
Synopsis
Synopsis
0
CEP/COS
0
VMF
0
Australia
US Medicaid
NA
Annual Reports
NA
Regulatory FDF Prices
NA
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers

1. Aller-chlor
2. Antihistaminico Llorens
3. Chlo-amine
4. Chlor-100
5. Chlor-trimeton
6. Chlor-tripolon
7. Chlorphenamine
8. Chlorpheniramine
9. Chlorpheniramine Tannate
10. Chlorpro
11. Chlorprophenpyridamine
12. Chlorspan 12
13. Chlortab-4
14. Cloro-trimeton
15. Efidac 24
16. Kloromin
17. Maleate, Chlorpheniramine
18. Piriton
19. Tannate, Chlorpheniramine
20. Teldrin
1. 113-92-8
2. Chlorphenamine Maleate
3. Neorestamin
4. Synistamin
5. Piriton
6. Chlor-trimeton
7. Antagonate
8. Kloromin
9. Chlorphenamine Hydrogen Maleate
10. Histaspan
11. Pyridamal
12. Efidac
13. Chlorpheniramine (maleate)
14. Nci-c55265
15. Polaronil
16. Chlorphenamine Maleates
17. V1q0o9oj9z
18. Chlorpheniramine Maleate Salt
19. Chlorprophenpyridamine Maleate
20. Chloropheniramine Maleate
21. Chebi:3645
22. 3-(4-chlorophenyl)-n,n-dimethyl-3-(pyridin-2-yl)propan-1-amine Maleate
23. Nsc-756684
24. Chlortrimeton
25. Allerclor
26. Allergin
27. Chlormene
28. Ibioton
29. Lorphen
30. Alunex
31. Dehist
32. Piriex
33. Chlor-tripolon
34. C-meton
35. (z)-but-2-enedioic Acid;3-(4-chlorophenyl)-n,n-dimethyl-3-pyridin-2-ylpropan-1-amine
36. Histadur Dura-tabs
37. Chlor-trimeton Od
38. Pyridamal 100
39. Pyridamal-100
40. Polaronil [german]
41. Carbinoxamide Maleate
42. Chlor-trimeton Allergy
43. Phenalin
44. Chlor-trimeton Injection
45. Histaspan-plus
46. Histaspan-d
47. Medique Chlorphen
48. Dl-chlorpheniramine Maleate
49. Ru-tuss Capsules
50. Smr000466271
51. Chloroprophenpyridamine Maleate
52. Ccris 1418
53. 3-(4-chlorophenyl)-n,n-dimethyl-3-pyridin-2-ylpropan-1-aminium (2z)-3-carboxyprop-2-enoate
54. Chlor-trimeton 12 Hour
55. Efidac 24 Chlorpheniramine Maleate
56. Sr-01000075261
57. M.p. Chlorcaps T.d.
58. Einecs 204-037-5
59. Mfcd00069225
60. Chlorpheniramine Maleate Oros Tablets
61. Unii-v1q0o9oj9z
62. Nasohist
63. Puermin
64. Corfen
65. Lohist
66. Chlor-trimeton Repetabs, Syrup, And Tablets
67. Sine Off
68. Sildec Pe
69. Alka-seltzer Plus
70. Prestwick_57
71. Chlorphenaminemaleate
72. Chlorpheniramine Maleate [usp:jan]
73. Teldrin (tn)
74. (+/-)-chlorpheniramine Maleate Salt
75. Chlorophenaminemaleate
76. Zodryl Ac 25
77. Zodryl Ac 30
78. Zodryl Ac 40
79. Zodryl Ac 50
80. Zodryl Ac 60
81. Zodryl Ac 80
82. Chlor-trimeton (tn)
83. 1-p-chlorophenyl-1-(2-pyridyl)-3-dimethylaminopropane Maleate
84. Dl-2-(p-chloro-alpha-2-(dimethylamino)ethylbenzyl)pyridine Maleate
85. 1-(n,n-dimethylamino)-3-(p-chlorophenyl-3-alpha-pyridyl)propane Maleate
86. 1-(p-chlorphenyl)-1-(2-pyridyl)-3-dimethylaminopropan Maleat [german]
87. 2-(p-chloro-alpha-(2-(dimethylamino)ethyl)benzyl)pyridine Maleate (1:1)
88. Chembl1659
89. (y)-chlorpheniramine Maleate
90. Chlorphenamini Hydrogenomaleas
91. Schembl33357
92. (?)-chlorpheniramine Maleate
93. Mls000758311
94. Mls001148230
95. Mls001424173
96. (+/-)-chlorpheniramine Maleate
97. Hms501n18
98. Hy-b0286a
99. (a+/-)-chlorpheniramine Maleate
100. Amf0009
