
Synopsis
Synopsis
0
CEP/COS
0
JDMF
0
VMF
0
EDQM
0
USP
0
JP
0
Others
0
South Africa
0
Listed Dossiers
DRUG PRODUCT COMPOSITIONS
Regulatory FDF Prices
NA
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
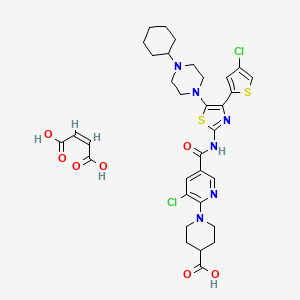

1. E5501 Maleate
2. 677007-74-8
3. Avatrombopag Maleate [usan]
4. Avatrombopag Meleate
5. Avatrombopag (maleate)
6. Gdw7m2p1is
7. E-5501 Maleate
8. (z)-but-2-enedioic Acid;1-[3-chloro-5-[[4-(4-chlorothiophen-2-yl)-5-(4-cyclohexylpiperazin-1-yl)-1,3-thiazol-2-yl]carbamoyl]pyridin-2-yl]piperidine-4-carboxylic Acid
9. 4-piperidinecarboxylic Acid, 1-(3-chloro-5-(((4-(4-chloro-2-thienyl)-5-(4-cyclohexyl-1- Piperazinyl)-2-thiazolyl)amino)carbonyl)-2-pyridinyl)-, (2z)-2-butenedioate (1:1)
10. Unii-gdw7m2p1is
11. Doptelet (tn)
12. Akr-501 Monomaleate
13. Chembl2105758
14. Schembl19610454
15. Avatrombopag Maleate [mi]
16. Avatrombopag Maleate (jan/usan)
17. Avatrombopag Maleate [jan]
18. Dtxsid001027855
19. 677007-74-8 (maleate)
20. Hy-13463a
21. Avatrombopag Maleate [who-dd]
22. Avatrombopag Maleate [orange Book]
23. Cs-0025423
24. D10307
25. A902459
26. Q27279052
27. 1-(3-chloro-5-((4-(4-chlorothiophen-2-yl)-5-(4-cyclohexylpiperazin-1-yl)thiazol-2- Yl)carbamoyl)pyridin-2-yl)piperidine-4-carboxylic Acid Mono-(2z)-but-2-enedioate
28. 1-[3-chloro-5-[[4-(4-chlorothiophen-2-yl)-5-(4-cyclohexylpiperazin-1-yl)-1,3-thiazol-2-yl]carbamoyl]pyridin-2-yl]piperidine-4-carboxylic Acid;avatrombopag
| Molecular Weight | 765.7 g/mol |
|---|---|
| Molecular Formula | C33H38Cl2N6O7S2 |
| Hydrogen Bond Donor Count | 4 |
| Hydrogen Bond Acceptor Count | 14 |
| Rotatable Bond Count | 9 |
| Exact Mass | 764.1620453 g/mol |
| Monoisotopic Mass | 764.1620453 g/mol |
| Topological Polar Surface Area | 233 Ų |
| Heavy Atom Count | 50 |
| Formal Charge | 0 |
| Complexity | 1050 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 1 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 2 |
Doptelet is indicated for the treatment of severe thrombocytopenia in adult patients with chronic liver disease who are scheduled to undergo an invasive procedure.
Doptelet is indicated for the treatment of primary chronic immune thrombocytopenia (ITP) in adult patients who are refractory to other treatments (e. g. corticosteroids, immunoglobulins).
Treatment of Idiopathic Thrombocytopenia Purpura (ITP), Treatment of Thrombocytopenic Purpura Secondary to Liver Disease
B02BX
 Biophore is a research-driven global pharmaceutical company focused on niche APIs for the generic industry.
Biophore is a research-driven global pharmaceutical company focused on niche APIs for the generic industry.
 Click Us!
Click Us! 
GDUFA
DMF Review : Reviewed
Rev. Date : 2025-10-22
Pay. Date : 2025-09-17
DMF Number : 41015
Submission : 2024-12-28
Status : Active
Type : II

 Granules India Limited has high volume world-class facilities for APIs, PFIs, & FDFs, serving customers in over 80 countries.
Granules India Limited has high volume world-class facilities for APIs, PFIs, & FDFs, serving customers in over 80 countries.

