
Synopsis
Synopsis
0
CEP/COS
0
JDMF
0
EU WC
0
KDMF
0
NDC API
0
VMF
0
Listed Suppliers
0
EDQM
0
USP
0
JP
0
Others
0
FDF Dossiers
0
FDA Orange Book
0
Europe
0
Canada
0
Australia
0
South Africa
0
Listed Dossiers
DRUG PRODUCT COMPOSITIONS
0
US Patents
0
US Exclusivities
0
Health Canada Patents
US Medicaid
NA
Annual Reports
NA
Regulatory FDF Prices
NA
0
API
0
FDF
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
0
News #PharmaBuzz
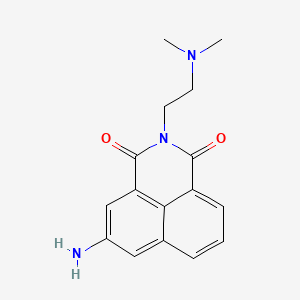

1. 5-amino-2-(2-(dimethylamino)ethyl)-1h-benz(de)isoquinoline-1,3(2h)-dione
2. 5-amino-2-(2-dimethylaminoethyl)benzo(de)isoquinolin-1,3-dione
3. Benzisoquinolinedione
4. Fa 142
5. M-fa 142
6. M-fa-142
7. Mfa 142
8. Mfa-142
9. Nafidimide
10. Nsc 308847
11. Nsc-308847
1. 69408-81-7
2. Nafidimide
3. Quinamed
4. Benzisoquinolinedione
5. Xanafide
6. 5-aminomitonafide
7. Nsc-308847
8. As1413
9. M-fa 142
10. Fa 142
11. Nsc308847
12. As-1413
13. 3-amino-n-(2-(dimethylamino)ethyl)naphthalimide
14. 5-amino-2-[2-(dimethylamino)ethyl]benzo[de]isoquinoline-1,3-dione
15. Mls000757039
16. Chembl428676
17. Amonifide
18. 1q8d39n37l
19. Amonafide (usan)
20. 1h-benz(de)isoquinoline-1,3(2h)-dione, 5-amino-2-(2-(dimethylamino)ethyl)-
21. Naphthalimide, 3-amino-n-(2-(dimethylamino)ethyl)-
22. Amonafide [usan]
23. 5-amino-2-(2-(dimethylamino)ethyl)-1h-benzo[de]isoquinoline-1,3(2h)-dione
24. Amonafide [inn]
25. Amonafidum [latin]
26. Amonafida [spanish]
27. Amonafidum
28. Amonafida
29. Smr000528907
30. Amonafide [usan:inn]
31. As-1413(amonafide)
32. Nsc 308847
33. Brn 5344673
34. Amonofide
35. Unii-1q8d39n37l
36. 5-amino-2-[2-(dimethylamino)ethyl]-1h-benzo[de]isoquinoline-1,3(2h)-dione
37. Amonafide-[d6]
38. M-fa-142
39. Bida
40. 3-amino-n-(2-(dimethylamino)ethyl)-1,8-naphthalindicarboximid
41. Amonafide [mart.]
42. 1h-benz(de)isoquinoline-1,3(2h)-dione, 5-amino-2-(dimethylamino)ethyl-
43. Ncimech_000383
44. Amonafide [who-dd]
45. Schembl27226
46. Mls006010419
47. Mls006011267
48. Chebi:94661
49. Cgx-571
50. Dtxsid30219562
51. Hms3654h19
52. 5-amino-2-(2-dimethylaminoethyl)benzo[de]isoquinoline-1,3-dione
53. Zinc4214836
54. Bdbm50033894
55. Ccg-35694
56. Nsc758251
57. S1367
58. Akos015850731
59. Bcp9000321
60. Cs-0496
61. Db05022
62. Nci-308847
63. Nsc-758251
64. Nsc308847,as1413
65. Ncgc00346513-01
66. Ac-22581
67. As-56124
68. Hy-10982
69. Nci60_002643
70. Ft-0662118
71. Sw219946-1
72. A24926
73. D10090
74. 408a817
75. Q4747531
76. Brd-k56334280-001-02-8
77. Brd-k56334280-001-03-6
78. 1h-benz[de]isoquinoline-1, 5-amino-2-[2-(dimethylamino)ethyl]-
79. 5-amino-2-(2-dimethylamino-ethyl)-benzo[de]isoquinoline-1,3-dione
80. 1h-benz[de]isoquinoline-1,3(2h)-dione,5-amino-2-[2-(dimethylamino)ethyl]-
81. 5-amino-2-(2-dimethylamino-ethyl)-benzo[de]isoquinoline-1,3-dione (amonafide)
82. 5-amino-2-(2-dimethylamino-ethyl)-benzo[de]isoquinoline-1,3-dione(amonafide)
83. 5-amino-2-[2-(dimethylamino)ethyl]-1h-benz[de]isoquinoline-1,3(2h)-dione;amonafide.