101. 1-(p-chlorphenyl)-1-(2-pyridyl)-3-dimethylaminopropan Maleat
102. Hms1568g16
103. Hms2051l14
104. Hms2093n19
105. Hms2095g16
106. Hms2230f21
107. Hms2232b23
108. Hms3260f04
109. Hms3712g16
110. Hms3884o10
111. Pharmakon1600-01505530
112. Tox21_500261
113. Ac-267
114. Chlorpheniramine Maleate [mi]
115. Nsc759156
116. S1816
117. Chlorpheniramine Maleate (jp17/usp)
118. Chlorpheniramine Maleate [jan]
119. Akos015951220
120. Chlorphenamine Maleate [mart.]
121. Pyridine, 2-(p-chloro-alpha-(2-(dimethylamino)ethyl)benzyl)-, Maleate (1:1)
122. Ccg-101036
123. Ccg-220117
124. Ccg-221565
125. Chlorphenamine Maleate [who-dd]
126. Lp00261
127. Nc00286
128. Nsc 756684
129. 2-pyridinepropanamine, .gamma.-(4-chlorophenyl)-n,n-dimethyl-, (z)-2-butenedioate (1:1)
130. 2-pyridinepropanamine, Gamma-(4-chlorophenyl)-n,n-dimethyl-, (z)-2-butenedioate (1:1)
131. Chlorpheniramine Maleate [vandf]
132. Chlorpheniramine Maleate [usp-rs]
133. Ncgc00093720-01
134. Ncgc00093720-02
135. Ncgc00260946-01
136. As-13167
137. Smr000653461
138. Chlorphenamine Maleate [ep Monograph]
139. Eu-0100261
140. Sw196372-4
141. Chlorpheniramine Maleate [orange Book]
142. C 3025
143. C07780
144. Chlorpheniramine Maleate [usp Monograph]
145. Contac Component Chlorpheniramine Maleate
146. D00665
147. D70131
148. Drize Component Chlorpheniramine Maleate
149. Ornade Component Chlorpheniramine Maleate
150. Chlorphenamine Hydrogen Maleate [who-ip]
151. Demazin Component Chlorpheniramine Maleate
152. Isoclor Component Chlorpheniramine Maleate
153. ( Inverted Question Mark)-chlorpheniramine Maleate
154. Chlorpheniramine Maleate Component Of Contac
155. Chlorpheniramine Maleate Component Of Drize
156. Chlorpheniramine Maleate Component Of Ornade
157. Triaminic Component Chlorpheniramine Maleate
158. Chlorphenamini Hydrogenomaleas [who-ip Latin]
159. Chlorpheniramine Maleate 100 Microg/ml In Methanol
160. Chlorpheniramine Maleate Component Of Demazin
161. Chlorpheniramine Maleate Component Of Isoclor
162. Sr-01000075261-1
163. Sr-01000075261-9
164. W-108612
165. Chlorpheniramine Maleate Component Of Triaminic
166. Q27106157
167. F2173-0923
168. Codimal-l.a. 12 Component Chlorpheniramine Maleate
169. Advil Allergy Sinus Component Chlorpheniramine Maleate
170. Chlorpheniramine Maleate Component Of Advil Allergy Sinus
171. Chlorpheniramine Maleate Component Of Codimal-l.a. 12
172. (+/-)-chlorpheniramine Maleate Salt >=99% (perchloric Acid Titration)
173. (+/-)-chlorpheniramine Maleate Salt, >=99% (perchloric Acid Titration)
174. [3-(4-chloro-phenyl)-3-pyridin-2-yl-propyl]-dimethyl-amine Maleate
175. Chlorphenamine Maleate, European Pharmacopoeia (ep) Reference Standard
176. 2-(p-chloro-.alpha.-(2-(dimethylamino)ethyl)benzyl)pyridine Maleate (1:1)
177. 2-pyridinepropanamine, Gamma-(4-chlorophenyl)-n,n-dimethyl-, (2z)-2-butenedioate (1:1)
178. Chlorphenamine Maleate (chlorpheniramine Maleate) 1.0 Mg/ml In Methanol (as Free Base)
179. Chlorpheniramine Maleate, Pharmaceutical Secondary Standard; Certified Reference Material
