



API Suppliers

US DMFs Filed

CEP/COS Certifications

JDMFs Filed
0
Other Certificates
Other Suppliers
0
0

USA (Orange Book)

Europe

Canada
0

Australia
0

South Africa
Uploaded Dossiers
U.S. Medicaid
0
Annual Reports
0
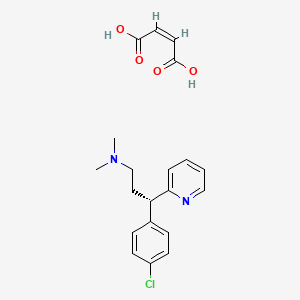

1. Dexchlorpheniramine
2. Dexchlorpheniramine Maleate (1:1), (+-)-isomer
3. Dexchlorpheniramine Maleate (1:1), (r)-isomer
4. Dexchlorpheniramine Maleate (1:1), (s)-isomer
5. Dexchlorpheniramine Monohydrochloride
6. Dexchlorpheniramine Sodium Maleate (1:1)
7. Dexchlorpheniramine, (+-)-isomer
8. Dexchlorpheniramine, (r)-isomer
9. Dexchlorpheniramine, Monohydrochloride, (s)-isomer
10. Dexclor
11. Dexclorfeniramine
12. Dextrochlorpheniramine Maleate
13. Polaramin
14. Polaramine
15. Polargen Td
16. Polaronil
1. 2438-32-6
2. Polaramine
3. (+)-chlorpheniramine Maleate
4. D-chlorpheniramine Maleate
5. Dexchloropheniramine Maleate
6. Mylaramine
7. Dexchlorpheniramine (maleate)
8. Chlorpheniramine D-form Maleate
9. S-(+)-chlorpheniramine Maleate Salt
10. Dexchlorpheniramine Maleate, Usp
11. Chebi:4465
12. B10yd955qw
13. Chlorpheniramine Maleate, (s)-
14. Dexchlorpheniramine Maleate [usp]
15. (z)-but-2-enedioic Acid;(3s)-3-(4-chlorophenyl)-n,n-dimethyl-3-pyridin-2-ylpropan-1-amine
16. Nsc-759156
17. Dextrochlorpheniramine Maleate
18. Dexchlorpheniramine Maleate (usp)
19. (s)-3-(4-chlorophenyl)-n,n-dimethyl-3-(pyridin-2-yl)propan-1-amine Maleate
20. (gamma-(4-chlorophenyl)-pyridine-2-propyl)(dimethyl)ammonium Hydrogen (z)-maleate
21. Sr-01000075763
22. Unii-b10yd955qw
23. Polar Amine
24. S-(+)-chlorpheniramine Maleate
25. Rescon Jr
26. Rescon Mx
27. Mylaramine (tn)
28. Polaramine (tn)
29. (s)-chlorpheniramine Maleate Salt
30. Einecs 219-450-6
31. Mfcd00079046
32. Dexchlorpheniraminmaleat
33. Mls001401384
34. Schembl119500
35. Chembl1200927
36. Dtxsid001017216
37. Hms2052e15
38. Hms2233i15
39. Hms3260f10
40. D-chlorpheniramine Maleate (jp17)
41. Hy-b1062
42. Tox21_500264
43. Bdbm50247890
44. (+)-2-(p-chloro-alpha-(2-(dimethylamino)ethyl)benzyl)pyridine Maleate (1:1)
45. Akos015962825
46. Ccg-101121
47. Cs-4618
48. Db09555
49. Lp00264
50. Nc00371
51. Nsc 759156
52. (2z)-but-2-enedioic Acid; [(3s)-3-(4-chlorophenyl)-3-(pyridin-2-yl)propyl]dimethylamine
53. D-chlorpheniramine Maleate [jan]
54. Ncgc00093722-01
55. Ncgc00093722-02
56. Ncgc00093722-03
57. Ncgc00260949-01
58. 2-pyridinepropanamine, .gamma.-(4-chlorophenyl)-n,n-dimethyl-, (s)-, (z)-2-butenedioate (1:1)
59. Ac-19009
60. As-17464
61. Smr000471617
62. Dexchlorpheniramine Maleate [vandf]
63. Dexchlorpheniramine Maleate [usp-rs]
64. Dexchlorpheniramine Maleate [who-dd]
65. Chlorpheniramine D-form Maleate [mi]
66. Eu-0100264
67. C 4915
68. C07783
69. D00668
70. Dexchlorpheniramine Maleate [ep Impurity]
71. Dexchlorpheniramine Maleate [orange Book]
72. 438d326
73. A924268
74. Dexchlorpheniramine Maleate [ep Monograph]
75. Dexchlorpheniramine Maleate [usp Monograph]
76. Sr-01000075763-1
77. Sr-01000075763-4
78. Q27106393
79. (gammas)-gamma-(4-chlorophenyl)-n,n-dimethyl-2-pyridinepropanamine Maleate
80. Dexchlorpheniramine Maleate, European Pharmacopoeia (ep) Reference Standard
81. Gammas-(4-chlorophenyl)-n,n-dimethyl-2-pyridinepropanamine, 2z-butenedioate
82. (+)-2-(p-chloro-.alpha.-(2-(dimethylamino)ethyl)benzyl)pyridine Maleate (1:1)
83. Dexchlorpheniramine Maleate, United States Pharmacopeia (usp) Reference Standard
84. Pyridine, 2-(p-chloro-alpha-(2-(dimethylamino)ethyl)benzyl)-, Maleate (1:1), (+)-
85. 2-pyridinepropanamine, Gamma-(4-chlorophenyl)-n,n-dimethyl-, (s)-, (z)-2-butenedioate (1:1)
| Molecular Weight | 390.9 g/mol |
|---|---|
| Molecular Formula | C20H23ClN2O4 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 6 |
| Rotatable Bond Count | 7 |
| Exact Mass | 390.1346349 g/mol |
| Monoisotopic Mass | 390.1346349 g/mol |
| Topological Polar Surface Area | 90.7 Ų |
