
Synopsis
Synopsis
0
API Suppliers
0
USDMF
0
CEP/COS
0
JDMF
0
EU WC
0
KDMF
0
NDC API
0
VMF
0
Listed Suppliers
0
FDF Dossiers
0
FDA Orange Book
0
Europe
0
Canada
0
Australia
0
South Africa
0
Listed Dossiers
DRUG PRODUCT COMPOSITIONS
0
US Patents
0
US Exclusivities
0
Health Canada Patents
US Medicaid
NA
Annual Reports
NA
Regulatory FDF Prices
NA
0
FDF
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
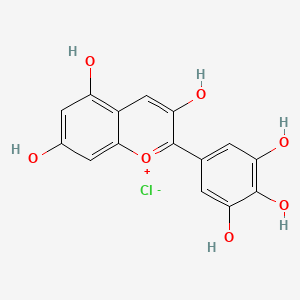

1. Delphinidin Chloride
1. Delphinidin Chloride
2. 528-53-0
3. Delphinidine
4. Delphinidol
5. Ephdine
6. Delfinidol Chloride
7. 8012-95-1
8. 3,3',4',5,5',7-hexahydroxyflavylium Chloride
9. Delphinidin (chloride)
10. 1-benzopyrylium, 3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-, Chloride
11. Em6md4aehe
12. 2-(3,4,5-trihydroxyphenyl)chromenylium-3,5,7-triol Chloride
13. Idb 1056
14. 2-(3,4,5-trihydroxyphenyl)chromenylium-3,5,7-triol;chloride
15. Chebi:38701
16. 3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)benzopyrylium Chloride
17. 3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-1-benzopyrylium Chloride
18. 3,3',4',5,5',7-hexahydroxy-2-phenylbenzopyrylium Chloride
19. 3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)chromenylium Chloride
20. Delphinidine; Delphinidol; Ephdine; Idb 1056;delfinidol Chloride; Delphinidin
21. Chembl590878
22. Delphinidin Chloride, Analytical Standard
23. Ccris 2518
24. Unii-em6md4aehe
25. Einecs 208-437-0
26. Paraffin Oils
27. Delphinidinchloride
28. 2-(3,4,5-trihydroxyphenyl)-1-benzopyrylium-3,5,7-triol
29. Mfcd00016663
30. Delphinidin [mi]
31. Schembl22369
32. Flavylium 3,3',4',5,5',7-hexahydroxy-, Chloride
33. Idb-1056
34. Dtxsid701019982
35. 3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)chromenium Chloride
36. Bcp15819
37. Hy-n2409
38. 3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)-1-benzopyrylium Chloride (1:1)
39. Akos027326494
40. Ac-35150
41. As-78369
42. Cs-0022610
43. Ft-0645153
44. Ft-0665679
45. E88811
46. Q367258
47. 3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)chromenyliumchloride
| Molecular Weight | 338.69 g/mol |
|---|---|
| Molecular Formula | C15H11ClO7 |
| Hydrogen Bond Donor Count | 6 |
| Hydrogen Bond Acceptor Count | 7 |
| Rotatable Bond Count | 1 |
| Exact Mass | 338.0193304 g/mol |
| Monoisotopic Mass | 338.0193304 g/mol |
| Topological Polar Surface Area | 122 Ų |
| Heavy Atom Count | 23 |
| Formal Charge | 0 |
| Complexity | 380 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 2 |
In severe cases of constipation, such as with fecal impaction, mineral oil and stool softener laxatives administered orally or rectally are indicated to soften the impacted feces. To help complete the evacuation of the impacted colon, a rectal stimulation or saline laxative may follow.
USP Convention. USPDI-Drug Information for the Health Care Professional. 14th ed. Volume I. Rockville, MD: United States Pharmacopeial Convention, Inc., 1994. (Plus Updates)., p. 1704
Medication (vet): orally, as a laxative with light grades (low viscosity) even having some advantage in animals over heavy grades (high viscosity).
Rossoff, I.S. Handbook of Veterinary Drugs. New York: Springer Publishing Company, 1974., p. 366
MEDICATION (VET): A preparation that contains 0.5% neomycin, 1% carbaryl, 9% sulfacetamide, 0.5% tetracaine, and 88.1% mineral oil is used in treatment of ear infections and ear mite infestations of small animals, including rabbits...
Booth, N.H., L.E. McDonald (eds.). Veterinary Pharmacology and Therapeutics. 5th ed. Ames, Iowa: Iowa State University Press, 1982., p. 668
Increase water retention in the stool by coating surfaces of stool and intestines with a water-immiscible film. Lubricant effect eases passage of contents through intestines. Emulsification of lubricant tends to enhance its ability to soften stool mass.
USP Convention. USPDI-Drug Information for the Health Care Professional. 14th ed. Volume I. Rockville, MD: United States Pharmacopeial Convention, Inc., 1994. (Plus Updates)., p. 1705
For more Therapeutic Uses (Complete) data for Mineral oil (7 total), please visit the HSDB record page.
In the 1940s, oleothorax (paraffin oil instillation) was widely used to treat patients with apical tuberculosis. The oil plombage should have been removed after a few years; however, since oleothoraces were usually asymptomatic, removal was uncommon. These in the meantime elderly patients are at risk of late complications, such as rupture of the oleothorax and aspiration of oil. We report the case of a 69-year-old man with a spontaneous rupture of an oleothorax leading to oil aspiration, lipid pneumonia and culture-proven disseminated tuberculosis with fatal outcome. Unexpected positive PCR for M. tuberculosis-DNA in tracheal secretions was one of the leading signs in this case. Thus oil plombage in patients with oleothorax may be "time bombs". Primary physicians should be aware of this life-threatening complication.
PMID:9857431 Kniehl E et al; Wien Klin Wochenschr 110 (20): 725-8 (1998)
In recent years, the oral use of mineral oil has not been advocated because of the possibility of interference with the absorption of fat-soluble vitamins and the danger of pulmonary aspiration. The dose required for the former effect exceeds that normally used in clinical practice. ... Oral mineral oil should not be given to patients with swallowing abnormalities.
American Medical Association, Council on Drugs. AMA Drug Evaluations Annual 1994. Chicago, IL: American Medical Association, 1994., p. 951
Oral mineral oil is not recommended for bedridden elderly patients since they are more prone to aspiration of oil droplets, which amy produce lipid pneumonia.
USP Convention. USPDI-Drug Information for the Health Care Professional. 14th ed. Volume I. Rockville, MD: United States Pharmacopeial Convention, Inc., 1994. (Plus Updates)., p. 1705
Oral mineral oil is not recommended for children up to 6 years of age since patients in this age group are more prone to aspiration of oil droplets, which may produce lipid pneumonia.
USP Convention. USPDI-Drug Information for the Health Care Professional. 14th ed. Volume I. Rockville, MD: United States Pharmacopeial Convention, Inc., 1994. (Plus Updates)., p. 1705
For more Drug Warnings (Complete) data for Mineral oil (6 total), please visit the HSDB record page.
1= Practically non-toxic: probable oral lethal dose (human) above 15 g/kg, more than 1 quart (2.2 lb) for 70 kg person (150 lb).
Gosselin, R.E., R.P. Smith, H.C. Hodge. Clinical Toxicology of Commercial Products. 5th ed. Baltimore: Williams and Wilkins, 1984., p. II-156
Vet: ... parenterally, in ... vaccines to prolong antigen effect. Histological studies indicate that great deal of ... oil remains at injection site for long time, with macrophages picking up small amounts and transporting it through lymphatic system. Its ultimate fate is still unknown.
Rossoff, I.S. Handbook of Veterinary Drugs. New York: Springer Publishing Company, 1974., p. 367
When large amounts of liquid petrolatum were fed to rabbits, rats and guinea pigs, small quantities were deposited in the mesenteric lymph nodes and, in several cases, in the intestinal mucosa, liver and spleen.
IARC. Monographs on the Evaluation of the Carcinogenic Risk of Chemicals to Humans. Geneva: World Health Organization, International Agency for Research on Cancer, 1972-PRESENT. (Multivolume work). Available at: https://monographs.iarc.fr/ENG/Classification/index.php, p. V33 132 (1984)
Tritiated mineral oil was administered orally or by ip injection to Sprague-Dawley and Holtzman rats. Five hours after oral administration of 0.66 mL/kg body weight, 1.5% of the dose had been absorbed unchanged, and an additional 1.5% was found in the carcasses as non-mineral oil substances. Liver, fat, kidney, brain and spleen contained mineral oil. Within two days, only 0.3% remained in the animals. After ip administration, the mineral oil was retained to a greater extent, and only 11% had been excreted in the feces eight days after treatment.
IARC. Monographs on the Evaluation of the Carcinogenic Risk of Chemicals to Humans. Geneva: World Health Organization, International Agency for Research on Cancer, 1972-PRESENT. (Multivolume work). Available at: https://monographs.iarc.fr/ENG/Classification/index.php, p. V33 133 (1984)
In general, mineral oil is absorbed to only a limited extent from the gastrointestinal tract. However, it as found in the liver, spleen, mesenteric and portal-hepatic lymph nodes and lungs of a man known to have ingested large amounts of liquid paraffin over many years. Mineral oil has been observed by differential staining and gas chromatography (GC) procedures in the lung tissue of many individuals who routinely use mineral oil by oral or nasal administration
IARC. Monographs on the Evaluation of the Carcinogenic Risk of Chemicals to Humans. Geneva: World Health Organization, International Agency for Research on Cancer, 1972-PRESENT. (Multivolume work). Available at: https://monographs.iarc.fr/ENG/Classification/index.php, p. V33 136 (1984)
For more Absorption, Distribution and Excretion (Complete) data for Mineral oil (10 total), please visit the HSDB record page.
Market Place

