
Synopsis
Synopsis
0
EU WC
0
KDMF
0
FDA Orange Book
DRUG PRODUCT COMPOSITIONS
0
US Patents
0
US Exclusivities
0
Health Canada Patents
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers

1. Dextran
2. Dextran 40000
3. Dextran 70
4. Dextran 75
5. Dextran 80
6. Dextran B 1355
7. Dextran B 1355 S
8. Dextran B-1355
9. Dextran B-1355-s
10. Dextran B1355
11. Dextran B512
12. Dextran Derivatives
13. Dextran M 70
14. Dextran T 40
15. Dextran T 500
16. Dextran T 70
17. Dextran T-40
18. Dextran T-500
19. Dextrans
20. Hemodex
21. Hyskon
22. Infukoll
23. Macrodex
24. Polyglucin
25. Promit
26. Rheodextran
27. Rheoisodex
28. Rheomacrodex
29. Rheopolyglucin
30. Rondex
31. Saviosol
1. L3l3xyp7mp
2. Lmwd
3. Schembl206877
4. Chembl1697742
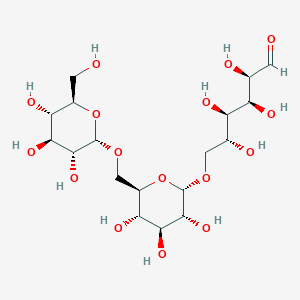
5. O-alpha-d-glucopyranosyl-(1.6)-o-alpha-d-glucopyranosyl-(1.6)-d-glucose
6. Hy-n0913a
7. Dtxsid701317190
8. 6-.alpha.-isomaltosylglucose
9. Zinc64622163
10. Cs-0109495
11. D-glucose, O-.alpha.-d-glucopyranosyl-(1->6)-o-.alpha.-d-glucopyranosyl-(1->6)-
12. O-alpha-d-glucopyranosyl-(1-->6)-o-alpha-d-glucopyranosyl-(1-->6)-d-glucose
13. Wurcs=2.0/2,3,2/[o2122h][a2122h-1a_1-5]/1-2-2/a6-b1_b6-c1
14. (2r,3s,4r,5r)-2,3,4,5-tetrahydroxy-6-(((2s,3r,4s,5s,6r)-3,4,5-trihydroxy-6-((((2s,3r,4s,5s,6r)-3,4,5-trihydroxy-6-(hydroxymethyl)oxan-2-yl)oxy)methyl)oxan-2-yl)oxy)hexanal
| Molecular Weight | 504.4 g/mol |
|---|---|
| Molecular Formula | C18H32O16 |
| XLogP3 | -7.2 |
| Hydrogen Bond Donor Count | 11 |
| Hydrogen Bond Acceptor Count | 16 |
| Rotatable Bond Count | 11 |
| Exact Mass | 504.16903493 g/mol |
| Monoisotopic Mass | 504.16903493 g/mol |
| Topological Polar Surface Area | 277 Ų |
| Heavy Atom Count | 34 |
| Formal Charge | 0 |
| Complexity | 625 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 14 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
Anticoagulants
Agents that prevent BLOOD CLOTTING. (See all compounds classified as Anticoagulants.)
Plasma Substitutes
Any liquid used to replace blood plasma, usually a saline solution, often with serum albumins, dextrans or other preparations. These substances do not enhance the oxygen- carrying capacity of blood, but merely replace the volume. They are also used to treat dehydration. (See all compounds classified as Plasma Substitutes.)
GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 1183
Submission : 1968-05-07
Status : Inactive
Type : II

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 1184
Submission : 1968-05-07
Status : Inactive
Type : II

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 19584
Submission : 2006-06-30
Status : Active
Type : II

USDMF
GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 462
Submission : 1962-01-08
Status : Inactive
Type : II

Portfolio PDF
Product Web Link
Virtual Booth
Digital Content
Website
Corporate PDF
USDMF
GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 9447
Submission : 1991-12-05
Status : Inactive
Type : II

Portfolio PDF
Product Web Link
Virtual Booth
Digital Content
Website
Corporate PDF
USDMF
GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 353
Submission : 1959-11-16
Status : Inactive
Type : II

Portfolio PDF
Product Web Link
Virtual Booth
Digital Content
Website
Corporate PDF
USDMF
GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 2573
Submission : 1975-12-03
Status : Inactive
Type : II

Portfolio PDF
Product Web Link
Virtual Booth
Digital Content
Website
Corporate PDF
 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results] Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.

