated through 2 distinct transactivation domains (ie, AF-1 and AF-2) of the receptor. The estrogen receptor also mediates gene transcription using different response elements (ie, AP-1) and other signal pathways. Recent advances in the molecular pharmacology of estrogen and estrogen receptors have resulted in the development of selective estrogen receptor modulators (eg, clomiphene, raloxifene, tamoxifen, toremifene), agents that bind and activate the estrogen receptor but that exhibit tissue-specific effects distinct from estrogen. Tissue-specific estrogen-agonist or -antagonist activity of these drugs appears to be related to structural differences in their estrogen receptor complex (eg, specifically the surface topography of AF-2 for raloxifene) compared with the estrogen (estradiol)-estrogen receptor complex. A second estrogen receptor also has been identified, and existence of at least 2 estrogen receptors (ER-alpha, ER-beta) may contribute to the tissue-specific activity of selective modulators. While the role of the estrogen receptor in bone, cardiovascular tissue, and the CNS continues to be studied, emerging evidence indicates that the mechanism of action of estrogen receptors in these tissues differs from the manner in which estrogen receptors function in reproductive tissue. /Estrogen General Statement/
American Society of Health System Pharmacists; AHFS Drug Information 2010. Bethesda, MD. (2010), p. 3130
Intracellular cytosol-binding proteins for estrogens have been identified in estrogen-responsive tissues including the female genital organs, breasts, pituitary, and hypothalamus. The estrogen-binding protein complex (ie, cytosol-binding protein and estrogen) distributes into the cell nucleus where it stimulates DNA, RNA, and protein synthesis. The presence of these receptor proteins is responsible for the palliative response to estrogen therapy in women with metastatic carcinoma of the breast. /Estrogen General Statement/
American Society of Health System Pharmacists; AHFS Drug Information 2010. Bethesda, MD. (2010), p. 3130
Estrogens have generally favorable effects on blood cholesterol and phospholipid concentrations. Estrogens reduce LDL-cholesterol and increase HDL-cholesterol concentrations in a dose-related manner. The decrease in LDL-cholesterol concentrations associated with estrogen therapy appears to result from increased LDL catabolism, while the increase in triglyceride concentrations is caused by increased production of large, triglyceride-rich, very-low-density lipoproteins (VLDLs); changes in serum HDL-cholesterol concentrations appear to result principally from an increase in the cholesterol and apolipoprotein A-1 content of HDL2- and a slight increase in HDL3-cholesterol. /Estrogen General Statement/
American Society of Health System Pharmacists; AHFS Drug Information 2010. Bethesda, MD. (2010), p. 3130
For more Mechanism of Action (Complete) data for ESTRONE (7 total), please visit the HSDB record page.
Date of Issue : 2022-06-30
Valid Till : 2025-07-02
Written Confirmation Number : WC-0161A2
Address of the Firm :

NDC Package Code : 22552-0022
Start Marketing Date : 2011-05-13
End Marketing Date : 2026-12-31
Dosage Form (Strength) : POWDER (1g/g)
Marketing Category : BULK INGREDIENT

Date of Issue : 2022-08-31
Valid Till : 2025-07-02
Written Confirmation Number : WC-0162Amended
Address of the Firm :








 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
Related Excipient Companies

Excipients by Applications
REF. STANDARDS & IMPURITIES

ANALYTICAL

ABOUT THIS PAGE
33
PharmaCompass offers a list of Estrone API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Estrone manufacturer or Estrone supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Estrone manufacturer or Estrone supplier.
A Estrone manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Estrone, including repackagers and relabelers. The FDA regulates Estrone manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Estrone API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
click here to find a list of Estrone manufacturers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PhamaCompass.
A Estrone supplier is an individual or a company that provides Estrone active pharmaceutical ingredient (API) or Estrone finished formulations upon request. The Estrone suppliers may include Estrone API manufacturers, exporters, distributors and traders.
click here to find a list of Estrone suppliers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PharmaCompass.
A Estrone written confirmation (Estrone WC) is an official document issued by a regulatory agency to a Estrone manufacturer, verifying that the manufacturing facility of a Estrone active pharmaceutical ingredient (API) adheres to the Good Manufacturing Practices (GMP) regulations of the importing country. When exporting Estrone APIs or Estrone finished pharmaceutical products to another nation, regulatory agencies frequently require a Estrone WC (written confirmation) as part of the regulatory process.
click here to find a list of Estrone suppliers with Written Confirmation (WC) on PharmaCompass.
National Drug Code is a comprehensive database maintained by the FDA that contains information on all drugs marketed in the US. This directory includes information about finished drug products, unfinished drug products, and compounded drug products, including those containing Estrone as an active pharmaceutical ingredient (API).
The FDA updates the NDC directory daily. The NDC numbers for Estrone API and other APIs are published in this directory by the FDA.
The NDC unfinished drugs database includes product listing information submitted for all unfinished drugs, such as active pharmaceutical ingredients (APIs), drugs intended for further processing and bulk drug substances for compounding.
Pharmaceutical companies that manufacture Estrone as an active pharmaceutical ingredient (API) must furnish the FDA with an updated record of all drugs that they produce, prepare, propagate, compound, or process for commercial distribution in the US at their facilities.
The NDC directory also contains data on finished compounded human drug products that contain Estrone and are produced by outsourcing facilities. While these outsourcing facilities are not mandated to assign a Estrone NDC to their finished compounded human drug products, they may choose to do so.
click here to find a list of Estrone suppliers with NDC on PharmaCompass.
Estrone Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Estrone GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Estrone GMP manufacturer or Estrone GMP API supplier for your needs.
A Estrone CoA (Certificate of Analysis) is a formal document that attests to Estrone's compliance with Estrone specifications and serves as a tool for batch-level quality control.
Estrone CoA mostly includes findings from lab analyses of a specific batch. For each Estrone CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Estrone may be tested according to a variety of international standards, such as European Pharmacopoeia (Estrone EP), Estrone JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Estrone USP).

