
Synopsis
Synopsis
0
CEP/COS
0
JDMF
0
KDMF
0
NDC API
0
VMF
0
Canada
DRUG PRODUCT COMPOSITIONS
0
US Patents
0
US Exclusivities
0
Health Canada Patents
0
FDF
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
0
News #PharmaBuzz

1. Acaril
2. Acarosan
3. Ansar
4. Antiscabiosum
5. Ascabiol
6. Benzanil
7. Benzemul
8. Benzylbenzoaat Smeersel Fna
9. Novoscabin
1. 120-51-4
2. Ascabiol
3. Benzoic Acid Benzyl Ester
4. Benzoic Acid, Phenylmethyl Ester
5. Benylate
6. Novoscabin
7. Benzoic Acid, Benzyl Ester
8. Scabiozon
9. Scabitox
10. Scobenol
11. Ascabin
12. Phenylmethyl Benzoate
13. Benzyl Phenylformate
14. Benzylets
15. Colebenz
16. Peruscabin
17. Scabagen
18. Scabanca
19. Benzylbenzoate
20. Vanzoate
21. Scabide
22. Venzonate
23. Benzyl Benzenecarboxylate
24. Benzylis Benzoas
25. Benzyl Alcohol Benzoic Ester
26. Benzylbenzenecarboxylate
27. Benzylester Kyseliny Benzoove
28. Benzoic Acid Phenylmethylester
29. Fema No. 2138
30. Nsc 8081
31. Nsc-8081
32. Chembl1239
33. Antiscabiosum
34. Chebi:41237
35. N863nb338g
36. Ncgc00094981-03
37. Peruscabina
38. Dsstox_cid_9153
39. Benzylum Benzoicum
40. Dsstox_rid_78686
41. Dsstox_gsid_29153
42. Benzoesaeurebenzylester
43. Caswell No. 082
44. Venzoate
45. Fema Number 2138
46. Benzyl Benzoate, Analytical Standard
47. Benzyl Benzoate (natural)
48. Bzm
49. Cas-120-51-4
50. Smr000471875
51. Hsdb 208
52. Einecs 204-402-9
53. Benzylester Kyseliny Benzoove [czech]
54. Epa Pesticide Chemical Code 009501
55. Benzylbenzoat
56. Brn 2049280
57. Benzyl Benzoate [usp:jan]
58. Acarobenzyl
59. Benzevan
60. Bengal
61. Benzyl-benzoate
62. Unii-n863nb338g
63. Ai3-00523
64. 1dzm
65. Benylate (tn)
66. Benzoic Acid Benzyl
67. Mfcd00003075
68. Spectrum_001240
69. Benzoic Acid-benzyl Ester
70. Spectrum2_000532
71. Spectrum3_001757
72. Spectrum4_000773
73. Spectrum5_001128
74. Wln: Rvo1r
75. Benzyl Benzoate, >=99%
76. Ec 204-402-9
77. Schembl3038
78. Benzyl Benzoate [ii]
79. Benzyl Benzoate [mi]
80. Benzyl Benzoate Bp98
81. Bspbio_003494
82. Kbiogr_001186
83. Kbioss_001720
84. 4-09-00-00307 (beilstein Handbook Reference)
