
Synopsis
Synopsis
0
VMF
0
Australia
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
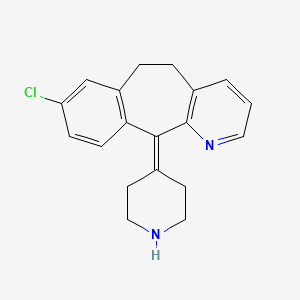

1. Aerius
2. Clarinex
3. Descarboethoxyloratadine
4. Descarboethoxyloratadine Acetate
5. Neoclarityn
6. Sch 34117
7. Sch-34117
1. 100643-71-8
2. Clarinex
3. Descarboethoxyloratadine
4. Desloratidine
5. Neoclarityn
6. Aerius
7. Azomyr
8. Sch-34117
9. Denosin
10. Allex
11. Opulis
12. Sch 34117
13. 8-chloro-11-piperidin-4-ylidene-6,11-dihydro-5h-benzo[5,6]cyclohepta[1,2-b]pyridine
14. Descarboethoxyoratidine
15. Desloratadine Actavis
16. Dasselta
17. Desloratadine Teva
18. Mfcd00871949
19. 8-chloro-11-(piperidin-4-ylidene)-6,11-dihydro-5h-benzo[5,6]cyclohepta[1,2-b]pyridine
20. Nsc-759824
21. Chembl1172
22. Loratadine Related Compound A
23. 8-chloro-6,11-dihydro-11-(4-piperidinylidene)-5h-benzo(5,6)cyclohepta(1,2-b)pyridine
24. Mls000559042
25. Chebi:291342
26. Fvf865388r
27. Nsc675447
28. 5h-benzo[5,6]cyclohepta[1,2-b]pyridine, 8-chloro-6,11-dihydro-11-(4-piperidinylidene)-
29. 13-chloro-2-piperidin-4-ylidene-4-azatricyclo[9.4.0.03,8]pentadeca-1(11),3(8),4,6,12,14-hexaene
30. Ncgc00159325-02
31. Smr000149358
32. Desloratadine [usan]
33. Clarinex Reditabs
34. 8-chloro-6,11-dihydro-11-(4-piperidinylidene)-5h-benzo[5,6]cyclohepta[1,2-b]pyridine
35. Dsstox_cid_24196
36. Dsstox_rid_80112
37. Dsstox_gsid_44196
38. Desalex
39. 5h-benzo(5,6)cyclohepta(1,2-b)pyridine, 8-chloro-6,11-dihydro-11-(4-piperidinylidene)-
40. 8-chloro-11-piperidin-4-ylidene-5,6-dihydrobenzo[1,2]cyclohepta[2,4-b]pyridine
41. 8-chloro-6,11-dihydro-11-(4-piperdinylidene)- 5h-benzo[5,6]cyclohepta[1,2-b]pyridine
42. Clarinex (tn)
43. 13-chloro-2-(piperidin-4-ylidene)-4-azatricyclo[9.4.0.0^{3,8}]pentadeca-1(11),3(8),4,6,12,14-hexaene
44. 8-chloro-11-(piperidin-4-ylidene)-6,11-dihydro-5h-benzo[5,6]cyclohepta[1,2-b]pyridine (descarboethoxyloratadine; Desloratadine)
45. Cas-100643-71-8
46. Sr-01000668962
47. Unii-fvf865388r
48. Desloratadine [usan:inn:ban]
49. 4-(8-chloro-5,6-dihydro-11h-benzo[5,6]cyclohepta(1,2-b]pyridin-11-ylidene)-piperidine
50. 4-(8-chloro-5,6-dihydro-11h-benzo[5,6]cyclohepta[1,2-b]pyridin-11-ylidene)piperidine
51. 4-{8-chloro-5,6-dihydro-11h-benzo[5,6]cyclohepta[1,2-b]pyridin-11-ylidene}-piperidine
52. 4-{8-chloro-5,6-dihydro-11h-benzo[5,6]cyclohepta[1,2-b]pyridin-11-ylidene}piperidine