Certificate Number : R1-CEP 2003-165 - Rev 01
Status : Valid
Issue Date : 2022-01-03
Type : Chemical
Substance Number : 1506

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.

Certificate Number : R1-CEP 1999-065 - Rev 03
Status : Valid
Issue Date : 2022-01-03
Type : Chemical
Substance Number : 1001

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.

Certificate Number : R1-CEP 1999-064 - Rev 03
Status : Valid
Issue Date : 2022-01-03
Type : Chemical
Substance Number : 1000

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.

Certificate Number : R1-CEP 1999-063 - Rev 03
Status : Valid
Issue Date : 2022-01-03
Type : Chemical
Substance Number : 999

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results] Registration Number : 217MF11169
Registrant's Address : 41, 2-chome, Sasazuka-cho, Nishi-ku, Nagoya City, Aichi Prefecture
Initial Date of Registration : 2005-12-12
Latest Date of Registration : 2018-10-01

Dextran sulfate sodium sulfur 18
Registration Number : 218MF10826
Registrant's Address : 2-41 Sasazuka-cho, Nishi-ku, Nagoya City, Aichi Prefecture
Initial Date of Registration : 2006-10-20
Latest Date of Registration : 2006-10-20

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]NDC Package Code : 49442-5001
Start Marketing Date : 2011-11-01
End Marketing Date : 2026-12-31
Dosage Form (Strength) : POWDER (100g/100g)
Marketing Category : DRUG FOR FURTHER PROCESSING

NDC Package Code : 49442-5600
Start Marketing Date : 2015-07-01
End Marketing Date : 2026-12-31
Dosage Form (Strength) : POWDER (1g/g)
Marketing Category : BULK INGREDIENT

NDC Package Code : 49442-5040
Start Marketing Date : 2006-09-01
End Marketing Date : 2026-12-31
Dosage Form (Strength) : POWDER (1g/g)
Marketing Category : BULK INGREDIENT

NDC Package Code : 49442-5070
Start Marketing Date : 2011-11-01
End Marketing Date : 2026-12-31
Dosage Form (Strength) : POWDER (100g/100g)
Marketing Category : DRUG FOR FURTHER PROCESSING

NDC Package Code : 49442-5800
Start Marketing Date : 2015-06-01
End Marketing Date : 2026-12-31
Dosage Form (Strength) : POWDER (1g/g)
Marketing Category : BULK INGREDIENT

NDC Package Code : 49442-5060
Start Marketing Date : 2011-11-01
End Marketing Date : 2026-12-31
Dosage Form (Strength) : POWDER (100g/100g)
Marketing Category : DRUG FOR FURTHER PROCESSING

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]VMF Number : 336
Submission : 1959-05-18
Status : Active
Type : II

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
 SWATI - Transforming science into solutions with 60+ years of expertise, global accreditations, and pioneering biotech innovation.
SWATI - Transforming science into solutions with 60+ years of expertise, global accreditations, and pioneering biotech innovation.
About the Company : Swati Spentose Pvt. Ltd. (SSPL), part of the 60-year-old V Group, is a globally trusted pharmaceutical manufacturer supplying to 70+ countries. We specialize in high-quality APIs a...
About the Company : PT. Cahaya Mandiri Farma is a leading company in the pharmaceutical industry, dedicated to producing high-quality healthcare products for humans. We are committed to excellence thr...

About the Company : Xi'an Wanlong Pharmaceutical Co., Ltd was founded in 1995 and it is comprehensive modern pharmaceutical enterprise which mainly produces formulations, API and intermediates, GMP & ...