85. Mls001066412
86. Mls001336003
87. Mls001336004
88. Benzyl Benzoate [fcc]
89. Benzyl Benzoate [jan]
90. Benzyloxy Phenyl Ketone
91. Divk1c_000204
92. Spectrum1503002
93. Spbio_000543
94. Benzyl Benzoate (jp17/usp)
95. Benzyl Benzoate [fhfi]
96. Benzyl Benzoate [hsdb]
97. Benzyl Benzoate [inci]
98. Zinc1021
99. Benzyl Benzoate [mart.]
100. Dtxsid8029153
101. Benzoic Acid,benzyl Ester
102. Hms500k06
103. Kbio1_000204
104. Kbio2_001720
105. Kbio2_004288
106. Kbio2_006856
107. Kbio3_002714
108. Benzyl Benzoate [usp-rs]
109. Benzyl Benzoate [who-dd]
110. Benzyl Benzoate [who-ip]
111. Nsc8081
112. Ninds_000204
113. Hms1921p16
114. Hms2092f20
115. Hms2269d24
116. Pharmakon1600-01503002
117. Hy-b0935
118. Tox21_111372
119. Tox21_201337
120. Tox21_303418
121. Bdbm50134035
122. Ccg-39578
123. Nsc758204
124. S4599
125. Stl183088
126. Benzyl Benzoate [ep Impurity]
127. Benzyl Benzoate [orange Book]
128. Akos003495939
129. Benzyl Benzoate [ep Monograph]
130. Benzyl Benzoate [usp Impurity]
131. Benzyl Benzoate, >=99%, Fcc, Fg
132. Tox21_111372_1
133. Benzyl Benzoate [usp Monograph]
134. Db00676
135. Nsc-758204
136. Benzylis Benzoas [who-ip Latin]
137. Idi1_000204
138. Benzyl Benzoate, For Synthesis, 99.0%
139. Ncgc00094981-01
140. Ncgc00094981-02
141. Ncgc00094981-04
142. Ncgc00094981-05
143. Ncgc00094981-07
144. Ncgc00257502-01
145. Ncgc00258889-01
146. Ac-17033
147. Ls-14279
148. Sbi-0051748.p002
149. Db-041563
150. B0064
151. Ft-0622708
152. Benzyl Benzoate, Natural, >=99%, Fcc, Fg
153. Benzyl Benzoate, Reagentplus(r), >=99.0%
154. Benzyl Benzoate, Saj First Grade, >=98.0%
155. Benzyl Benzoate, Tested According To Ph.eur.
156. A14577
157. A19449
158. Benzyl Benzoate, Saj Special Grade, >=99.0%
159. D01138
160. Ab00052298_07
161. Benzyl Benzoate, Vetec(tm) Reagent Grade, 98%
162. Benzyl Benzoate, Meets Usp Testing Specifications
163. Q413755
164. Sr-01000763773
165. Benzoic Acid-benzyl Ester 5000 Microg/ml In Hexane
166. Q-200696
167. Sr-01000763773-2
168. Brd-k52072429-001-06-1
169. Benzoic Acid-benzyl Ester 100 Microg/ml In Acetonitrile
170. Benzoic Acid Benzyl Ester; Benzoic Acid Phenylmethyl Ester
171. Benzyl Benzoate, Certified Reference Material, Tracecert(r)
172. Benzyl Benzoate, United States Pharmacopeia (usp) Reference Standard
173. Benzyl Benzoate, Pharmaceutical Secondary Standard; Certified Reference Material
| Molecular Weight | 212.24 g/mol |
|---|---|
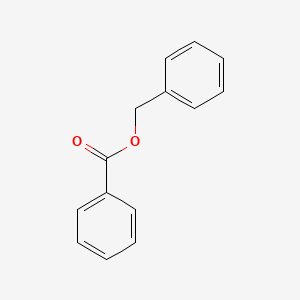
| Molecular Formula | C14H12O2 |
| XLogP3 | 4 |
| Hydrogen Bond Donor Count | 0 |
| Hydrogen Bond Acceptor Count | 2 |
| Rotatable Bond Count | 4 |
| Exact Mass | 212.083729621 g/mol |
| Monoisotopic Mass | 212.083729621 g/mol |
| Topological Polar Surface Area | 26.3 Ų |
| Heavy Atom Count | 16 |
| Formal Charge | 0 |
| Complexity | 213 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
Benzyl benzoate is used as an alternative agent for the topical treatment of scabies and also has been used for the topical treatment of pediculosis (lice infestation). ... Benzyl benzoate lotion, in an approximate concentration of 28% w/w, is applied topically. The drug should not be administered orally. Containers of the lotion should be shaken before using.
American Society of Health-System Pharmacists; Drug Information 2018. Bethesda, MD. 2018
Scabicide, pediculicide.