84. 5-amino-2-[2-(dimethylamino)ethyl]-1h-benzo[de]isoquinoline-1,3(2h)-dione #
85. 11-amino-3-[2-(dimethylamino)ethyl]-3-azatricyclo[7.3.1.0?,??]trideca-1(13),5,7,9,11-pentaene-2,4-dione
86. Amonafidedihydrochloride,1h-benz[de]isoquinoline-1,3(2h)-dione,5-amino-2-[2-(dimethylamino)ethyl]-
| Molecular Weight | 283.32 g/mol |
|---|---|
| Molecular Formula | C16H17N3O2 |
| XLogP3 | 1.5 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 4 |
| Rotatable Bond Count | 3 |
| Exact Mass | 283.132076794 g/mol |
| Monoisotopic Mass | 283.132076794 g/mol |
| Topological Polar Surface Area | 66.6 Ų |
| Heavy Atom Count | 21 |
| Formal Charge | 0 |
| Complexity | 437 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
Investigated for use/treatment in breast cancer, ovarian cancer, and prostate cancer.
Antiviral Agents
Agents used in the prophylaxis or therapy of VIRUS DISEASES. Some of the ways they may act include preventing viral replication by inhibiting viral DNA polymerase; binding to specific cell-surface receptors and inhibiting viral penetration or uncoating; inhibiting viral protein synthesis; or blocking late stages of virus assembly. (See all compounds classified as Antiviral Agents.)
Antineoplastic Agents
Substances that inhibit or prevent the proliferation of NEOPLASMS. (See all compounds classified as Antineoplastic Agents.)
Enzyme Inhibitors
Compounds or agents that combine with an enzyme in such a manner as to prevent the normal substrate-enzyme combination and the catalytic reaction. (See all compounds classified as Enzyme Inhibitors.)
Intercalating Agents
Agents that are capable of inserting themselves between the successive bases in DNA, thus kinking, uncoiling or otherwise deforming it and therefore preventing its proper functioning. They are used in the study of DNA. (See all compounds classified as Intercalating Agents.)
Amonafide is a DNA intercalating agent and inhibitor of topoisomerase II that has been extensively studied in patients with malignant solid tumors. Amonafide has also been studied in patients with AML.
ABOUT THIS PAGE
70
PharmaCompass offers a list of Amonafide API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Amonafide manufacturer or Amonafide supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Amonafide manufacturer or Amonafide supplier.
A Amonafide manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Amonafide, including repackagers and relabelers. The FDA regulates Amonafide manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Amonafide API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
A Amonafide supplier is an individual or a company that provides Amonafide active pharmaceutical ingredient (API) or Amonafide finished formulations upon request. The Amonafide suppliers may include Amonafide API manufacturers, exporters, distributors and traders.
click here to find a list of Amonafide suppliers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PharmaCompass.
A Amonafide DMF (Drug Master File) is a document detailing the whole manufacturing process of Amonafide active pharmaceutical ingredient (API) in detail. Different forms of Amonafide DMFs exist exist since differing nations have different regulations, such as Amonafide USDMF, ASMF (EDMF), JDMF, CDMF, etc.
A Amonafide DMF submitted to regulatory agencies in the US is known as a USDMF. Amonafide USDMF includes data on Amonafide's chemical properties, information on the facilities and procedures used, and details about packaging and storage. The Amonafide USDMF is kept confidential to protect the manufacturer’s intellectual property.
click here to find a list of Amonafide suppliers with USDMF on PharmaCompass.
Amonafide Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Amonafide GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Amonafide GMP manufacturer or Amonafide GMP API supplier for your needs.
A Amonafide CoA (Certificate of Analysis) is a formal document that attests to Amonafide's compliance with Amonafide specifications and serves as a tool for batch-level quality control.
Amonafide CoA mostly includes findings from lab analyses of a specific batch. For each Amonafide CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Amonafide may be tested according to a variety of international standards, such as European Pharmacopoeia (Amonafide EP), Amonafide JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Amonafide USP).