53. Desloratadine-[d7]
54. Desloratadine- Bio-x
55. Mk-4117
56. Cpd000149358
57. Opera_id_1891
58. Desloratadine [mi]
59. Desloratadine [inn]
60. Desloratadine [jan]
61. Schembl4425
62. Desloratadine [vandf]
63. Mls000759406
64. Mls001201801
65. Mls001424247
66. Desloratadine [mart.]
67. Sch34117
68. Desloratadine Ratiopharm
69. Desloratadine [usp-rs]
70. Desloratadine [who-dd]
71. Desloratadine (jan/usp/inn)
72. Gtpl7157
73. Zinc1261
74. Desloratadine [ema Epar]
75. Dtxsid1044196
76. Loratadine Related Compound A Rs
77. Hms2052h05
78. Hms2090c06
79. Hms2093f19
80. Hms3394h05
81. Hms3652o15
82. Hms3715j15
83. Hms3885c18
84. Pharmakon1600-01505393
85. Desloratadine [orange Book]
86. Albb-027276
87. Bcp02340
88. Desloratadine For System Suitability
89. Hy-b0539
90. Desloratadine [ep Monograph]
91. Tox21_111574
92. Bbl010777
93. Bdbm50073179
94. Desloratadine [usp Monograph]
95. Nsc759824
96. S4012
97. Stk586537
98. Akos000280921
99. Tox21_111574_1
100. Ac-1279
101. Ccg-101162
102. Db00967
103. Ks-1048
104. Nc00412
105. Nsc 675447
106. Nsc 759824
107. Nsc-675447
108. Sb17503
109. Clarinex-d Component Desloratadine
110. Desloratadine 100 Microg/ml In Methanol
111. Desloratadine, Powder, >=98% (hplc)
112. Ncgc00159325-03
113. Ncgc00159325-04
114. Ncgc00159325-05
115. Bd164361
116. Loratadine Ep Impurity D (desloratadine)
117. Sbi-0206828.p001
118. Loratadine Impurity D [ep Impurity]
119. D3787
120. Desloratadine Component Of Clarinex-d
121. Ft-0602522
122. Ft-0666048
123. Sw197792-3
124. A19515
125. D03693
126. H11943
127. Loratadine Related Compound A [usp-rs]
128. Ab00456701-11
129. Ab00456701-13
130. Ab00456701_14
131. Ab00456701_15
132. 643d718
133. L001025
134. Q418060
135. Loratadine Related Compound A [usp Impurity]
136. Q-200936
137. Rupatadine Fumarate Impurity B [ep Impurity]
138. Sr-01000668962-4
139. Sr-01000668962-5
140. Sr-01000668962-8
141. Brd-k82357231-001-13-4
142. Desloratadine Teva (authorized: Rhinitis, Urticaria)
143. Desloratadine Actavis (authorized: Rhinitis, Urticaria)
144. Desloratadine, European Pharmacopoeia (ep) Reference Standard
145. Desloratadine Ratiopharm (authorized: Rhinitis, Urticaria)
146. Desloratadine, United States Pharmacopeia (usp) Reference Standard
147. 4-(8-chloro-5,6-dihydro-11 H-benzo[5,6]cyclohepta[1,2-b]pyridin-11-ylidene)piperidine
148. 4-(8-chloro-5,6-dihydro-11h-benzo-[5,6]cyclohepta(1,2-b]pyridin-11-ylidene)-piperidine
149. 4-(8-chloro-5,6-dihydro-11h-benzo[5,6]cyclohepta [1,2-b]pyridin-11-ylidene) Piperidine
150. 4-(8-chloro-5,6-dihydro-11h-benzo[5,6]cyclohepta[1,2-b]pyridin-11-ylidene) Piperidine
151. 8-chloro-11-(4-piperidinylidene)-6,11-dihydro-5h- Benzo[4,5]cyclohepta[2,1-b]pyridine
152. 8-chloro-11-(4-piperidyliden)-6,11-dihydro-5h-benzo[5,6]cyclohepta [1,2-b]pyridine
153. 8-chloro-11-(4-piperidyliden)-6,11-dihydro-5h-benzo[5,6]cyclohepta[1,2-b]pyridine
154. 8-chloro-11-(4-piperidylidene)-6,11-dihydro-5h-benzo[5.6]cyclohepta[1,2-b]pyridine
155. 8-chloro-6,11-dihydro-11-(4-piperidylidene)-5h-benzo[5,6]cyclohepta[1,2-b]pyridine
156. Desloratadine For System Suitability, European Pharmacopoeia (ep) Reference Standard
