
Synopsis
Synopsis
0
USDMF
0
CEP/COS
0
EU WC
0
KDMF
0
NDC API
0
VMF
0
FDA Orange Book
0
Europe
0
Canada
0
South Africa
0
Listed Dossiers
DRUG PRODUCT COMPOSITIONS
0
US Patents
0
US Exclusivities
0
Health Canada Patents
US Medicaid
NA
Annual Reports
NA
Regulatory FDF Prices
NA
0
FDF
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
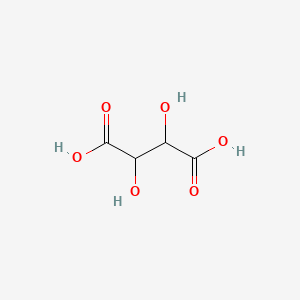

1. (r*,r*)-(+-)-2,3-dihydroxybutanedioic Acid, Monoammonium Monosodium Salt
2. Aluminum Tartrate
3. Ammonium Tartrate
4. Calcium Tartrate
5. Calcium Tartrate Tetrahydrate
6. Mn(iii) Tartrate
7. Potassium Tartrate
8. Seignette Salt
9. Sodium Ammonium Tartrate
10. Sodium Potassium Tartrate
11. Sodium Tartrate
12. Stannous Tartrate
13. Tartaric Acid, ((r*,r*)-(+-))-isomer
14. Tartaric Acid, (r*,s*)-isomer
15. Tartaric Acid, (r-(r*,r*))-isomer
16. Tartaric Acid, (s-(r*,r*))-isomer
17. Tartaric Acid, Ammonium Sodium Salt, (1:1:1) Salt, (r*,r*)-(+-)-isomer
18. Tartaric Acid, Calcium Salt, (r-r*,r*)-isomer
19. Tartaric Acid, Monoammonium Salt, (r-(r*,r*))-isomer
20. Tartrate
1. 2,3-dihydroxysuccinic Acid
2. Dl-tartaric Acid
3. 2,3-dihydroxybutanedioic Acid
4. 526-83-0
5. 133-37-9
6. Racemic Acid
7. Traubensaure
8. Racemic Tartaric Acid
9. Dl-tartrate
10. Paratartaric Acid
11. Paratartaric Aicd
12. Uvic Acid
13. Acidum Tartaricum
14. Butanedioic Acid, 2,3-dihydroxy-
15. Resolvable Tartaric Acid
16. Nsc62778
17. Tartaric Acid D,l
18. E-7050 (2s,3s)-2,3-dihydroxysuccinic Acid
19. Tartaric Acid, L-(+)-
20. 2,3-dihydroxy-succinic Acid
21. Baros
22. Chebi:15674
23. Dl-2,3-dihydroxybutanedioic Acid
24. (2rs,3rs)-tartaric Acid
25. Tartrate
26. 1007601-97-9
27. Nsc 148314
28. Butanedioic Acid, 2,3-dihydroxy-, (2r,3r)-rel-
29. Butanedioic Acid, 2,3-dihydroxy-(r*,r*)-(.+/-.)-
30. Butanedioic Acid, 2,3-dihydroxy-, (r*,r*)-
31. 868-14-4
32. (.+-.)-tartaric Acid
33. (2r,3r)-2,3-dihydroxybernsteinsaeure
34. Tartaric Acid (van)
35. Kyselina Vinna [czech]
36. Nsc155080
37. Tartaric Acid [usan:jan]
38. D-alpha,beta-dihydroxysuccinic Acid
39. Nsc-148314
40. Kyselina 2,3-dihydroxybutandiova [czech]
41. (+) Tartaric Acid
42. (-) Tartaric Acid
43. 1,2-dihydroxyethane-1,2-dicarboxylic Acid
44. Ai3-06298
45. 1,2-dicarboxylic Acid
46. Wln: Qvyqyqvq
47. (-) D-tartaric Acid
48. Sal Tartar (salt/mix)
49. Tartaric Acid, (dl)-
50. Butanedioic Acid, 2,3-dihydroxy- (r-(r*,r*))-
51. Butanedioic Acid, 2,3-dihydroxy-, [s-(r*,r*)]-
52. Malic Acid, 3-hydroxy-
53. Laevo-(+)-tartaric Acid
54. 2,3-dihydroxysuccinicacid
55. Dextro,laevo-tartaric Acid
56. Succinic Acid,3-dihydroxy
57. Schembl848
58. Bmse000167
59. Succinic Acid,3-dihydroxy-
60. (.+/-.)-tartaric Acid
61. Dsstox_cid_26986
62. Dsstox_rid_82036
63. Dsstox_gsid_46986
64. Oprea1_827092
65. Tartaric Acid, (l)
66. Tartaric Acid, (.+-.)-
67. Butanedioic Acid,3-dihydroxy-
68. Chembl333714
69. Dihydroxysuccinic Acid, (dl)-
70. Tartaric Acid, (.+/-.)-
71. L+tartaric Acid Fcc, Nf, Usp
72. Dtxsid501031477
73. Hms3370m15
74. (+)-2,3-dihydroxybutanedioic Acid
75. Bcp14303
76. Tox21_302052
77. Bbl011588
78. Mfcd00064206
79. Nsc133735
80. Nsc148314
81. Nsc608773
82. S2997
83. Stk387106
84. 2,3-dihydroxysuccinic Acid, (dl)-
85. 3-carboxy-2,3-dihydroxypropanoic Acid
86. Akos000120086
87. Akos016844048
88. Nsc-133735
89. Nsc-608773
90. Sb44180
91. Sb44181
92. Smp2_000051
93. D-.alpha.,.beta.-dihydroxysuccinic Acid
94. Ncgc00256063-01
95. Ncgc00347131-03
96. As-10983
97. Cas-133-37-9
98. Nci60_001102
99. (+)-2,3-dihydroxy-1,4-butanedioic Acid
100. Db-016129
101. Db-016159
102. Db-042899
103. Am20110247
104. Cs-0022654
105. Ft-0624346
106. Ft-0625514
107. Ft-0628018
108. Ft-0628243
109. Ft-0656080
110. Ft-0772946
111. Ft-0773804
112. T0001
113. (+/-)-2,3-dihydroxy-1,4-butanedioic Acid
114. A22866
115. Butanedioic Acid,3-dihydroxy- [r-(r*,r*)]-
116. 133d379
117. A829202
118. Q194322
119. Butanedioic Acid,3-dihydroxy-, (r*,r*)-(.+-.)-
120. F2191-0230
121. Z1258943354
122. 1,2-dihydroxyethane-1,2-dicarboxylic Acid;2,3-dihydrosuccinic Acid
123. (2s,3s)-(-)-tartaric Acid; D(-)-threaric Acid;d(-)-dihydroxysuccinic Acid
124. Copper, Mixt. With [r-(r*,r*)]-2,3-dihydroxybutanedioic Acid Monopotassium Salt
| Molecular Weight | 150.09 g/mol |
|---|---|
| Molecular Formula | C4H6O6 |
| XLogP3 | -1.9 |
| Hydrogen Bond Donor Count | 4 |
| Hydrogen Bond Acceptor Count | 6 |
| Rotatable Bond Count | 3 |
| Exact Mass | 150.01643791 g/mol |
| Monoisotopic Mass | 150.01643791 g/mol |
| Topological Polar Surface Area | 115 Ų |
| Heavy Atom Count | 10 |
| Formal Charge | 0 |
| Complexity | 134 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 2 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
Stress incontinence, female



