
Synopsis
0
CEP/COS
0
JDMF
0
EU WC
0
NDC API
0
VMF
0
FDA Orange Book
0
Canada
0
Australia
0
South Africa
DRUG PRODUCT COMPOSITIONS
0
US Patents
0
US Exclusivities
0
Health Canada Patents
Annual Reports
NA
Regulatory FDF Prices
NA
0
FDF
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
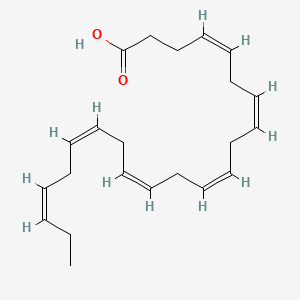

1. Acid, Docosahexaenoic
2. Acids, Docosahexaenoic
3. Acids, Docosahexenoic
4. Docosahexaenoate
5. Docosahexaenoic Acid (all-z Isomer)
6. Docosahexaenoic Acid Dimer (all-z Isomer)
7. Docosahexaenoic Acid, 3,6,9,12,15,18-isomer
8. Docosahexaenoic Acid, 4,7,10,13,16,19-(all-z-isomer)
9. Docosahexaenoic Acid, 4,7,10,13,16,19-(all-z-isomer), Cerium Salt
10. Docosahexaenoic Acid, 4,7,10,13,16,19-(all-z-isomer), Cesium Salt
11. Docosahexaenoic Acid, 4,7,10,13,16,19-(all-z-isomer), Potassium Salt
12. Docosahexaenoic Acid, 4,7,10,13,16,19-(z,z,z,z,z,e-isomer)
13. Docosahexaenoic Acid, 4,7,10,13,16,19-isomer
14. Docosahexaenoic Acid, 4,7,10,13,16,19-isomer, Sodium Salt
15. Docosahexaenoic Acid, Sodium Salt
16. Docosahexaenoic Acids
17. Docosahexenoic Acids
1. Doconexent
2. Cervonic Acid
3. 6217-54-5
4. Cis-4,7,10,13,16,19-docosahexaenoic Acid
5. Docosahexaenoate
6. (4z,7z,10z,13z,16z,19z)-docosa-4,7,10,13,16,19-hexaenoic Acid
7. All-cis-dha
8. Doconexento
9. Doconexentum
10. Doxonexent
11. Aquagrow Advantage
12. All-z-docosahexaenoic Acid
13. Doconexent [inn]
14. Martek Dha Hm
15. Ropufa 60
16. All-cis-4,7,10,13,16,19-docosahexaenoic Acid
17. (4z,7z,10z,13z,16z,19z)-docosahexaenoic Acid
18. Docosaheaenoic-acid
19. Docosahexaenoic Acid (all-z)
20. Ccris 7670
21. All-cis-docosa-4,7,10,13,16,19-hexaenoic Acid
22. Zad9okh9jc
23. Docosahexanoic Acid
24. (all-z)-4,7,10,13,16,19-docosahexaenoic Acid
25. Docosa-4,7,10,13,16,19-hexaenoic Acid
26. (4z,7z,10z,13z,16z,19z)-4,7,10,13,16,19-docosahexaenoic Acid
27. Chembl367149
28. Docosahexaenoic Acid(dha)
29. Chebi:28125
30. Delta4,7,10,13,16,19-docosahexaenoic Acid
31. 4-cis,7-cis,10-cis,13-cis,16-cis,19-cis-docosahexaenoic Acid
32. 4z,7z,10z,13z,16z,19z-docosahexaenoic Acid
33. Docosahexaenoic Acid (c22:6 N3)
34. Omega-3 Marine Triglycerides
35. Mfcd00065722
36. 4,7,10,13,16,19-docosahexaenoic Acid, (all-z)-
37. Fa 22:6
38. Efalex
39. Ncgc00161345-04
40. C22:6 (n-3)
41. Docosahexaenoic Acid (22:6 N-3)
42. 22:6 N-3
43. 22:6(n-3)
44. Cis-4,7,10,13,16,19-docosahexanoic Acid
45. 4,7,10,13,16,19-docosahexaenoic Acid
46. C22:6n-3,6,9,12,15,18
47. Monolife 50
48. Marinol D 50tg
49. Sr-05000002130
50. Unii-zad9okh9jc
51. Doconexentum [inn-latin]
52. Doconexento [inn-spanish]
53. Dha-[21,21,22,22,22-d5]
54. Cervonate
55. Dtxsid5040465
56. 1fdq
57. Algal Dha
58. Docohexanenoic Acid
59. Docosahexanenoic Acid
60. Omega 3 Fatty Acid
61. All-z-docosahexaenoate
62. Spectrum5_002062
63. Docosahexaenoic Acid (6ci)
64. Dsstox_cid_20465
65. Dsstox_rid_79498
66. Dsstox_gsid_40465
67. Schembl19577
68. Bspbio_001298
69. Docoshexaenoic Acid (powder)
70. Mls004773950
71. Bml3-b02
72. Gtpl1051
73. Retriacyl (proposed Trade Name)
74. Bcbcmap01_000145
75. Docosahexaenoic Acid [mi]
76. Hms1361a20
77. Hms1791a20
78. Hms1989a20
79. Hms3402a20
80. Hms3649j15
81. Docosahexaenoic Acid [inci]
82. Hy-b2167
83. Zinc4474564
84. Docosahexaenoic Acid [vandf]
85. Tox21_111992
86. Bdbm50210259
87. Docosahexaenoic Acid [mart.]
88. Lmfa01030185
89. Docosahexaenoic Acid [usp-rs]
90. Docosahexaenoic Acid [who-dd]
91. Akos015962159
92. Ac-1010
93. Ccg-207958
94. Ccg-208135
95. Cs-6261
96. Db03756
97. Kl-0761
98. Idi1_033768
99. 4,7,10,13,16,19-docosahexaenoate
100. Ncgc00161345-01
101. Ncgc00161345-02
102. Ncgc00161345-03
103. Ncgc00161345-05
104. Ncgc00161345-07
105. All Cis- Docosahexaenoic Acid (cis-dha)
106. Smr001881493
107. Cas-6217-54-5
108. Cis-4,7,10,13,16,19-docosahexanoate
109. D2226
110. S6454
111. C06429
112. H10987
113. Ab01563379_01
114. Docosahexaenoic Acid (dha) (c22:6 N3)
115. Docosahexaenoic Acid-rich Single Cell Oil
116. 217d545
117. Q423345
118. Sr-05000002130-1
119. Sr-05000002130-4
120. Brd-k39965020-001-02-6
121. 4,7,10,13,16,19-docosahexaenoic Acid, (all Cis)-
122. A320050000
123. Cis-4,7,10,13,16,19-docosahexaenoic Acid, >=98%
124. Fa(22:6(4z,7z,10z,13z,16z,19z))
125. Z,z,z,z,z,z-docosa-4,7,10,13,16,19-hexaenoic Acid
126. (all-z)-4,7,10,13,16,19-docosahexaenoic Acid, Dha
127. 800e8e72-bbf4-46f7-a60b-b8f2b54669c7
128. C22h32o2 (cis-4,7,10,13,16,19-docosahexaenoic Acid)
129. 4,7,10,13,16,19-docosahexaenoic Acid, (all-z)- (8ci)
130. Cis-4,7,10,13,16,19-docosahexaenoic Acid, Analytical Standard
131. Docosa-4z,7z,10z,13z,16z,19z-hexaenoic Acid (22:6, N-3)