GDUFA
DMF Review : Reviewed
Rev. Date : 2025-12-16
Pay. Date : 2025-11-18
DMF Number : 41872
Submission : 2025-07-01
Status : Active
Type : II

NDC Package Code : 62207-025
Start Marketing Date : 2025-03-12
End Marketing Date : 2026-12-31
Dosage Form (Strength) : POWDER (1kg/kg)
Marketing Category : DRUG FOR FURTHER PROCESSING

 ChemWerth works in generic API development & supply, non-infringement patent strategy development and regulatory support.
ChemWerth works in generic API development & supply, non-infringement patent strategy development and regulatory support.
 Metrochem has been delivering customized volume & quality products to customers across the world, taking utmost care of their needs.
Metrochem has been delivering customized volume & quality products to customers across the world, taking utmost care of their needs.
 Nuray is an expert in the synthesis of Niche novel APIs, the first to launch Generics, NCEs, Advanced Intermediates // USFDA certified.
Nuray is an expert in the synthesis of Niche novel APIs, the first to launch Generics, NCEs, Advanced Intermediates // USFDA certified.

Date of Issue : 2024-02-20
Valid Till : 2026-12-06
Written Confirmation Number : WC-0416
Address of the Firm :
 Honour is a leading global CDMO and specialty chemicals manufacturer with seven world-class sites delivering quality-driven solutions.
Honour is a leading global CDMO and specialty chemicals manufacturer with seven world-class sites delivering quality-driven solutions.

GDUFA
DMF Review : Reviewed
Rev. Date : 2026-01-20
Pay. Date : 2025-11-28
DMF Number : 43091
Submission : 2025-11-27
Status : Active
Type : II

NDC Package Code : 82708-0014
Start Marketing Date : 2018-05-21
End Marketing Date : 2026-12-31
Dosage Form (Strength) : POWDER (1kg/kg)
Marketing Category : BULK INGREDIENT

 Hiray Pharma Solutions partners with global innovators to develop, manufacture and commercialize high-quality APIs and intermediates.
Hiray Pharma Solutions partners with global innovators to develop, manufacture and commercialize high-quality APIs and intermediates.
 Honour is a leading global CDMO and specialty chemicals manufacturer with seven world-class sites delivering quality-driven solutions.
Honour is a leading global CDMO and specialty chemicals manufacturer with seven world-class sites delivering quality-driven solutions.

NDC Package Code : 82708-0015
Start Marketing Date : 2018-05-21
End Marketing Date : 2026-12-31
Dosage Form (Strength) : POWDER (1kg/kg)
Marketing Category : BULK INGREDIENT


GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 38491
Submission : 2023-06-26
Status : Active
Type : II

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
 Biophore is a research-driven global pharmaceutical company focused on niche APIs for the generic industry.
Biophore is a research-driven global pharmaceutical company focused on niche APIs for the generic industry.
GDUFA
DMF Review : Complete
Rev. Date : 2025-10-22
Pay. Date : 2025-09-17
DMF Number : 41015
Submission : 2024-12-28
Status : Active
Type : II
 Granules India Limited has high volume world-class facilities for APIs, PFIs, & FDFs, serving customers in over 80 countries.
Granules India Limited has high volume world-class facilities for APIs, PFIs, & FDFs, serving customers in over 80 countries.
GDUFA
DMF Review : Complete
Rev. Date : 2025-12-16
Pay. Date : 2025-11-18
DMF Number : 41872
Submission : 2025-07-01
Status : Active
Type : II
 Honour is a leading global CDMO and specialty chemicals manufacturer with seven world-class sites delivering quality-driven solutions.
Honour is a leading global CDMO and specialty chemicals manufacturer with seven world-class sites delivering quality-driven solutions.
GDUFA
DMF Review : Complete
Rev. Date : 2026-01-20
Pay. Date : 2025-11-28
DMF Number : 43091
Submission : 2025-11-27
Status : Active
Type : II
GDUFA
DMF Review : Complete
Rev. Date : 2025-01-10
Pay. Date : 2024-12-04
DMF Number : 35784
Submission : 2021-03-31
Status : Active
Type : II

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 38491
Submission : 2023-06-26
Status : Active
Type : II

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 41921
Submission : 2025-05-15
Status : Active
Type : II

GDUFA
DMF Review : Complete
Rev. Date : 2025-06-10
Pay. Date : 2025-04-23
DMF Number : 41803
Submission : 2025-04-30
Status : Active
Type : II

USDMF
GDUFA
DMF Review : Complete
Rev. Date : 2026-04-03
Pay. Date : 2026-01-02
DMF Number : 43384
Submission : 2026-01-06
Status : Active
Type : II

Portfolio PDF
Product Web Link
Virtual Booth
Digital Content
Website
Corporate PDF
USDMF
GDUFA
DMF Review : Complete
Rev. Date : 2025-01-06
Pay. Date : 2024-12-16
DMF Number : 40697
Submission : 2024-10-28
Status : Active
Type : II