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results] Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Details:
OcuMension will acquire outright, Tears Natural Forte, Bion Tears, Alcaine, Fluorescite, and Cyclogyl, in the Chinese market and AR-15512, a novel topical drug candidate for dry eye.
Lead Product(s): HYDROXYPROPYLMETHYLCELLULOSE,Dextrans,Glycerine
Therapeutic Area: Ophthalmology Brand Name: Tears Naturale Forte
Study Phase: Approved FDFProduct Type: Carbohydrate
Sponsor: OcuMension Therapeutics
Deal Size: Undisclosed Upfront Cash: Undisclosed
Deal Type: Acquisition August 12, 2024

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Lead Product(s) : HYDROXYPROPYLMETHYLCELLULOSE,Dextrans,Glycerine
Therapeutic Area : Ophthalmology
Highest Development Status : Approved FDF
Partner/Sponsor/Collaborator : OcuMension Therapeutics
Deal Size : Undisclosed
Deal Type : Acquisition
OcuMension Acquires Alcon Eye Drops with 16.7% Equity, Strategic Partnership Begins
Details : OcuMension will acquire outright, Tears Natural Forte, Bion Tears, Alcaine, Fluorescite, and Cyclogyl, in the Chinese market and AR-15512, a novel topical drug candidate for dry eye.
Product Name : Tears Naturale Forte
Product Type : Carbohydrate
Upfront Cash : Undisclosed
August 12, 2024

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Details:
ILB® to inhibit infection of human cells by four serotypes of Dengue virus (DENV1-4), two strains of Zika virus (African and Asian) and Yellow Fever virus (vaccine strain YF17D) assessed by immunofluorescence of viral particles.
Lead Product(s): LMW-Dextran Sulfate,Dextrans,Inapplicable
Therapeutic Area: Infections and Infectious Diseases Brand Name: ILB
Study Phase: PreclinicalProduct Type: Miscellaneous
Sponsor: Undisclosed
Deal Size: Inapplicable Upfront Cash: Inapplicable
Deal Type: Inapplicable September 07, 2022

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Lead Product(s) : LMW-Dextran Sulfate,Dextrans,Inapplicable
Therapeutic Area : Infections and Infectious Diseases
Highest Development Status : Preclinical
Partner/Sponsor/Collaborator : Undisclosed
Deal Size : Inapplicable
Deal Type : Inapplicable
Tikomed's Drug Candidate ILB® Inhibited Infection of Human Cells by Dengue, Zika and Yellow Fever...
Details : ILB® to inhibit infection of human cells by four serotypes of Dengue virus (DENV1-4), two strains of Zika virus (African and Asian) and Yellow Fever virus (vaccine strain YF17D) assessed by immunofluorescence of viral particles.
Product Name : ILB
Product Type : Miscellaneous
Upfront Cash : Inapplicable
September 07, 2022

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Details:
These studies show that ILB® releases, re-distributes and modulates the bioactivity of endogeneous heparin-binding growth factors that target disease compromised nervous tissues to initiate a cascade of transcriptional, metabolic and immunological effects.
Lead Product(s): LMW-Dextran Sulfate,Dextrans,Inapplicable
Therapeutic Area: Neurology Brand Name: ILB
Study Phase: Phase IIProduct Type: Miscellaneous
Sponsor: Undisclosed
Deal Size: Inapplicable Upfront Cash: Inapplicable
Deal Type: Inapplicable August 31, 2022

Tikomed's Ilb® Mobilizes and Modulates Key Growth Factors That Trigger a Cascade of Neuroprotecti...
Details : These studies show that ILB® releases, re-distributes and modulates the bioactivity of endogeneous heparin-binding growth factors that target disease compromised nervous tissues to initiate a cascade of transcriptional, metabolic and immunological effec...
Product Name : ILB
Product Type : Miscellaneous
Upfront Cash : Inapplicable
August 31, 2022

