
Synopsis
Synopsis
0
USDMF
0
JDMF
0
NDC API
0
VMF
0
EDQM
0
USP
0
JP
0
Others
0
FDA Orange Book
DRUG PRODUCT COMPOSITIONS
0
US Patents
0
US Exclusivities
0
Health Canada Patents
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
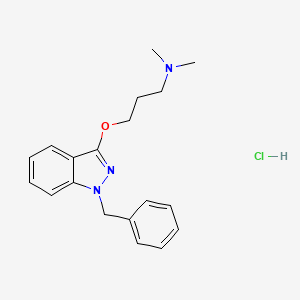

1. Benzidamine
2. Benzindamine
3. Benzydamine
4. Benzydamine Monohydrochloride
5. Difflam
6. Hydrochloride, Benzydamine
7. Lonol
8. Monohydrochloride, Benzydamine
9. Novo Benzydamine
10. Novo-benzydamine
11. Opalgyne
12. Pms Benzydamine
13. Pms-benzydamine
14. Ratio Benzydamine
15. Ratio-benzydamine
16. Rosalgin
17. Sun Benz
18. Sun-benz
19. Tantum
20. Tantum Topico
1. 132-69-4
2. Benzydamine Hcl
3. Benzidamine Hydrochloride
4. Benzindamine Hydrochloride
5. Tantum
6. Benzydamine (hydrochloride)
7. Af-864
8. Nsc-759276
9. 132-69-4 (hcl)
10. K2gi407r4q
11. Benalgin
12. {3-[(1-benzyl-1h-indazol-3-yl)oxy]propyl}dimethylamine Hydrochloride
13. 1-benzyl-3-(3-(dimethylamino)propoxy)-1h-indazole Monohydrochloride
14. 3-((1-benzyl-1h-indazol-3-yl)oxy)-n,n-dimethylpropan-1-amine Hydrochloride
15. 1-propanamine, N,n-dimethyl-3-((1-(phenylmethyl)-1h-indazol-3-yl)oxy)-, Monohydrochloride
16. Benzidan
17. Benzyrin
18. Difflam
19. Dorinamin
20. Enzamin
21. Epirotin
22. Flogaton
23. Imotryl
24. Oxinazin
25. Ririlim
26. Riripen
27. Salyzoron
28. Tamas
29. Verax
30. 1-benzyl-3-(3-(dimethylamino)propoxy)-1h-indazole Hydrochloride
31. 1-benzyl-3-(3-[dimethylamino]propoxy)-1h-indazole Hydrochloride
32. Dsstox_cid_25293
33. Dsstox_rid_80786
34. Dsstox_gsid_45293
35. 1-benzyl-3-[3-(dimethylamino)propoxy]-1h-indazole Hydrochloride
36. 3-(1-benzylindazol-3-yl)oxy-n,n-dimethylpropan-1-amine;hydrochloride
37. Smr000544402
38. Ncgc00016397-01
39. Cas-132-69-4
40. Einecs 205-076-0
41. Af 864
42. Af864
43. Benzydamine Hydrochloride [usan:jan]
44. Unii-k2gi407r4q
45. Sr-01000799149
46. Benzydamine, Hcl
47. Prestwick_403
48. Tantum (tn)
49. 1-benzyl-3-gamma-dimethylaminopropoxy-1h-indazole Hydrochloride
50. 1-propanamine, N,n-dimethyl-3-((1-(phenylmethyl)-1h-indazol-3-yl)oxy)-, Hydrochloride
51. Schembl42644
52. Benzydaminehydrochloride
53. Mls001216204
54. Mls001306431
55. Mls001333107
56. Mls001333108
57. Mls002222210
58. Not Available;benzydamine Hcl
59. Benzydamine-[d6] Hydrochloride
60. Chembl1528134
61. Dtxsid1045293
62. Chebi:31266
63. Hms1568l16
64. Pharmakon1600-01505975
65. Bcp12133
66. Tox21_110420
67. Hy-30235a
68. Mfcd00078957
69. Nsc759276
70. S4165
71. Benzydamine Hydrochloride (jan/usan)
72. Benzydamine Hydrochloride [mi]
73. 1h-indazole, 1-benzyl-3-(3-(dimethylamino)propoxy)-, Monohydrochloride
74. 3-((1-benzyl-1h-indazol-3-yl)oxy)-n,n-dimethylpropylamine Monohydrochloride
75. Akos015994635
76. Tox21_110420_1
77. Ac-6791
78. Benzydamine Hydrochloride [jan]
79. Ccg-213582
80. Cs-4463
81. Hs-1002
82. Nsc 759276
83. Benzydamine Hydrochloride [usan]
84. Benzydamine Hydrochloride [mart.]
85. Ncgc00016397-04
86. Benzydamine Hydrochloride [who-dd]
87. 1-benzyl-3-hydroxy-1h-indazolesodiumsalt
88. B3751
89. Ft-0662621
90. Sw196646-3
91. Benzydamine Hydrochloride, Analytical Standard
92. A14562
93. Benzydamine Hydrochloride [ep Monograph]
94. D01410
95. H10439
96. A854848
97. J-006209
98. Sr-01000799149-4
99. Q27281858
100. 1-benzyl-3-(3-(dimethylamino) Propoxy)-1h Indozale Hydrochloride
101. 3-(1-benzyl-1h-indazol-3-yloxy)-n,n-dimethylpropan-1-amine Hydrochloride
| Molecular Weight | 345.9 g/mol |
|---|---|
| Molecular Formula | C19H24ClN3O |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 3 |
| Rotatable Bond Count | 7 |
| Exact Mass | 345.1607901 g/mol |
| Monoisotopic Mass | 345.1607901 g/mol |
| Topological Polar Surface Area | 30.3 Ų |
| Heavy Atom Count | 24 |
| Formal Charge | 0 |
| Complexity | 344 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 2 |
Anti-Inflammatory Agents
Substances that reduce or suppress INFLAMMATION. (See all compounds classified as Anti-Inflammatory Agents.)
 BioXera Pharma: A trusted partner for high-quality human and veterinary APIs and intermediates across global regulated markets.
BioXera Pharma: A trusted partner for high-quality human and veterinary APIs and intermediates across global regulated markets.
 Tenatra connects Indian manufacturers with global buyers through active partners in Germany, Switzerland, Belgium, Spain & Turkey.
Tenatra connects Indian manufacturers with global buyers through active partners in Germany, Switzerland, Belgium, Spain & Turkey.