132. (4z,7z,10z,13z,16z, 19z)-docosa-4,7,10,13,16,19-hexaenoic Acid
133. (4z,7z,10z,13z,16z,19z)-docosa-4, 7,10,13,16,19-hexaenoic Acid
134. (4z,7z,10z,13z,16z,19z)-docosa-4,7,10,13,16,19- Hexaenoic Acid
135. Cis-4,7,13,16,19-docosahexaenoic Acid (stabilized With Vitamine E)
136. 4,7,10,13,16,19-docosahexaenoic Acid, (4z,7z,10z,13z,16z,19z)- (9ci)
137. Docosahexaenoic Acid (dha) (c22:6) (constituent Of Krill Oil) [dsc]
138. 1024594-51-1
139. Cis-4,7,10,13,16,19-docosahexaenoic Acid, 500 Mug/ml In Ethanol, Certified Reference Material
| Molecular Weight | 328.5 g/mol |
|---|---|
| Molecular Formula | C22H32O2 |
| XLogP3 | 6.2 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 2 |
| Rotatable Bond Count | 14 |
| Exact Mass | 328.240230259 g/mol |
| Monoisotopic Mass | 328.240230259 g/mol |
| Topological Polar Surface Area | 37.3 Ų |
| Heavy Atom Count | 24 |
| Formal Charge | 0 |
| Complexity | 462 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 6 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
Used as a high-docosahexaenoic acid (DHA) oral supplement.
Treatment of Retinitis Pigmentosa
DHA in the central nervous system is found in the phospholipid bilayers where it modulates the physical environment and increase the free volume within the membrane bilayer. It influences the G-protein coupled receptor activity and affects transmembrane transport and cell interaction with the exterior world. It is also reported to promote apoptosis, neuronal differentiation and ion channel activity. Like other polyunsaturated fatty acids, DHA acts as a ligand at PPARs that plays an anti-inflammatory effect and regulate inflammatory gene expression and NFB activation. DHA also gives rise to resolvins and related compounds (e.g., protectins) through pathways involving cyclooxygenase and lipoxygenase enzymes to resolve the inflammatory responses.
Absorption
Like other omega-3 fatty acids, DHA is hydrolyzed from the intestines and delivered through the lymphatic circulation. Plasma DHA concentrations increase in a dose-dependent and saturable manner.
Volume of Distribution
DHA is the most abundant n3 fatty acid in membranes and is present in all organs. It is also the most variable among organs and is particularly abundant in neural tissue, such as brain and retina, where it is several hundred-fold more abundant than EPA.
DHA can be metabolized into DHA-derived specialized pro-resolving mediators (SPMs), DHA epoxides, electrophilic oxo-derivatives (EFOX) of DHA, neuroprostanes, ethanolamines, acylglycerols, docosahexaenoyl amides of amino acids or neurotransmitters, and branched DHA esters of hydroxy fatty acids, among others. It is converted to 17-hydroperoxy-DHA derivatives via COX-2 and 15-LOX and 5-LOX activity. These derivatives are further converted into D-series resolvins and protectins with potent anti-inflammatory potential and potent neuroprotective effect. DHA may also be metabolized to 19,20-epoxydocosapentaenoic acids (EDPs) and isomers via CYP2C9 activity. Epoxy metabolites are reported to mediate anti-tumor activity by inhibiting angiogenesis, tumor growth, and metastasis.
Approximately 20 hours.
DHA and its conversion to other lipid signalling moleccules compete with the arachidonic acid cascade from endogenous phospholipids and shift the inflammatory state to being more anti-inflammatory. DHA inhibits endotoxin-stimulated production of IL-6 and IL-8 in human endothelial cells. Derivatives of DHA are anti-inflammatory lipid mediators. Lipid mediators resolvin D1 and protectin D1 all inhibit transendothelial migration of neutrophils, so preventing neutrophilic infiltration at sites of inflammation, resolvin D1 inhibits IL-1 production, and protectin D1 inhibits TNF and IL-1 production. Monoxydroxy derivative of DHA converted by LOX inhibit thromboxane-induced platelet aggregation. DHA supplementation has also shown to reduce the levels of serum C-reactive protein (CRP) and other circulating markers of inflammation such as neutrophils in hypertriglyceridemic men. DHA acts as a ligand at peroxisome proliferator-activated receptor (PPAR) gamma and alpha that regulate lipid signalling molecule-mediated transduction pathways and modulate inflammation. As a natural ligand, DHA induces a protective effect in retinal tissues by activating retinoid x receptors and subsequent ERK/MAPK signaling pathway in photoreceptors to promote their survival and differentiation, stimulating the expression of antiapoptotic proteins such as Bcl-2 and preserving mitochondrial membrane potential.
Related Excipient Companies