Portfolio PDF
Product Web Link
Virtual Booth
Digital Content
Website
Corporate PDF
USDMF
GDUFA
DMF Review : Complete
Rev. Date : 2026-04-24
Pay. Date : 2026-03-16
DMF Number : 43423
Submission : 2025-12-31
Status : Active
Type : II

Portfolio PDF
Product Web Link
Virtual Booth
Digital Content
Website
Corporate PDF
 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results] Nuray is an expert in the synthesis of Niche novel APIs, the first to launch Generics, NCEs, Advanced Intermediates // USFDA certified.
Nuray is an expert in the synthesis of Niche novel APIs, the first to launch Generics, NCEs, Advanced Intermediates // USFDA certified.
Date of Issue : 2024-02-20
Valid Till : 2026-12-06
Written Confirmation Number : WC-0416
Address of the Firm : Plot No. 111, SIDCO Industrial Estate, Kakkalur, Thiruvallur 602 003, Tamil Nadu...
 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]Registrant Name : Handok Consumption Co., Ltd.
Registration Date : 2024-11-26
Registration Number : Su318-7-ND
Manufacturer Name : Sterling Wisconsin LLC
Manufacturer Address : W130 N10497 Washington Drive Germantown, WI, 53022, USA

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results] Biophore is a research-driven global pharmaceutical company focused on niche APIs for the generic industry.
Biophore is a research-driven global pharmaceutical company focused on niche APIs for the generic industry.
About the Company : Biophore, founded in 2007, develops and manufactures niche and complex pharmaceutical products. With USFDA- and EU-approved API facilities, a dedicated intermediates site and an R&...
 Granules India Limited has high volume world-class facilities for APIs, PFIs, & FDFs, serving customers in over 80 countries.
Granules India Limited has high volume world-class facilities for APIs, PFIs, & FDFs, serving customers in over 80 countries.
About the Company : Granules is a fast-growing, vertically integrated pharma company based in Hyderabad, committed to operational excellence, quality, and customer service. It manufactures across the ...
 ChemWerth works in generic API development & supply, non-infringement patent strategy development and regulatory support.
ChemWerth works in generic API development & supply, non-infringement patent strategy development and regulatory support.
About the Company : ChemWerth, established in 1982, is a US-headquartered full-service generic API company. It supplies cGMP-quality APIs to regulated markets worldwide, with exclusive partnerships fo...
 Metrochem has been delivering customized volume & quality products to customers across the world, taking utmost care of their needs.
Metrochem has been delivering customized volume & quality products to customers across the world, taking utmost care of their needs.
About the Company : Established in 2004, Metrochem API is one of the fastest-growing APIs, pellets & intermediates manufacturers. It has 6 dedicated manufacturing facilities for its 3 core product gro...
 Nuray is an expert in the synthesis of Niche novel APIs, the first to launch Generics, NCEs, Advanced Intermediates // USFDA certified.
Nuray is an expert in the synthesis of Niche novel APIs, the first to launch Generics, NCEs, Advanced Intermediates // USFDA certified.
About the Company : Nuray Chemicals Pvt Ltd, established in 2012 near Chennai, is an API manufacturer for highly regulated markets. Its state-of-the-art R&D facility specializes in synthesizing NCEs, ...
 Honour is a leading global CDMO and specialty chemicals manufacturer with seven world-class sites delivering quality-driven solutions.
Honour is a leading global CDMO and specialty chemicals manufacturer with seven world-class sites delivering quality-driven solutions.
About the Company : Honour is a leading global CDMO and trusted manufacturer of specialty chemicals and ingredients, with seven world-class facilities meeting global safety and quality standards. Thro...
About the Company : Operational since 2024, ApiSyn Healthcare Pvt. Ltd. is a rapidly growing cGMP manufacturer and CDMO specializing in APIs, intermediates, peptides, and amino acids across two dedica...
 Hiray Pharma Solutions partners with global innovators to develop, manufacture and commercialize high-quality APIs and intermediates.
Hiray Pharma Solutions partners with global innovators to develop, manufacture and commercialize high-quality APIs and intermediates.
About the Company : Hiray Pharma Solutions is a global end-to-end CDMO specializing in APIs and intermediates. The company offers integrated services across Process R&D, Analytical R&D, manufacturing,...
About the Company : Aptra Synthesis Private Limited, is a reliable and vertically integrated manufacturer of APIs and intermediates, boasting state-of-the-art manufacturing facilities that adhere to G...

About the Company : Kimia Biosciences, established in 1993, is focused on manufacturing bulk drugs for various therapeutic segments. With a strong emphasis on excellence and advanced technologies, Kim...