Certificate Number : CEP 2021-325 - Rev 02
Issue Date : 2025-07-11
Type : Chemical
Substance Number : 2759
Status : Valid

Date of Issue : 2025-10-17
Valid Till : 2027-07-07
Written Confirmation Number : WC-0156
Address of the Firm :

Registrant Name : Daeshin Muyak Co., Ltd.
Registration Date : 2025-06-23
Registration Number : 20250623-211-J-1932
Manufacturer Name : Bal Pharma Limited.
Manufacturer Address : 61-B, Bommasandra Industrial Area Bangalore - 560 099, India





Certificate Number : CEP 2021-203 - Rev 01
Issue Date : 2024-01-17
Type : Chemical
Substance Number : 2759
Status : Valid



 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info : Generic
Registration Country : South Africa
Brand Name : THROFLAM-CO
Dosage Form : MOW
Dosage Strength : 22.5mg/15 ml
Packaging : 200X1mg/15 ml
Approval Date :
Application Number :
Regulatory Info : Generic
Registration Country : South Africa
 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info : Generic
Registration Country : South Africa
Brand Name : Throflam-Co
Dosage Form : SPO
Dosage Strength : 22.5mg/15ml
Packaging : 30X1mg/15ml
Approval Date :
Application Number :
Regulatory Info : Generic
Registration Country : South Africa
 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info : Generic
Registration Country : South Africa
Brand Name : Throflam
Dosage Form : MOW
Dosage Strength : 22.5mg/15ml
Packaging : 200X1mg/15ml
Approval Date :
Application Number :
Regulatory Info : Generic
Registration Country : South Africa
 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info : Generic
Registration Country : South Africa
Brand Name : Throflam
Dosage Form : SPO
Dosage Strength : 22.5mg/15ml
Packaging : 30X1mg/15ml
Approval Date :
Application Number :
Regulatory Info : Generic
Registration Country : South Africa
 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info : Originator
Registration Country : South Africa
Brand Name : Andolex-C Oral Gel
Dosage Form : GEO
Dosage Strength : 10mg/g
Packaging : 1X1mg/g
Approval Date :
Application Number :
Regulatory Info : Originator
Registration Country : South Africa

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info : Originator
Registration Country : South Africa
Brand Name : Andolex-C Spray
Dosage Form : SPO
Dosage Strength : 22.5mg/15ml
Packaging : 50X1mg/15ml
Approval Date :
Application Number :
Regulatory Info : Originator
Registration Country : South Africa