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Details:
The collaboration will support TikoMed's lead drug platform candidate ILB®, a broad-spectrum small molecule drug candidate with curative potential and clinical development, regulatory and commercial strategies.
Lead Product(s): LMW-Dextran Sulfate,Dextrans,Inapplicable
Therapeutic Area: Neurology Brand Name: ILB
Study Phase: Phase IIProduct Type: Miscellaneous
Sponsor: IQVIA
Deal Size: Undisclosed Upfront Cash: Undisclosed
Deal Type: Collaboration May 05, 2022

Details : The collaboration will support TikoMed's lead drug platform candidate ILB®, a broad-spectrum small molecule drug candidate with curative potential and clinical development, regulatory and commercial strategies.
Product Name : ILB
Product Type : Miscellaneous
Upfront Cash : Undisclosed
May 05, 2022

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Details:
The data from the rodent model studying severe traumatic brain injury (sTBI), showed that a single post-injury injection of ILB® has beneficial effects on the metabolic damages caused by sTBI.
Lead Product(s): LMW-Dextran Sulfate,Dextrans,Inapplicable
Therapeutic Area: Trauma (Emergency, Injury, Surgery) Brand Name: ILB
Study Phase: PreclinicalProduct Type: Miscellaneous
Sponsor: Undisclosed
Deal Size: Inapplicable Upfront Cash: Inapplicable
Deal Type: Inapplicable September 14, 2020

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Lead Product(s) : LMW-Dextran Sulfate,Dextrans,Inapplicable
Therapeutic Area : Trauma (Emergency, Injury, Surgery)
Highest Development Status : Preclinical
Partner/Sponsor/Collaborator : Undisclosed
Deal Size : Inapplicable
Deal Type : Inapplicable
Tikomed's ILB® Restores Brain Energy Metabolism Following Severe Traumatic Brain Injury in the Ra...
Details : The data from the rodent model studying severe traumatic brain injury (sTBI), showed that a single post-injury injection of ILB® has beneficial effects on the metabolic damages caused by sTBI.
Product Name : ILB
Product Type : Miscellaneous
Upfront Cash : Inapplicable
September 14, 2020

Details:
ILB (Dextran Sulfate) is a Other Large Molecule drug candidate, which is currently being evaluated in phase II clinical studies for the treatment of Amyotrophic Lateral Sclerosis.
Lead Product(s): Dextrans,Inapplicable
Therapeutic Area: Neurology Brand Name: ILB
Study Phase: Phase IIProduct Type: Miscellaneous
Sponsor: Tikomed | University Hospital Birmingham | Neuregenix
Deal Size: Inapplicable Upfront Cash: Inapplicable
Deal Type: Inapplicable October 15, 2018

A Safety and Tolerability Study of ILB® in Patients With Amyotrophic Lateral Sclerosis (ALS)
Details : ILB (Dextran Sulfate) is a Other Large Molecule drug candidate, which is currently being evaluated in phase II clinical studies for the treatment of Amyotrophic Lateral Sclerosis.
Product Name : ILB
Product Type : Miscellaneous
Upfront Cash : Inapplicable
October 15, 2018

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Details:
ILB (Dextran Sulfate) is a Other Large Molecule drug candidate, which is currently being evaluated in phase II clinical studies for the treatment of Amyotrophic Lateral Sclerosis.
Lead Product(s): Dextrans,Inapplicable
Therapeutic Area: Neurology Brand Name: ILB
Study Phase: Phase IIProduct Type: Miscellaneous
Sponsor: Undisclosed
Deal Size: Inapplicable Upfront Cash: Inapplicable
Deal Type: Inapplicable August 03, 2018

Details : ILB (Dextran Sulfate) is a Other Large Molecule drug candidate, which is currently being evaluated in phase II clinical studies for the treatment of Amyotrophic Lateral Sclerosis.
Product Name : ILB
Product Type : Miscellaneous
Upfront Cash : Inapplicable
August 03, 2018

Details:
Dextran is a Other Small Molecule drug candidate, which is currently being evaluated in phase III clinical studies for the treatment of Brain Injuries, Traumatic.
Lead Product(s): Dextrans,Inapplicable
Therapeutic Area: Trauma (Emergency, Injury, Surgery) Brand Name: Undisclosed
Study Phase: Phase IIIProduct Type: Miscellaneous
Sponsor: Defence Research and Development Canada
Deal Size: Inapplicable Upfront Cash: Inapplicable
Deal Type: Inapplicable July 17, 2012