180. Chlorpheniramine Maleate, United States Pharmacopeia (usp) Reference Standard
181. Chlorpheniramine Maleate Salt Solution, 1.0 Mg/ml In Methanol (as Free Base), Ampule Of 1 Ml, Certified Reference Material
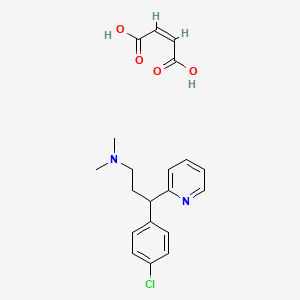
1. Chlorpheniaramine Maleate
2. Chloropheniramine Maleate
3. Chlorphenamine Maleate
| Molecular Weight | 390.9 g/mol |
|---|---|
| Molecular Formula | C20H23ClN2O4 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 6 |
| Rotatable Bond Count | 7 |
| Exact Mass | 390.1346349 g/mol |
| Monoisotopic Mass | 390.1346349 g/mol |
| Topological Polar Surface Area | 90.7 Ų |
| Heavy Atom Count | 27 |
| Formal Charge | 0 |
| Complexity | 368 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 1 |
| Defined Bond Stereocenter Count | 1 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 2 |
| 1 of 2 | |
|---|---|
| Drug Name | Chlorpheniramine maleate |
| Drug Label | Ed Chlortan Tablets are yellow, round, plain bottom, debossed "PH above 012" on top.... |
| Active Ingredient | Chlorpheniramine maleate |
| Dosage Form | Tablet, extended release |
| Route | Oral |
| Strength | 12mg |
| Market Status | Over the Counter |
| Company | Avanthi |
| 2 of 2 | |
|---|---|
| Drug Name | Chlorpheniramine maleate |
| Drug Label | Ed Chlortan Tablets are yellow, round, plain bottom, debossed "PH above 012" on top.... |
| Active Ingredient | Chlorpheniramine maleate |
| Dosage Form | Tablet, extended release |
| Route | Oral |
| Strength | 12mg |
| Market Status | Over the Counter |
| Company | Avanthi |
Histamine H1 Antagonists
Drugs that selectively bind to but do not activate histamine H1 receptors, thereby blocking the actions of endogenous histamine. Included here are the classical antihistaminics that antagonize or prevent the action of histamine mainly in immediate hypersensitivity. They act in the bronchi, capillaries, and some other smooth muscles, and are used to prevent or allay motion sickness, seasonal rhinitis, and allergic dermatitis and to induce somnolence. The effects of blocking central nervous system H1 receptors are not as well understood. (See all compounds classified as Histamine H1 Antagonists.)
Antipruritics
Agents, usually topical, that relieve itching (pruritus). (See all compounds classified as Antipruritics.)
Anti-Allergic Agents
Agents that are used to treat allergic reactions. Most of these drugs act by preventing the release of inflammatory mediators or inhibiting the actions of released mediators on their target cells. (From AMA Drug Evaluations Annual, 1994, p475) (See all compounds classified as Anti-Allergic Agents.)
 LGM Pharma accelerates & optimizes the new product pathway from early development through commercialization.
LGM Pharma accelerates & optimizes the new product pathway from early development through commercialization.
 Malladi is a leader in Ephedrine, Pseudoephedrine Salts & Phenylephrine HCl // USFDA, EDQM, ANSM, KFDA, and TGA inspected.
Malladi is a leader in Ephedrine, Pseudoephedrine Salts & Phenylephrine HCl // USFDA, EDQM, ANSM, KFDA, and TGA inspected.