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info : Originator
Registration Country : South Africa
Brand Name : Andolex Analgesic Lozenges
Dosage Form : LOZ
Dosage Strength : 3mg
Packaging : 24X1mg
Approval Date :
Application Number :
Regulatory Info : Originator
Registration Country : South Africa

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info : Originator
Registration Country : South Africa
Brand Name : Andolex-C Loz Raspberry
Dosage Form : LOZ
Dosage Strength : 3mg
Packaging : 16X1mg
Approval Date :
Application Number :
Regulatory Info : Originator
Registration Country : South Africa

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info : Originator
Registration Country : South Africa
Brand Name : Oranix
Dosage Form : MOW
Dosage Strength : 22.5 mg/15ml
Packaging : 200X1mg/15ml
Approval Date :
Application Number :
Regulatory Info : Originator
Registration Country : South Africa

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info : Generic
Registration Country : South Africa
Brand Name : Gesoral Oral Spray
Dosage Form : SPO
Dosage Strength : 22.5mg/15ml
Packaging : 30X1mg/15ml
Approval Date :
Application Number :
Regulatory Info : Generic
Registration Country : South Africa

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results] Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info : Generic
Registration Country : Turkey
Brand Name :
Dosage Form : Oral Spray
Dosage Strength : 0.15%
Packaging :
Approval Date :
Application Number :
Regulatory Info : Generic
Registration Country : Turkey

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Packaging :
Regulatory Info : Generic
Dosage : Oral Spray
Dosage Strength : 0.15%
Brand Name :
Approval Date :
Application Number :
Registration Country : Turkey

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info :
Registration Country : Saudi Arabia
Brand Name : Unitum Gel
Dosage Form : Gel
Dosage Strength : 50MG
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : Saudi Arabia

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Packaging :
Regulatory Info :
Dosage : Gel
Dosage Strength : 50MG
Brand Name : Unitum Gel
Approval Date :
Application Number :
Registration Country : Saudi Arabia

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info :
Registration Country : Ukraine
Brand Name : Septefril® Verde From...
Dosage Form : Spray
Dosage Strength : 1.5MG/1ML
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : Ukraine

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Packaging :
Regulatory Info :
Dosage : Spray
Dosage Strength : 1.5MG/1ML
Brand Name : Septefril® Verde From...
Approval Date :
Application Number :
Registration Country : Ukraine

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info :
Registration Country : Ukraine
Brand Name : Septefril® Verde From...
Dosage Form : Spray
Dosage Strength : 3MG/1ML
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : Ukraine

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Packaging :
Regulatory Info :
Dosage : Spray
Dosage Strength : 3MG/1ML
Brand Name : Septefril® Verde From...
Approval Date :
Application Number :
Registration Country : Ukraine

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info :
Registration Country : South Korea
Benzydamine Hydrochloride; Cetylpyridinium Chloride Anhydrous
Brand Name : Morgle One Q
Dosage Form : Spray
Dosage Strength : 1.5MG/1ML; 5MG/1ML
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : South Korea

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Packaging :
Regulatory Info :
Benzydamine Hydrochloride; Cetylpyridinium Chloride Anhydrous
Dosage : Spray
Dosage Strength : 1.5MG/1ML; 5MG/1ML
Brand Name : Morgle One Q
Approval Date :
Application Number :
Registration Country : South Korea

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info :
Registration Country : Poland
Brand Name : Hascosept
Dosage Form : Oral Solution
Dosage Strength : 1.5MG/GM
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : Poland

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Packaging :
Regulatory Info :
Dosage : Oral Solution
Dosage Strength : 1.5MG/GM
Brand Name : Hascosept
Approval Date :
Application Number :
Registration Country : Poland

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info :
Registration Country : India
Benzydamine Hydrochloride; Pholcodine; Cetylpyridinium
Brand Name :
Dosage Form : Lozenge
Dosage Strength : 1.5MG; 5.5MG; 1.33MG
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : India

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Packaging :
Regulatory Info :
Benzydamine Hydrochloride; Pholcodine; Cetylpyridinium
Dosage : Lozenge
Dosage Strength : 1.5MG; 5.5MG; 1.33MG
Brand Name :
Approval Date :
Application Number :
Registration Country : India