Lead Product(s) : Dextrans,Inapplicable
Therapeutic Area : Trauma (Emergency, Injury, Surgery)
Highest Development Status : Phase III
Partner/Sponsor/Collaborator : Defence Research and Development Canada
Deal Size : Inapplicable
Deal Type : Inapplicable
Details : Dextran is a Other Small Molecule drug candidate, which is currently being evaluated in phase III clinical studies for the treatment of Brain Injuries, Traumatic.
Product Name : Undisclosed
Product Type : Miscellaneous
Upfront Cash : Inapplicable
July 17, 2012

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results] Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info : Generic
Registration Country : South Korea
Brand Name : NUMAREN
Dosage Form : EYE DROP
Dosage Strength : 3MG; 1MG
Packaging : 15mL, 0.6mL,30ea
Approval Date :
Application Number :
Regulatory Info : Generic
Registration Country : South Korea

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Packaging : 15mL, 0.6mL,30ea
Regulatory Info : Generic
Dosage : EYE DROP
Dosage Strength : 3MG; 1MG
Brand Name : NUMAREN
Approval Date :
Application Number :
Registration Country : South Korea

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info : Generic
Registration Country : South Korea
Tetrahydrozoline; Dextrans; N-Vinyl-2-Pyrrolidone; Polyethylene Glycol Monostearate
Brand Name : CLEAR-DROP
Dosage Form : EYE DROP
Dosage Strength : 0.5MG/ML; 2MG/ML; 10...
Packaging : 15mL
Approval Date :
Application Number :
Regulatory Info : Generic
Registration Country : South Korea

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Packaging : 15mL
Regulatory Info : Generic
Tetrahydrozoline; Dextrans; N-Vinyl-2-Pyrrolidone; Polyethylene Glycol Monostearate
Dosage : EYE DROP
Dosage Strength : 0.5MG/ML; 2MG/ML; 10...
Brand Name : CLEAR-DROP
Approval Date :
Application Number :
Registration Country : South Korea

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info :
Registration Country : Turkey
Brand Name : SLEZOL-D
Dosage Form : EYE DROPS
Dosage Strength : 3MG; 1MG
Packaging : 15 ML/BOTTLE
Approval Date :
Application Number :
Regulatory Info :
Registration Country : Turkey

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Packaging : 15 ML/BOTTLE
Regulatory Info :
Dosage : EYE DROPS
Dosage Strength : 3MG; 1MG
Brand Name : SLEZOL-D
Approval Date :
Application Number :
Registration Country : Turkey

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info :
Registration Country : Turkey
Hypromellose API; Dextrans; Glycerine
Brand Name : SLEZOL FORTE
Dosage Form : EYE DROPS
Dosage Strength : 3MG; 1MG; 2MG
Packaging : 15 ML/BOTTLE
Approval Date :
Application Number :
Regulatory Info :
Registration Country : Turkey

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Packaging : 15 ML/BOTTLE
Regulatory Info :
Hypromellose API; Dextrans; Glycerine
Dosage : EYE DROPS
Dosage Strength : 3MG; 1MG; 2MG
Brand Name : SLEZOL FORTE
Approval Date :
Application Number :
Registration Country : Turkey

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]Related Excipient Companies