REF. STANDARDS & IMPURITIES

ABOUT THIS PAGE
76
PharmaCompass offers a list of Delphinidin Chloride API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Delphinidin Chloride manufacturer or Delphinidin Chloride supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Delphinidin Chloride manufacturer or Delphinidin Chloride supplier.
A Delphinidin Chloride manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Delphinidin Chloride, including repackagers and relabelers. The FDA regulates Delphinidin Chloride manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Delphinidin Chloride API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
A Delphinidin Chloride supplier is an individual or a company that provides Delphinidin Chloride active pharmaceutical ingredient (API) or Delphinidin Chloride finished formulations upon request. The Delphinidin Chloride suppliers may include Delphinidin Chloride API manufacturers, exporters, distributors and traders.
Delphinidin Chloride Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Delphinidin Chloride GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Delphinidin Chloride GMP manufacturer or Delphinidin Chloride GMP API supplier for your needs.
A Delphinidin Chloride CoA (Certificate of Analysis) is a formal document that attests to Delphinidin Chloride's compliance with Delphinidin Chloride specifications and serves as a tool for batch-level quality control.
Delphinidin Chloride CoA mostly includes findings from lab analyses of a specific batch. For each Delphinidin Chloride CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Delphinidin Chloride may be tested according to a variety of international standards, such as European Pharmacopoeia (Delphinidin Chloride EP), Delphinidin Chloride JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Delphinidin Chloride USP).