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]Global Sales Information

Market Place

Reply
11 Nov 2025
Reply
23 Apr 2024
Reply
15 Sep 2023
Reply
11 Sep 2018
 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
Patents & EXCLUSIVITIES
ABOUT THIS PAGE
78
PharmaCompass offers a list of Avatrombopag Maleate API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Avatrombopag Maleate manufacturer or Avatrombopag Maleate supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Avatrombopag Maleate manufacturer or Avatrombopag Maleate supplier.
A Avatrombopag manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Avatrombopag, including repackagers and relabelers. The FDA regulates Avatrombopag manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Avatrombopag API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
click here to find a list of Avatrombopag manufacturers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PhamaCompass.
A Avatrombopag supplier is an individual or a company that provides Avatrombopag active pharmaceutical ingredient (API) or Avatrombopag finished formulations upon request. The Avatrombopag suppliers may include Avatrombopag API manufacturers, exporters, distributors and traders.
click here to find a list of Avatrombopag suppliers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PharmaCompass.
A Avatrombopag DMF (Drug Master File) is a document detailing the whole manufacturing process of Avatrombopag active pharmaceutical ingredient (API) in detail. Different forms of Avatrombopag DMFs exist exist since differing nations have different regulations, such as Avatrombopag USDMF, ASMF (EDMF), JDMF, CDMF, etc.
A Avatrombopag DMF submitted to regulatory agencies in the US is known as a USDMF. Avatrombopag USDMF includes data on Avatrombopag's chemical properties, information on the facilities and procedures used, and details about packaging and storage. The Avatrombopag USDMF is kept confidential to protect the manufacturer’s intellectual property.
click here to find a list of Avatrombopag suppliers with USDMF on PharmaCompass.
In Korea, the Ministry of Food and Drug Safety (MFDS) is in charge of regulating pharmaceutical products and services.
Pharmaceutical companies submit a Avatrombopag Drug Master File in Korea (Avatrombopag KDMF) to the MFDS, which includes comprehensive information about the production, processing, facilities, materials, packaging, and testing of Avatrombopag. The MFDS reviews the Avatrombopag KDMF as part of the drug registration process and uses the information provided in the Avatrombopag KDMF to evaluate the safety and efficacy of the drug.
After submitting a Avatrombopag KDMF to the MFDS, the registered manufacturer can provide importers or distributors with the registration number without revealing confidential information to Korean business partners. Applicants seeking to register their Avatrombopag API can apply through the Korea Drug Master File (KDMF).
click here to find a list of Avatrombopag suppliers with KDMF on PharmaCompass.
A Avatrombopag written confirmation (Avatrombopag WC) is an official document issued by a regulatory agency to a Avatrombopag manufacturer, verifying that the manufacturing facility of a Avatrombopag active pharmaceutical ingredient (API) adheres to the Good Manufacturing Practices (GMP) regulations of the importing country. When exporting Avatrombopag APIs or Avatrombopag finished pharmaceutical products to another nation, regulatory agencies frequently require a Avatrombopag WC (written confirmation) as part of the regulatory process.
click here to find a list of Avatrombopag suppliers with Written Confirmation (WC) on PharmaCompass.
National Drug Code is a comprehensive database maintained by the FDA that contains information on all drugs marketed in the US. This directory includes information about finished drug products, unfinished drug products, and compounded drug products, including those containing Avatrombopag as an active pharmaceutical ingredient (API).
The FDA updates the NDC directory daily. The NDC numbers for Avatrombopag API and other APIs are published in this directory by the FDA.
The NDC unfinished drugs database includes product listing information submitted for all unfinished drugs, such as active pharmaceutical ingredients (APIs), drugs intended for further processing and bulk drug substances for compounding.
Pharmaceutical companies that manufacture Avatrombopag as an active pharmaceutical ingredient (API) must furnish the FDA with an updated record of all drugs that they produce, prepare, propagate, compound, or process for commercial distribution in the US at their facilities.
The NDC directory also contains data on finished compounded human drug products that contain Avatrombopag and are produced by outsourcing facilities. While these outsourcing facilities are not mandated to assign a Avatrombopag NDC to their finished compounded human drug products, they may choose to do so.
click here to find a list of Avatrombopag suppliers with NDC on PharmaCompass.
Avatrombopag Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Avatrombopag GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Avatrombopag GMP manufacturer or Avatrombopag GMP API supplier for your needs.
A Avatrombopag CoA (Certificate of Analysis) is a formal document that attests to Avatrombopag's compliance with Avatrombopag specifications and serves as a tool for batch-level quality control.
Avatrombopag CoA mostly includes findings from lab analyses of a specific batch. For each Avatrombopag CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Avatrombopag may be tested according to a variety of international standards, such as European Pharmacopoeia (Avatrombopag EP), Avatrombopag JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Avatrombopag USP).