Dosage Form : Orodispersible Tablet
Grade : Oral, Topical
Application : Disintegrants & Superdisintegrants
Excipient Details : Glycine is used as a disintegrant in solid dosage forms such as orally disintegrating tablets and in topical formulations such as emulsions & creams.
Pharmacopoeia Ref : NA
Technical Specs : Molecular weight: 75.07 g/mol
Ingredient(s) : Glycine Excipient
 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]Dosage Form : Tablet
Grade : Oral
 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]Dosage Form : Injectable / Parenteral
Grade : Parenteral, Oral, Topical
 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]Dosage Form : Ophthalmic Solution
Grade : Biopharma Grade
Brand Name : Calcium Chloride Dihidrat...
Application : Parenteral
Excipient Details : Used as a buffering agent in liquid formulation like ophthalmics, parenterals, pharma/biopharma processes.
Pharmacopoeia Ref : On Request
Technical Specs : Low bacteria endotoxins, low bioburden (TAMC/TYMC). Customised packaging (from grs to kilograms)
Ingredient(s) : Calcium Chloride Dihidrato
Dosage Form : Injectable / Parenteral
Grade : Biopharma Grade
Brand Name : Dextrose Anhydrous
Application : Parenteral
Excipient Details : Used as an important carbon source for cell culture during upstream process. Use for vaccines, recombinant proteins, antibodies manufacturing.
Pharmacopoeia Ref : On Request
Technical Specs : Low bacteria endotoxins, low bioburden (TAMC/TYMC). Customised packaging (from grs to kilograms)
Ingredient(s) : D Glucose Excipient
Dosage Form : Injectable / Parenteral
Grade : Biopharma Grade
Application : Thickeners and Stabilizers
Excipient Details : Used as a chelating agent in cell culture media, downstream and as a chelator stabilizer in Biopharmaceutical formulations.
Pharmacopoeia Ref : On Request
Technical Specs : Low bacteria endotoxins, low bioburden (TAMC/TYMC). Customised packaging (from grs to kilograms)
Ingredient(s) : Disodium Edetate Excipient
Dosage Form : Injectable / Parenteral
Grade : Biopharma Grade
Application : Parenteral
Excipient Details : Used as an ingredient in cell culture media in upstream, as a buffer elution in downstream and as a buffering agent in formulations.
Pharmacopoeia Ref : On Request
Technical Specs : Low bacteria endotoxins, low bioburden (TAMC/TYMC). Customised packaging (from grs to kilograms)
Ingredient(s) : Glycine Excipient
Dosage Form : Injectable / Parenteral
Grade : Biopharma Grade
Brand Name : Magnesium Chloride Hexahy...
Application : Parenteral
Excipient Details : Used as a tonicity agent in cell culture in upstream process and injectable formulations.
Pharmacopoeia Ref : On Request
Technical Specs : Low bacteria endotoxins, low bioburden (TAMC/TYMC). Customised packaging (from grs to kilograms)
Ingredient(s) : Magnesium Chloride Hexahydrate Excipient
Dosage Form : Injectable / Parenteral
Grade : Biopharma Grade
Brand Name : Potassium Chloride
Application : Parenteral
Excipient Details : Source of potassium and chloride in cell culture, also used as an osmotic regulator.
Pharmacopoeia Ref : On Request
Technical Specs : Low bacteria endotoxins, low bioburden (TAMC/TYMC). Customised packaging (from grs to kilograms)
Ingredient(s) : Potassium Chloride Excipient
Dosage Form : Injectable / Parenteral
Grade : Biopharma Grade
Brand Name : Sodium Bicarbonate
Application : Parenteral
Excipient Details : Used as a buffering agent in cell culture media, purification processes and injectable formulations.
Pharmacopoeia Ref : On Request
Technical Specs : Low bacteria endotoxins, low bioburden (TAMC/TYMC). Customised packaging (from grs to kilograms)
Ingredient(s) : Sodium Bicarbonate Excipient
Dosage Form : Injectable / Parenteral
Grade : Biopharma Grade
Application : Parenteral
Excipient Details : Used as an osmolarity agent in culture media, tonicity adjuster in ophthalmics and parenterals solutions.
Pharmacopoeia Ref : On Request
Technical Specs : Low bacteria endotoxins, low bioburden (TAMC/TYMC). Customised packaging (from grs to kilograms)
Ingredient(s) : Sodium Chloride Excipient
Dosage Form : Injectable / Parenteral
Grade : Biopharma Grade
Brand Name : Trisodium Citrate
Application : Parenteral
Excipient Details : Used as a buffering agent in purification process during downstream and in the final formulation.
Pharmacopoeia Ref : On Request
Technical Specs : Low bacteria endotoxins, low bioburden (TAMC/TYMC). Customised packaging (from grs to kilograms)
Ingredient(s) : Trisodium Citrate Excipient
Dosage Form : Injectable / Parenteral
Grade : Biopharma Grade
Application : Surfactant & Foaming Agents
Excipient Details : Used as surfactant agent in cell culture media, in protein purification and in injectable formulations.
Pharmacopoeia Ref : On Request
Technical Specs : Low bacteria endotoxins, low bioburden (TAMC/TYMC). Customised packaging (from grs to kilograms)
Ingredient(s) : Polysorbate 80
 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]Excipients by Applications
Global Sales Information