157. Desloratidine, Pharmaceutical Secondary Standard; Certified Reference Material
158. Loratadine Related Compound A, United States Pharmacopeia (usp) Reference Standard
159. 13-chloro-2-(piperidin-4-ylidene)-4-azatricyclo[9.4.0.0^{3,8}]pentadeca-1(11),3,5,7,12,14-hexaene
160. 8-chloro-11-(4-piperidylidene)-6,11-dihydro-5h-benzo[5,6]cyclohepta[1,2-b]pyridine
161. 8-chloro-6,11-dihydro-11(4-piperidylidene)-5h-benzo(5,6)cyclohepta(1,2-b) Pyridine
162. 8-chloro-6,11-dihydro-11-(4-piperidinylidene)-5h-benzo(5,6)cyclohepta(1,2-bpyridine
163. 8-chloro-6,11-dihydro-11-(4-piperidinylidene)-5h-benzo(5,6)cyclohepta(1,2b)pyridine
| Molecular Weight | 310.8 g/mol |
|---|---|
| Molecular Formula | C19H19ClN2 |
| XLogP3 | 4.5 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 2 |
| Rotatable Bond Count | 0 |
| Exact Mass | 310.1236763 g/mol |
| Monoisotopic Mass | 310.1236763 g/mol |
| Topological Polar Surface Area | 24.9 Ų |
| Heavy Atom Count | 22 |
| Formal Charge | 0 |
| Complexity | 425 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
| 1 of 4 | |
|---|---|
| Drug Name | Clarinex |
| PubMed Health | Desloratadine (By mouth) |
| Drug Classes | Respiratory Agent |
| Drug Label | CLARINEX (desloratadine) Tablets are light blue, round, film-coated tablets containing 5 mg desloratadine, an antihistamine, to be administered orally. CLARINEX Tablets also contain the following excipients: dibasic calcium phosphate dihydrate USP, m... |
| Active Ingredient | Desloratadine |
| Dosage Form | Tablet, orally disintegrating; Tablet; Syrup; Solution |
| Route | oral; Oral |
| Strength | 2.5mg; 5mg; 0.5mg/ml |
| Market Status | Prescription |
| Company | Merck Sharp Dohme; Schering |
| 2 of 4 | |
|---|---|
| Drug Name | Desloratadine |
| PubMed Health | Desloratadine (By mouth) |
| Drug Classes | Respiratory Agent |
| Drug Label | Desloratadine tablets are blue, round, film-coated tablets debossed with logo L5 on one side containing 5 mg desloratadine, an antihistamine, to be administered orally. Desloratadine tablets also contain the following excipients: carnauba wax,... |
| Active Ingredient | Desloratadine |
| Dosage Form | Tablet, orally disintegrating; Tablet |
| Route | oral; Oral |
| Strength | 2.5mg; 5mg |
| Market Status | Tentative Approval; Prescription |
| Company | Perrigo R And D; Mylan Pharms; Glenmark Generics; Belcher Pharms; Lupin Pharms; Sun Pharm Inds; Reddys; Zydus Pharms Usa; Dr Reddys Labs; Sandoz; Orchid Hlthcare |
| 3 of 4 | |
|---|---|
| Drug Name | Clarinex |
| PubMed Health | Desloratadine (By mouth) |
| Drug Classes | Respiratory Agent |
| Drug Label | CLARINEX (desloratadine) Tablets are light blue, round, film-coated tablets containing 5 mg desloratadine, an antihistamine, to be administered orally. CLARINEX Tablets also contain the following excipients: dibasic calcium phosphate dihydrate USP, m... |
| Active Ingredient | Desloratadine |
| Dosage Form | Tablet, orally disintegrating; Tablet; Syrup; Solution |
| Route | oral; Oral |
| Strength | 2.5mg; 5mg; 0.5mg/ml |
| Market Status | Prescription |