O'Neil, M.J. (ed.). The Merck Index - An Encyclopedia of Chemicals, Drugs, and Biologicals. Cambridge, UK: Royal Society of Chemistry, 2013., p. 199
VET: Acaricide, pediculicide. Contraindicated in cats.
O'Neil, M.J. (ed.). The Merck Index - An Encyclopedia of Chemicals, Drugs, and Biologicals. Cambridge, UK: Royal Society of Chemistry, 2013., p. 199
Studies in animals showed that oral ingestion of large doses of benzyl benzoate resulted in progressive incoordination, CNS excitation, seizures, and death.
American Society of Health-System Pharmacists; Drug Information 2018. Bethesda, MD. 2018
When used in appropriate dosage, topically applied benzyl benzoate appears to have a low order of toxicity. Slight local irritation (especially of the male genitalia), itching, and allergic skin sensitivity may occur, and contact with the face, eyes, mucous membranes, and urethral meatus should be avoided. Repeated application of benzyl benzoate frequently causes contact dermatitis.
American Society of Health-System Pharmacists; Drug Information 2018. Bethesda, MD. 2018
Benzyl benzoate is contraindicated in patients with a history of hypersensitivity to the drug.
American Society of Health-System Pharmacists; Drug Information 2018. Bethesda, MD. 2018
Benzyl benzoate should not be applied to acutely inflamed skin or raw, weeping surfaces. If primary irritation or hypersensitivity occurs, treatment should be discontinued and the drug removed with soap and water.
American Society of Health-System Pharmacists; Drug Information 2018. Bethesda, MD. 2018
VET: Cats are particularly susceptible to this action, and the use of benzyl benzoate in this species is therefore contraindicated. The most prominent feature is nervous excitability. The clinical signs resemble those of poisoning by benzoic acid.
Humphreys, D.J. Veterinary Toxicology. 3rd ed. London, England: Bailliere Tindell, 1988., p. 131
Used to kill lice and the mites responsible for the skin condition scabies.
Benzyl benzoate is one of the older preparations used to treat scabies. Scabies is a skin infection caused by the mite sarcoptes scabiei. It is characterised by severe itching (particularly at night), red spots, and may lead to a secondary infection. Benzyl benzoate is lethal to this mite and so is useful in the treatment of scabies. It is also used to treat lice infestation of the head and body. Benzyl benzoate is not the treatment of choice for scabies due to its irritant properties.
Insecticides
Pesticides designed to control insects that are harmful to man. The insects may be directly harmful, as those acting as disease vectors, or indirectly harmful, as destroyers of crops, food products, or textile fabrics. (See all compounds classified as Insecticides.)
P - Antiparasitic products, insecticides and repellents
P03 - Ectoparasiticides, incl. scabicides, insecticides and repellents
P03A - Ectoparasiticides, incl. scabicides
P03AX - Other ectoparasiticides, incl. scabicides
P03AX01 - Benzyl benzoate
Absorption
No data are available on percutaneous absorption of benzyl benzoate. Some older studies have suggested some percutaneous absorption, however the amount was not quantified.
The maternal and fetal toxicity of benzyl benzoate, commonly used as antiparasitic insecticide, was evaluated in pregnant rats after a daily oral dose of 25 and 100 mg/kg. Biochemical, histopathological, and morphological examinations were performed. Dams were observed for maternal body weights and food and water consumption and subjected to caesarean section on (GD) 20. Maternal and fetal liver, kidney, heart, brain, and placenta were examined histopathologically under light microscope. Maternal and fetal liver and placenta were stained immunohistochemically for vascular endothelial growth factor (VEGF). Morphometric analysis of fetal body lengths, placental measurements, and fetal skeletal stainings was performed. Statistically significant alterations in biochemical parameters and placental and skeletal measurements were determined in treatment groups. In addition to histopathological changes, considerable differences were observed in the immunolocalization of VEGF in treatment groups. These results demonstrated that benzyl benzoate and its metabolites can transport to the placenta and eventually enter the fetuses.