| Heavy Atom Count | 27 |
| Formal Charge | 0 |
| Complexity | 368 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 1 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 1 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 2 |
| 1 of 2 | |
|---|---|
| Drug Name | Dexchlorpheniramine maleate |
| PubMed Health | Dexchlorpheniramine Maleate (By mouth) |
| Drug Classes | Respiratory Agent |
| Drug Label | Each 5 mL (teaspoonful) contains:Dexchlorpheniramine Maleate, USP2 mgAlcohol not more than7.0%Dexchlorpheniramine Maleate, USP, an antihistamine agent, is a white, odorless crystalline powder that is freely soluble in water. The molecular formula is... |
| Active Ingredient | Dexchlorpheniramine maleate |
| Dosage Form | Syrup |
| Route | Oral |
| Strength | 2mg/5ml |
| Market Status | Prescription |
| Company | Wockhardt |
| 2 of 2 | |
|---|---|
| Drug Name | Dexchlorpheniramine maleate |
| PubMed Health | Dexchlorpheniramine Maleate (By mouth) |
| Drug Classes | Respiratory Agent |
| Drug Label | Each 5 mL (teaspoonful) contains:Dexchlorpheniramine Maleate, USP2 mgAlcohol not more than7.0%Dexchlorpheniramine Maleate, USP, an antihistamine agent, is a white, odorless crystalline powder that is freely soluble in water. The molecular formula is... |
| Active Ingredient | Dexchlorpheniramine maleate |
| Dosage Form | Syrup |
| Route | Oral |
| Strength | 2mg/5ml |
| Market Status | Prescription |
| Company | Wockhardt |
Dexchlorpheniramine can be used in the treatment of perennial and seasonal allergic rhinitis, vasomotor rhiniti, allergic conjunctivitis due to inhalant allergens and foods, mild uncomplicated allergic skin manifestations of urticaria and angioedema, amelioration of allergic reactions to blood or plasma, and dermographism.
In allergic reactions, an allergen binds to IgE antibodies on mast cells and basophils. Once this occurs IgE receptors crosslink with each other triggering a series of events that eventually leads to cell-degranulation and the release of histamine (and other chemical mediators) from the mast cell or basophil. Histamine can react with local or widespread tissues through histamine receptors. Histamine, acting on H1-receptors, produces pruritis, vasodilatation, hypotension, flushing, headache, tachycardia, and bronchoconstriction. Histamine also increases vascular permeability and potentiates pain. Dexchlorpheniramine, is a histamine H1 antagonist of the alkylamine class. It competes with histamine for the normal H1-receptor sites on effector cells of the gastrointestinal tract, blood vessels and respiratory tract. It provides effective, temporary relief of sneezing, watery and itchy eyes, and runny nose due to hay fever and other upper respiratory allergies.
Histamine H1 Antagonists
Drugs that selectively bind to but do not activate histamine H1 receptors, thereby blocking the actions of endogenous histamine. Included here are the classical antihistaminics that antagonize or prevent the action of histamine mainly in immediate hypersensitivity. They act in the bronchi, capillaries, and some other smooth muscles, and are used to prevent or allay motion sickness, seasonal rhinitis, and allergic dermatitis and to induce somnolence. The effects of blocking central nervous system H1 receptors are not as well understood. (See all compounds classified as Histamine H1 Antagonists.)
Absorption
Oral bioavailability in rats 40.5%
Route of Elimination
Renal excretion
Volume of Distribution
321L
Clearance
9.8L/h
Hepatic metabolism. Major metabolism by CYP 2D6 and minor metabolism by 3A4, 2C11 and 2B1.
20-30 h
Competes with histamine for H1-receptor sites on effector cells in the gastrointestinal tract, blood vessels, and respiratory tract. Dexchlorpheniramine is the predominant active isomer of chlorpheniramine and is approximately twice as active as the racemic compound.