| Company | Merck Sharp Dohme; Schering |
| 4 of 4 | |
|---|---|
| Drug Name | Desloratadine |
| PubMed Health | Desloratadine (By mouth) |
| Drug Classes | Respiratory Agent |
| Drug Label | Desloratadine tablets are blue, round, film-coated tablets debossed with logo L5 on one side containing 5 mg desloratadine, an antihistamine, to be administered orally. Desloratadine tablets also contain the following excipients: carnauba wax,... |
| Active Ingredient | Desloratadine |
| Dosage Form | Tablet, orally disintegrating; Tablet |
| Route | oral; Oral |
| Strength | 2.5mg; 5mg |
| Market Status | Tentative Approval; Prescription |
| Company | Perrigo R And D; Mylan Pharms; Glenmark Generics; Belcher Pharms; Lupin Pharms; Sun Pharm Inds; Reddys; Zydus Pharms Usa; Dr Reddys Labs; Sandoz; Orchid Hlthcare |
For the relief of symptoms of seasonal allergic rhinitis, perennial (non-seasonal) allergic rhinitis. Desloratidine is also used for the sympomatic treatment of pruritus and urticaria (hives) associated with chronic idiopathic urticaria.
FDA Label
Azomyr is indicated for the relief of symptoms associated with:
- allergic rhinitis (see section 5. 1)
- urticaria (see section 5. 1)
Aerius is indicated for the relief of symptoms associated with:
- allergic rhinitis;
- urticaria.
Neoclarityn is indicated for the relief of symptoms associated with:
- allergic rhinitis
- urticaria
Treatment of allergic rhinitis and urticaria.
Desloratadine Teva is indicated for the relief of symptoms associated with:
- allergic rhinitis;
- urticaria.
Dasselta is indicated for the relief of symptoms associated with:
- allergic rhinitis;
- urticaria.
Desloratadine ratiopharm is indicated in adults for the relief of symptoms associated with:
- allergic rhinitis
- chronic idiopathic urticaria as initially diagnosed by a physician
Aerius is indicated for the relief of symptoms associated with:
- allergic rhinitis (see section 5. 1)
- urticaria (see section 5. 1)
Opulis is indicated for the relief of symptoms associated with:
- allergic rhinitis (see section 5. 1)
- urticaria (see section 5. 1)
Desloratadine is a long-acting second-generation H1-receptor antagonist which has a selective and peripheral H1-antagonist action. Histamine is a chemical that causes many of the signs that are part of allergic reactions, such as the swelling of tissues. Histamine is released from histamine-storing cells (mast cells) and attaches to other cells that have receptors for histamine. The attachment of the histamine to the receptors causes the cell to be "activated," releasing other chemicals which produce the effects that we associate with allergies. Desloratadine blocks one type of receptor for histamine (the H1 receptor) and thus prevents activation of cells by histamine. Unlike most other antihistamines, Desloratadine does not enter the brain from the blood and, therefore, does not cause drowsiness.
Cholinergic Antagonists
Drugs that bind to but do not activate CHOLINERGIC RECEPTORS, thereby blocking the actions of ACETYLCHOLINE or cholinergic agonists. (See all compounds classified as Cholinergic Antagonists.)