TEOFARMA Srl
Dosage Form :
Dosage Strength : Cream Derm 30 G 0.122% + 32,000 I.U./100 G...
Price Per Pack (Euro) : 7.37
Published in :
Country : Italy
RX/OTC/DISCN : Class C

Portfolio PDF
Product Web Link
Virtual Booth
Digital Content
Website
Corporate PDF
 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]Market Place

REF. STANDARDS & IMPURITIES

CAS Number : n/a
Quantity Per Vial : 0.9 mg
Sale Unit : 1
Order Code : Y0000070
Batch No : 1
Price (€) : 79
Storage : +5°C ± 3°C

CAS Number : 9004-54-0
Quantity Per Vial : 50 mg
Sale Unit : 1
Order Code : D0731005
Batch No : 1
Price (€) : 79
Storage : +5°C ± 3°C

CAS Number : 9004-54-0
Quantity Per Vial : 25 mg
Sale Unit : 1
Order Code : D0731000
Batch No : 1
Price (€) : 79
Storage : +5°C ± 3°C

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]ABOUT THIS PAGE
11
PharmaCompass offers a list of Dextrans API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, Price,and more, enabling you to easily find the right Dextrans manufacturer or Dextrans supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Dextrans manufacturer or Dextrans supplier.
PharmaCompass also assists you with knowing the Dextrans API Price utilized in the formulation of products. Dextrans API Price is not always fixed or binding as the Dextrans Price is obtained through a variety of data sources. The Dextrans Price can also vary due to multiple factors, including market conditions, regulatory modifications, or negotiated pricing deals.
A DEXTRAN POWDER manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of DEXTRAN POWDER, including repackagers and relabelers. The FDA regulates DEXTRAN POWDER manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. DEXTRAN POWDER API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
click here to find a list of DEXTRAN POWDER manufacturers with USDMF, JDMF, KDMF, CEP, GMP, COA and API Price related information on PhamaCompass.
A DEXTRAN POWDER supplier is an individual or a company that provides DEXTRAN POWDER active pharmaceutical ingredient (API) or DEXTRAN POWDER finished formulations upon request. The DEXTRAN POWDER suppliers may include DEXTRAN POWDER API manufacturers, exporters, distributors and traders.
click here to find a list of DEXTRAN POWDER suppliers with USDMF, JDMF, KDMF, CEP, GMP, COA and API Price related information on PharmaCompass.
A DEXTRAN POWDER DMF (Drug Master File) is a document detailing the whole manufacturing process of DEXTRAN POWDER active pharmaceutical ingredient (API) in detail. Different forms of DEXTRAN POWDER DMFs exist exist since differing nations have different regulations, such as DEXTRAN POWDER USDMF, ASMF (EDMF), JDMF, CDMF, etc.
A DEXTRAN POWDER DMF submitted to regulatory agencies in the US is known as a USDMF. DEXTRAN POWDER USDMF includes data on DEXTRAN POWDER's chemical properties, information on the facilities and procedures used, and details about packaging and storage. The DEXTRAN POWDER USDMF is kept confidential to protect the manufacturer’s intellectual property.
click here to find a list of DEXTRAN POWDER suppliers with USDMF on PharmaCompass.
The Pharmaceuticals and Medical Devices Agency (PMDA) established the Japan Drug Master File (JDMF), also known as the Master File (MF), to permit Japanese and foreign manufacturers of drug substances, intermediates, excipients, raw materials, and packaging materials (‘Products’) to voluntarily register confidential information about the production and management of their products in Japan.
The DEXTRAN POWDER Drug Master File in Japan (DEXTRAN POWDER JDMF) empowers DEXTRAN POWDER API manufacturers to present comprehensive information (e.g., production methods, data, etc.) to the review authority, i.e., PMDA (Pharmaceuticals & Medical Devices Agency).