Registration Number : 217MF10397
Registrant's Address : 1-1-2 Nishinomiyahama, Nishinomiya City, Hyogo Prefecture
Initial Date of Registration : 2005-08-10
Latest Date of Registration :




 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
Market Place

REF. STANDARDS & IMPURITIES

ABOUT THIS PAGE
29
PharmaCompass offers a list of DL-Tartaric acid API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right DL-Tartaric acid manufacturer or DL-Tartaric acid supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred DL-Tartaric acid manufacturer or DL-Tartaric acid supplier.
A DL-Tartaric acid manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of DL-Tartaric acid, including repackagers and relabelers. The FDA regulates DL-Tartaric acid manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. DL-Tartaric acid API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
click here to find a list of DL-Tartaric acid manufacturers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PhamaCompass.
A DL-Tartaric acid supplier is an individual or a company that provides DL-Tartaric acid active pharmaceutical ingredient (API) or DL-Tartaric acid finished formulations upon request. The DL-Tartaric acid suppliers may include DL-Tartaric acid API manufacturers, exporters, distributors and traders.
click here to find a list of DL-Tartaric acid suppliers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PharmaCompass.
The Pharmaceuticals and Medical Devices Agency (PMDA) established the Japan Drug Master File (JDMF), also known as the Master File (MF), to permit Japanese and foreign manufacturers of drug substances, intermediates, excipients, raw materials, and packaging materials (‘Products’) to voluntarily register confidential information about the production and management of their products in Japan.
The DL-Tartaric acid Drug Master File in Japan (DL-Tartaric acid JDMF) empowers DL-Tartaric acid API manufacturers to present comprehensive information (e.g., production methods, data, etc.) to the review authority, i.e., PMDA (Pharmaceuticals & Medical Devices Agency).
PMDA reviews the DL-Tartaric acid JDMF during the approval evaluation for pharmaceutical products. At the time of DL-Tartaric acid JDMF registration, PMDA checks if the format is accurate, if the necessary items have been included (application), and if data has been attached.
click here to find a list of DL-Tartaric acid suppliers with JDMF on PharmaCompass.
DL-Tartaric acid Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of DL-Tartaric acid GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right DL-Tartaric acid GMP manufacturer or DL-Tartaric acid GMP API supplier for your needs.
A DL-Tartaric acid CoA (Certificate of Analysis) is a formal document that attests to DL-Tartaric acid's compliance with DL-Tartaric acid specifications and serves as a tool for batch-level quality control.
DL-Tartaric acid CoA mostly includes findings from lab analyses of a specific batch. For each DL-Tartaric acid CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
DL-Tartaric acid may be tested according to a variety of international standards, such as European Pharmacopoeia (DL-Tartaric acid EP), DL-Tartaric acid JP (Japanese Pharmacopeia) and the US Pharmacopoeia (DL-Tartaric acid USP).