Histamine H1 Antagonists, Non-Sedating
A class of non-sedating drugs that bind to but do not activate histamine receptors (DRUG INVERSE AGONISM), thereby blocking the actions of histamine or histamine agonists. These antihistamines represent a heterogenous group of compounds with differing chemical structures, adverse effects, distribution, and metabolism. Compared to the early (first generation) antihistamines, these non-sedating antihistamines have greater receptor specificity, lower penetration of BLOOD-BRAIN BARRIER, and are less likely to cause drowsiness or psychomotor impairment. (See all compounds classified as Histamine H1 Antagonists, Non-Sedating.)
R06AX27
R06AX27
R06AX27
R06AX27
R06AX27
R06AX27
R06AX27
R06AX27
R06AX27
R06AX27
S76 | LUXPHARMA | Pharmaceuticals Marketed in Luxembourg | Pharmaceuticals marketed in Luxembourg, as published by d'Gesondheetskeess (CNS, la caisse nationale de sante, www.cns.lu), mapped by name to structures using CompTox by R. Singh et al. (in prep.). List downloaded from https://cns.public.lu/en/legislations/textes-coordonnes/liste-med-comm.html. Dataset DOI:10.5281/zenodo.4587355
R - Respiratory system
R06 - Antihistamines for systemic use
R06A - Antihistamines for systemic use
R06AX - Other antihistamines for systemic use
R06AX27 - Desloratadine
Absorption
Desloratadine administered orally for ten days to healthy volunteers as a 5 mg tablet once daily resulted in a mean Tmax of approximately 3 hours, a mean steady-state Cmax of 4 ng/ml, and a mean steady-state AUC of 56.9 ng\*hr/ml. A similar profile was observed using 10 ml of an oral solution containing 5 mg of desloratadine. Food was found not to affect desloratadine absorption.
Route of Elimination
Approximately 87% of a 14C-desloratadine dose was equally recovered in urine and feces as metabolic products.
Desloratadine is metabolized to the active metabolite 3-hydroxydesloratadine, which is subsequently glucuronidated.
Desloratadine is a known human metabolite of Rupatadine and loratadine.
S73 | METXBIODB | Metabolite Reaction Database from BioTransformer | DOI:10.5281/zenodo.4056560
Desloratadine has a mean plasma elimination half-life of approximately 27 hours.
Like other H1-blockers, Desloratadine competes with free histamine for binding at H1-receptors in the GI tract, uterus, large blood vessels, and bronchial smooth muscle. This blocks the action of endogenous histamine, which subsequently leads to temporary relief of the negative symptoms (eg. nasal congestion, watery eyes) brought on by histamine.
CAS Number : 5570-77-4
End Use API : Desloratadine
About The Company : Vamsi Labs, established in 1991, is a leading Indian API manufacturer specializing in anti-asthmatic, anti-migraine, and anti-psychotic APIs for domestic and in...
2-Cyano-3-[2(3-chloro phenyl) ethyl]pyridine (L-5)
CAS Number : 31255-57-9
End Use API : Desloratadine
About The Company : Tagoor Laboratories, established in 2018 and part of the Tagoor Group, provides APIs, advanced intermediates, and key starting materials for critical and high-g...
8-Chloro-6,11-dihydro-11-(1-methyl-4-piperidinylid...
CAS Number : 38092-89-6
End Use API : Desloratadine
About The Company : Tagoor Laboratories, established in 2018 and part of the Tagoor Group, provides APIs, advanced intermediates, and key starting materials for critical and high-g...
CAS Number : 3222-49-9
End Use API : Desloratadine
About The Company : Tagoor Laboratories, established in 2018 and part of the Tagoor Group, provides APIs, advanced intermediates, and key starting materials for critical and high-g...
3-(Chloromethyl)-5-methylpyridine hydrochloride
CAS Number : 1007089-84-0
End Use API : Desloratadine
About The Company : Tagoor Laboratories, established in 2018 and part of the Tagoor Group, provides APIs, advanced intermediates, and key starting materials for critical and high-g...