Excipients by Applications
Global Sales Information

Market Place

REF. STANDARDS & IMPURITIES

ANALYTICAL

ABOUT THIS PAGE
70
PharmaCompass offers a list of DHA API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right DHA manufacturer or DHA supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred DHA manufacturer or DHA supplier.
A DHA manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of DHA, including repackagers and relabelers. The FDA regulates DHA manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. DHA API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
click here to find a list of DHA manufacturers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PhamaCompass.
A DHA supplier is an individual or a company that provides DHA active pharmaceutical ingredient (API) or DHA finished formulations upon request. The DHA suppliers may include DHA API manufacturers, exporters, distributors and traders.
click here to find a list of DHA suppliers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PharmaCompass.
A DHA DMF (Drug Master File) is a document detailing the whole manufacturing process of DHA active pharmaceutical ingredient (API) in detail. Different forms of DHA DMFs exist exist since differing nations have different regulations, such as DHA USDMF, ASMF (EDMF), JDMF, CDMF, etc.
A DHA DMF submitted to regulatory agencies in the US is known as a USDMF. DHA USDMF includes data on DHA's chemical properties, information on the facilities and procedures used, and details about packaging and storage. The DHA USDMF is kept confidential to protect the manufacturer’s intellectual property.
click here to find a list of DHA suppliers with USDMF on PharmaCompass.
In Korea, the Ministry of Food and Drug Safety (MFDS) is in charge of regulating pharmaceutical products and services.
Pharmaceutical companies submit a DHA Drug Master File in Korea (DHA KDMF) to the MFDS, which includes comprehensive information about the production, processing, facilities, materials, packaging, and testing of DHA. The MFDS reviews the DHA KDMF as part of the drug registration process and uses the information provided in the DHA KDMF to evaluate the safety and efficacy of the drug.
After submitting a DHA KDMF to the MFDS, the registered manufacturer can provide importers or distributors with the registration number without revealing confidential information to Korean business partners. Applicants seeking to register their DHA API can apply through the Korea Drug Master File (KDMF).
click here to find a list of DHA suppliers with KDMF on PharmaCompass.
DHA Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of DHA GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right DHA GMP manufacturer or DHA GMP API supplier for your needs.
A DHA CoA (Certificate of Analysis) is a formal document that attests to DHA's compliance with DHA specifications and serves as a tool for batch-level quality control.
DHA CoA mostly includes findings from lab analyses of a specific batch. For each DHA CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
DHA may be tested according to a variety of international standards, such as European Pharmacopoeia (DHA EP), DHA JP (Japanese Pharmacopeia) and the US Pharmacopoeia (DHA USP).