PMDA reviews the DEXTRAN POWDER JDMF during the approval evaluation for pharmaceutical products. At the time of DEXTRAN POWDER JDMF registration, PMDA checks if the format is accurate, if the necessary items have been included (application), and if data has been attached.
click here to find a list of DEXTRAN POWDER suppliers with JDMF on PharmaCompass.
A DEXTRAN POWDER CEP of the European Pharmacopoeia monograph is often referred to as a DEXTRAN POWDER Certificate of Suitability (COS). The purpose of a DEXTRAN POWDER CEP is to show that the European Pharmacopoeia monograph adequately controls the purity of DEXTRAN POWDER EP produced by a given manufacturer. Suppliers of raw materials can prove the suitability of DEXTRAN POWDER to their clients by showing that a DEXTRAN POWDER CEP has been issued for it. The manufacturer submits a DEXTRAN POWDER CEP (COS) as part of the market authorization procedure, and it takes on the role of a DEXTRAN POWDER CEP holder for the record. Additionally, the data presented in the DEXTRAN POWDER CEP (COS) is managed confidentially and offers a centralized system acknowledged by numerous nations, exactly like the DEXTRAN POWDER DMF.
A DEXTRAN POWDER CEP (COS) is recognised by all 36 nations that make up the European Pharmacopoeia Convention. DEXTRAN POWDER CEPs may be accepted in nations that are not members of the Ph. Eur. at the discretion of the authorities there.
click here to find a list of DEXTRAN POWDER suppliers with CEP (COS) on PharmaCompass.
National Drug Code is a comprehensive database maintained by the FDA that contains information on all drugs marketed in the US. This directory includes information about finished drug products, unfinished drug products, and compounded drug products, including those containing DEXTRAN POWDER as an active pharmaceutical ingredient (API).
The FDA updates the NDC directory daily. The NDC numbers for DEXTRAN POWDER API and other APIs are published in this directory by the FDA.
The NDC unfinished drugs database includes product listing information submitted for all unfinished drugs, such as active pharmaceutical ingredients (APIs), drugs intended for further processing and bulk drug substances for compounding.
Pharmaceutical companies that manufacture DEXTRAN POWDER as an active pharmaceutical ingredient (API) must furnish the FDA with an updated record of all drugs that they produce, prepare, propagate, compound, or process for commercial distribution in the US at their facilities.
The NDC directory also contains data on finished compounded human drug products that contain DEXTRAN POWDER and are produced by outsourcing facilities. While these outsourcing facilities are not mandated to assign a DEXTRAN POWDER NDC to their finished compounded human drug products, they may choose to do so.
click here to find a list of DEXTRAN POWDER suppliers with NDC on PharmaCompass.
DEXTRAN POWDER Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of DEXTRAN POWDER GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, API price, and more, enabling you to easily find the right DEXTRAN POWDER GMP manufacturer or DEXTRAN POWDER GMP API supplier for your needs.
A DEXTRAN POWDER CoA (Certificate of Analysis) is a formal document that attests to DEXTRAN POWDER's compliance with DEXTRAN POWDER specifications and serves as a tool for batch-level quality control.
DEXTRAN POWDER CoA mostly includes findings from lab analyses of a specific batch. For each DEXTRAN POWDER CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
DEXTRAN POWDER may be tested according to a variety of international standards, such as European Pharmacopoeia (DEXTRAN POWDER EP), DEXTRAN POWDER JP (Japanese Pharmacopeia) and the US Pharmacopoeia (DEXTRAN POWDER USP).