CAS Number : 5570-77-4
End Use API : Desloratadine
About The Company : Tagoor Laboratories, established in 2018 and part of the Tagoor Group, provides APIs, advanced intermediates, and key starting materials for critical and high-g...
3(2-(3-Chloro phenyl) ethyl) cyano pyridine
CAS Number : 31255-55-7
End Use API : Desloratadine
About The Company : VASUDHA PHARMA CHEM LIMITED was incorporated, as a public limited company under the Companies Act, 1956 in 1994-95 at Hyderabad in the state of Telangana, India...

8-Chloro-5,6dihydro (1H)-benzo [5,6] cyclohepta (...
CAS Number : 31251-41-9
End Use API : Desloratadine
About The Company : VASUDHA PHARMA CHEM LIMITED was incorporated, as a public limited company under the Companies Act, 1956 in 1994-95 at Hyderabad in the state of Telangana, India...

8 chloro-11-[(N-Methyl)-4-piperidylidene]- 6, 11-d...
CAS Number : 38092-89-6
End Use API : Desloratadine
About The Company : VASUDHA PHARMA CHEM LIMITED was incorporated, as a public limited company under the Companies Act, 1956 in 1994-95 at Hyderabad in the state of Telangana, India...

cyclohepta (1,2b) pyridine (Methyl Loratadine)
CAS Number : 38092-89-6
End Use API : Desloratadine
About The Company : VASUDHA PHARMA CHEM LIMITED was incorporated, as a public limited company under the Companies Act, 1956 in 1994-95 at Hyderabad in the state of Telangana, India...

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]DRUG PRODUCT COMPOSITIONS

Related Excipient Companies

Excipients by Applications
Global Sales Information

Market Place

Patents & EXCLUSIVITIES
REF. STANDARDS & IMPURITIES

ANALYTICAL

ABOUT THIS PAGE
37
PharmaCompass offers a list of Desloratadine API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Desloratadine manufacturer or Desloratadine supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Desloratadine manufacturer or Desloratadine supplier.
A Azomyr manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Azomyr, including repackagers and relabelers. The FDA regulates Azomyr manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Azomyr API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
click here to find a list of Azomyr manufacturers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PhamaCompass.
A Azomyr supplier is an individual or a company that provides Azomyr active pharmaceutical ingredient (API) or Azomyr finished formulations upon request. The Azomyr suppliers may include Azomyr API manufacturers, exporters, distributors and traders.
click here to find a list of Azomyr suppliers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PharmaCompass.
A Azomyr DMF (Drug Master File) is a document detailing the whole manufacturing process of Azomyr active pharmaceutical ingredient (API) in detail. Different forms of Azomyr DMFs exist exist since differing nations have different regulations, such as Azomyr USDMF, ASMF (EDMF), JDMF, CDMF, etc.
A Azomyr DMF submitted to regulatory agencies in the US is known as a USDMF. Azomyr USDMF includes data on Azomyr's chemical properties, information on the facilities and procedures used, and details about packaging and storage. The Azomyr USDMF is kept confidential to protect the manufacturer’s intellectual property.
click here to find a list of Azomyr suppliers with USDMF on PharmaCompass.
The Pharmaceuticals and Medical Devices Agency (PMDA) established the Japan Drug Master File (JDMF), also known as the Master File (MF), to permit Japanese and foreign manufacturers of drug substances, intermediates, excipients, raw materials, and packaging materials (‘Products’) to voluntarily register confidential information about the production and management of their products in Japan.
The Azomyr Drug Master File in Japan (Azomyr JDMF) empowers Azomyr API manufacturers to present comprehensive information (e.g., production methods, data, etc.) to the review authority, i.e., PMDA (Pharmaceuticals & Medical Devices Agency).
PMDA reviews the Azomyr JDMF during the approval evaluation for pharmaceutical products. At the time of Azomyr JDMF registration, PMDA checks if the format is accurate, if the necessary items have been included (application), and if data has been attached.
click here to find a list of Azomyr suppliers with JDMF on PharmaCompass.