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]Global Sales Information

Market Place

Reply
04 Jun 2024
Reply
13 Oct 2023
Reply
13 Jun 2023
Reply
09 Jun 2023
Reply
17 Nov 2022
Reply
29 Jul 2022
Reply
26 Apr 2022
Reply
08 Jan 2022
Reply
23 Sep 2021
Reply
23 Jul 2021
 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
ABOUT THIS PAGE
43
PharmaCompass offers a list of Benzydamine Hydrochloride API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Benzydamine Hydrochloride manufacturer or Benzydamine Hydrochloride supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Benzydamine Hydrochloride manufacturer or Benzydamine Hydrochloride supplier.
A Benzydamine HCl manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Benzydamine HCl, including repackagers and relabelers. The FDA regulates Benzydamine HCl manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Benzydamine HCl API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
click here to find a list of Benzydamine HCl manufacturers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PhamaCompass.
A Benzydamine HCl supplier is an individual or a company that provides Benzydamine HCl active pharmaceutical ingredient (API) or Benzydamine HCl finished formulations upon request. The Benzydamine HCl suppliers may include Benzydamine HCl API manufacturers, exporters, distributors and traders.
click here to find a list of Benzydamine HCl suppliers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PharmaCompass.
In Korea, the Ministry of Food and Drug Safety (MFDS) is in charge of regulating pharmaceutical products and services.
Pharmaceutical companies submit a Benzydamine HCl Drug Master File in Korea (Benzydamine HCl KDMF) to the MFDS, which includes comprehensive information about the production, processing, facilities, materials, packaging, and testing of Benzydamine HCl. The MFDS reviews the Benzydamine HCl KDMF as part of the drug registration process and uses the information provided in the Benzydamine HCl KDMF to evaluate the safety and efficacy of the drug.
After submitting a Benzydamine HCl KDMF to the MFDS, the registered manufacturer can provide importers or distributors with the registration number without revealing confidential information to Korean business partners. Applicants seeking to register their Benzydamine HCl API can apply through the Korea Drug Master File (KDMF).
click here to find a list of Benzydamine HCl suppliers with KDMF on PharmaCompass.
A Benzydamine HCl CEP of the European Pharmacopoeia monograph is often referred to as a Benzydamine HCl Certificate of Suitability (COS). The purpose of a Benzydamine HCl CEP is to show that the European Pharmacopoeia monograph adequately controls the purity of Benzydamine HCl EP produced by a given manufacturer. Suppliers of raw materials can prove the suitability of Benzydamine HCl to their clients by showing that a Benzydamine HCl CEP has been issued for it. The manufacturer submits a Benzydamine HCl CEP (COS) as part of the market authorization procedure, and it takes on the role of a Benzydamine HCl CEP holder for the record. Additionally, the data presented in the Benzydamine HCl CEP (COS) is managed confidentially and offers a centralized system acknowledged by numerous nations, exactly like the Benzydamine HCl DMF.
A Benzydamine HCl CEP (COS) is recognised by all 36 nations that make up the European Pharmacopoeia Convention. Benzydamine HCl CEPs may be accepted in nations that are not members of the Ph. Eur. at the discretion of the authorities there.
click here to find a list of Benzydamine HCl suppliers with CEP (COS) on PharmaCompass.
A Benzydamine HCl written confirmation (Benzydamine HCl WC) is an official document issued by a regulatory agency to a Benzydamine HCl manufacturer, verifying that the manufacturing facility of a Benzydamine HCl active pharmaceutical ingredient (API) adheres to the Good Manufacturing Practices (GMP) regulations of the importing country. When exporting Benzydamine HCl APIs or Benzydamine HCl finished pharmaceutical products to another nation, regulatory agencies frequently require a Benzydamine HCl WC (written confirmation) as part of the regulatory process.
click here to find a list of Benzydamine HCl suppliers with Written Confirmation (WC) on PharmaCompass.
Benzydamine HCl Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Benzydamine HCl GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Benzydamine HCl GMP manufacturer or Benzydamine HCl GMP API supplier for your needs.
A Benzydamine HCl CoA (Certificate of Analysis) is a formal document that attests to Benzydamine HCl's compliance with Benzydamine HCl specifications and serves as a tool for batch-level quality control.
Benzydamine HCl CoA mostly includes findings from lab analyses of a specific batch. For each Benzydamine HCl CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Benzydamine HCl may be tested according to a variety of international standards, such as European Pharmacopoeia (Benzydamine HCl EP), Benzydamine HCl JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Benzydamine HCl USP).