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 33944
Submission : 2019-08-14
Status : Active
Type : II

Date of Issue : 2026-04-10
Valid Till : 2029-04-09
Written Confirmation Number : WC-0318
Address of the Firm :

NDC Package Code : 57218-765
Start Marketing Date : 2017-04-18
End Marketing Date : 2026-12-31
Dosage Form (Strength) : POWDER (1kg/kg)
Marketing Category : BULK INGREDIENT

Registrant Name : Ace Biopharm Co., Ltd.
Registration Date : 2026-04-14
Registration Number : 20260414-210-J-2179
Manufacturer Name : Malladi Drugs & Pharmaceuticals Limited
Manufacturer Address : Unit-5, Plot No: 49, 50, 55 & 56, IDA, Gajulamandyam, Attur (PO), Rnigunta-517 520, Tirupati District, Andhra Pradesh, India
 Supriya Lifesciences is a cGMP-compliant API manufacturing organization with a leadership position across key & niche products.
Supriya Lifesciences is a cGMP-compliant API manufacturing organization with a leadership position across key & niche products.

Registrant Name : IMCD Korea Co., Ltd.
Registration Date : 2021-04-12
Registration Number : 20201217-210-J-552(1)
Manufacturer Name : Supriya Lifescience Ltd
Manufacturer Address : A-5/2, Lote-Parshuram Industrial Area, MIDC, Taluka-Khed, Dist.-Ratnagiri, 415722, Maharashtra, India

| Available Reg Filing : ASMF, CN |

 Tenatra connects Indian manufacturers with global buyers through active partners in Germany, Switzerland, Belgium, Spain & Turkey.
Tenatra connects Indian manufacturers with global buyers through active partners in Germany, Switzerland, Belgium, Spain & Turkey.
 Supriya Lifesciences is a cGMP-compliant API manufacturing organization with a leadership position across key & niche products.
Supriya Lifesciences is a cGMP-compliant API manufacturing organization with a leadership position across key & niche products.

Registrant Name : Narsha Farm Co., Ltd.
Registration Date : 2024-09-05
Registration Number : 20201217-210-J-552(5)
Manufacturer Name : Supriya Lifescience Ltd.
Manufacturer Address : A-5/2, Lote Parshuram Industrial Area, MIDC Tal-Khed, Dist- Ratnagiri 415722, Maharashtra State, India

| Available Reg Filing : ASMF, CN |

 Supriya Lifesciences is a cGMP-compliant API manufacturing organization with a leadership position across key & niche products.
Supriya Lifesciences is a cGMP-compliant API manufacturing organization with a leadership position across key & niche products.

Registrant Name : Sanil Pharma Co., Ltd.
Registration Date : 2023-06-30
Registration Number : 20201217-210-J-552(4)
Manufacturer Name : Supriya Lifescience Ltd.
Manufacturer Address : A-5/2, Lote Parshuram Industrial Area, MIDC Tal-Khed, Dist- Ratnagiri 415722, Maharashtra State, India

| Available Reg Filing : ASMF, CN |

 Supriya Lifesciences is a cGMP-compliant API manufacturing organization with a leadership position across key & niche products.
Supriya Lifesciences is a cGMP-compliant API manufacturing organization with a leadership position across key & niche products.

Registrant Name : Gukjeon Co., Ltd.
Registration Date : 2022-04-27
Registration Number : 20201217-210-J-552(3)
Manufacturer Name : Supriya Lifescience Ltd.
Manufacturer Address : A-5/2, Lote Parshuram Industrial Area, MIDC, Tal-Khed, Dist-Ratnagiri, 415722, Maharashtra State, India_x000D_

| Available Reg Filing : ASMF, CN |

 Supriya Lifesciences is a cGMP-compliant API manufacturing organization with a leadership position across key & niche products.
Supriya Lifesciences is a cGMP-compliant API manufacturing organization with a leadership position across key & niche products.