PMID:21922633 Kockaya EA, Kilic A et al; Environ Toxicol 29 (1): 40-53 (2014)
The percutaneous absorption of benzyl benzoate measured in vivo in human and monkey studies. With the application sites occluded, 54% of the applied dose penetrated human skin in 24 hr compared with 69% absorption in the monkey skin.
PMID:2379896 Bronaugh RL et al; Food Chem Toxicol 28 (5): 369-74 (1990)
Rapidly hydrolyzed to benzoic acid and benzyl alcohol, which is further oxidized to benzoic acid. The benzoic acid is conjugated with glycine to form hippuric acid.
Converted to hippuric acid in vivo.
Gosselin, R.E., R.P. Smith, H.C. Hodge. Clinical Toxicology of Commercial Products. 5th ed. Baltimore: Williams and Wilkins, 1984., p. II-203
Benzyl benzoate exerts toxic effects on the nervous system of the parasite, resulting in its death. It is also toxic to mite ova, though its exact mechanism of action is unknown. In vitro, benzyl benzoate has been found to kill the Sarcoptes mite within 5 minutes.
Global Sales Information

Market Place

REF. STANDARDS & IMPURITIES

ABOUT THIS PAGE
45
PharmaCompass offers a list of Benzyl Benzoate API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Benzyl Benzoate manufacturer or Benzyl Benzoate supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Benzyl Benzoate manufacturer or Benzyl Benzoate supplier.
A Benzyl Benzoate manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Benzyl Benzoate, including repackagers and relabelers. The FDA regulates Benzyl Benzoate manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Benzyl Benzoate API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
click here to find a list of Benzyl Benzoate manufacturers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PhamaCompass.
A Benzyl Benzoate supplier is an individual or a company that provides Benzyl Benzoate active pharmaceutical ingredient (API) or Benzyl Benzoate finished formulations upon request. The Benzyl Benzoate suppliers may include Benzyl Benzoate API manufacturers, exporters, distributors and traders.
click here to find a list of Benzyl Benzoate suppliers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PharmaCompass.
A Benzyl Benzoate DMF (Drug Master File) is a document detailing the whole manufacturing process of Benzyl Benzoate active pharmaceutical ingredient (API) in detail. Different forms of Benzyl Benzoate DMFs exist exist since differing nations have different regulations, such as Benzyl Benzoate USDMF, ASMF (EDMF), JDMF, CDMF, etc.
A Benzyl Benzoate DMF submitted to regulatory agencies in the US is known as a USDMF. Benzyl Benzoate USDMF includes data on Benzyl Benzoate's chemical properties, information on the facilities and procedures used, and details about packaging and storage. The Benzyl Benzoate USDMF is kept confidential to protect the manufacturer’s intellectual property.
click here to find a list of Benzyl Benzoate suppliers with USDMF on PharmaCompass.
A Benzyl Benzoate written confirmation (Benzyl Benzoate WC) is an official document issued by a regulatory agency to a Benzyl Benzoate manufacturer, verifying that the manufacturing facility of a Benzyl Benzoate active pharmaceutical ingredient (API) adheres to the Good Manufacturing Practices (GMP) regulations of the importing country. When exporting Benzyl Benzoate APIs or Benzyl Benzoate finished pharmaceutical products to another nation, regulatory agencies frequently require a Benzyl Benzoate WC (written confirmation) as part of the regulatory process.
click here to find a list of Benzyl Benzoate suppliers with Written Confirmation (WC) on PharmaCompass.
Benzyl Benzoate Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Benzyl Benzoate GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Benzyl Benzoate GMP manufacturer or Benzyl Benzoate GMP API supplier for your needs.
A Benzyl Benzoate CoA (Certificate of Analysis) is a formal document that attests to Benzyl Benzoate's compliance with Benzyl Benzoate specifications and serves as a tool for batch-level quality control.
Benzyl Benzoate CoA mostly includes findings from lab analyses of a specific batch. For each Benzyl Benzoate CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Benzyl Benzoate may be tested according to a variety of international standards, such as European Pharmacopoeia (Benzyl Benzoate EP), Benzyl Benzoate JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Benzyl Benzoate USP).