In Korea, the Ministry of Food and Drug Safety (MFDS) is in charge of regulating pharmaceutical products and services.
Pharmaceutical companies submit a Azomyr Drug Master File in Korea (Azomyr KDMF) to the MFDS, which includes comprehensive information about the production, processing, facilities, materials, packaging, and testing of Azomyr. The MFDS reviews the Azomyr KDMF as part of the drug registration process and uses the information provided in the Azomyr KDMF to evaluate the safety and efficacy of the drug.
After submitting a Azomyr KDMF to the MFDS, the registered manufacturer can provide importers or distributors with the registration number without revealing confidential information to Korean business partners. Applicants seeking to register their Azomyr API can apply through the Korea Drug Master File (KDMF).
click here to find a list of Azomyr suppliers with KDMF on PharmaCompass.
A Azomyr CEP of the European Pharmacopoeia monograph is often referred to as a Azomyr Certificate of Suitability (COS). The purpose of a Azomyr CEP is to show that the European Pharmacopoeia monograph adequately controls the purity of Azomyr EP produced by a given manufacturer. Suppliers of raw materials can prove the suitability of Azomyr to their clients by showing that a Azomyr CEP has been issued for it. The manufacturer submits a Azomyr CEP (COS) as part of the market authorization procedure, and it takes on the role of a Azomyr CEP holder for the record. Additionally, the data presented in the Azomyr CEP (COS) is managed confidentially and offers a centralized system acknowledged by numerous nations, exactly like the Azomyr DMF.
A Azomyr CEP (COS) is recognised by all 36 nations that make up the European Pharmacopoeia Convention. Azomyr CEPs may be accepted in nations that are not members of the Ph. Eur. at the discretion of the authorities there.
click here to find a list of Azomyr suppliers with CEP (COS) on PharmaCompass.
A Azomyr written confirmation (Azomyr WC) is an official document issued by a regulatory agency to a Azomyr manufacturer, verifying that the manufacturing facility of a Azomyr active pharmaceutical ingredient (API) adheres to the Good Manufacturing Practices (GMP) regulations of the importing country. When exporting Azomyr APIs or Azomyr finished pharmaceutical products to another nation, regulatory agencies frequently require a Azomyr WC (written confirmation) as part of the regulatory process.
click here to find a list of Azomyr suppliers with Written Confirmation (WC) on PharmaCompass.
National Drug Code is a comprehensive database maintained by the FDA that contains information on all drugs marketed in the US. This directory includes information about finished drug products, unfinished drug products, and compounded drug products, including those containing Azomyr as an active pharmaceutical ingredient (API).
The FDA updates the NDC directory daily. The NDC numbers for Azomyr API and other APIs are published in this directory by the FDA.
The NDC unfinished drugs database includes product listing information submitted for all unfinished drugs, such as active pharmaceutical ingredients (APIs), drugs intended for further processing and bulk drug substances for compounding.
Pharmaceutical companies that manufacture Azomyr as an active pharmaceutical ingredient (API) must furnish the FDA with an updated record of all drugs that they produce, prepare, propagate, compound, or process for commercial distribution in the US at their facilities.
The NDC directory also contains data on finished compounded human drug products that contain Azomyr and are produced by outsourcing facilities. While these outsourcing facilities are not mandated to assign a Azomyr NDC to their finished compounded human drug products, they may choose to do so.
click here to find a list of Azomyr suppliers with NDC on PharmaCompass.
Azomyr Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Azomyr GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Azomyr GMP manufacturer or Azomyr GMP API supplier for your needs.
A Azomyr CoA (Certificate of Analysis) is a formal document that attests to Azomyr's compliance with Azomyr specifications and serves as a tool for batch-level quality control.
Azomyr CoA mostly includes findings from lab analyses of a specific batch. For each Azomyr CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Azomyr may be tested according to a variety of international standards, such as European Pharmacopoeia (Azomyr EP), Azomyr JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Azomyr USP).