Registrant Name : Daeshin Muyak Co., Ltd.
Registration Date : 2021-12-08
Registration Number : 20201217-210-J-552(2)
Manufacturer Name : Supriya Lifescience Ltd.
Manufacturer Address : A-5/2, Lote Parshuram IndustrialArea, MIDC, Tal-Khed, Dist-Ratnagiri, 415722 MaharashtraState, India

| Available Reg Filing : ASMF, CN |

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
DRUG PRODUCT COMPOSITIONS

Related Excipient Companies

Excipients by Applications
Market Place

Patents & EXCLUSIVITIES
REF. STANDARDS & IMPURITIES

ABOUT THIS PAGE
17
PharmaCompass offers a list of Chlorpheniramine Maleate API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Chlorpheniramine Maleate manufacturer or Chlorpheniramine Maleate supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Chlorpheniramine Maleate manufacturer or Chlorpheniramine Maleate supplier.
A Alunex manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Alunex, including repackagers and relabelers. The FDA regulates Alunex manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Alunex API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
click here to find a list of Alunex manufacturers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PhamaCompass.
A Alunex supplier is an individual or a company that provides Alunex active pharmaceutical ingredient (API) or Alunex finished formulations upon request. The Alunex suppliers may include Alunex API manufacturers, exporters, distributors and traders.
click here to find a list of Alunex suppliers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PharmaCompass.
A Alunex DMF (Drug Master File) is a document detailing the whole manufacturing process of Alunex active pharmaceutical ingredient (API) in detail. Different forms of Alunex DMFs exist exist since differing nations have different regulations, such as Alunex USDMF, ASMF (EDMF), JDMF, CDMF, etc.
A Alunex DMF submitted to regulatory agencies in the US is known as a USDMF. Alunex USDMF includes data on Alunex's chemical properties, information on the facilities and procedures used, and details about packaging and storage. The Alunex USDMF is kept confidential to protect the manufacturer’s intellectual property.
click here to find a list of Alunex suppliers with USDMF on PharmaCompass.
The Pharmaceuticals and Medical Devices Agency (PMDA) established the Japan Drug Master File (JDMF), also known as the Master File (MF), to permit Japanese and foreign manufacturers of drug substances, intermediates, excipients, raw materials, and packaging materials (‘Products’) to voluntarily register confidential information about the production and management of their products in Japan.
The Alunex Drug Master File in Japan (Alunex JDMF) empowers Alunex API manufacturers to present comprehensive information (e.g., production methods, data, etc.) to the review authority, i.e., PMDA (Pharmaceuticals & Medical Devices Agency).
PMDA reviews the Alunex JDMF during the approval evaluation for pharmaceutical products. At the time of Alunex JDMF registration, PMDA checks if the format is accurate, if the necessary items have been included (application), and if data has been attached.
click here to find a list of Alunex suppliers with JDMF on PharmaCompass.
In Korea, the Ministry of Food and Drug Safety (MFDS) is in charge of regulating pharmaceutical products and services.
Pharmaceutical companies submit a Alunex Drug Master File in Korea (Alunex KDMF) to the MFDS, which includes comprehensive information about the production, processing, facilities, materials, packaging, and testing of Alunex. The MFDS reviews the Alunex KDMF as part of the drug registration process and uses the information provided in the Alunex KDMF to evaluate the safety and efficacy of the drug.
After submitting a Alunex KDMF to the MFDS, the registered manufacturer can provide importers or distributors with the registration number without revealing confidential information to Korean business partners. Applicants seeking to register their Alunex API can apply through the Korea Drug Master File (KDMF).
click here to find a list of Alunex suppliers with KDMF on PharmaCompass.
A Alunex written confirmation (Alunex WC) is an official document issued by a regulatory agency to a Alunex manufacturer, verifying that the manufacturing facility of a Alunex active pharmaceutical ingredient (API) adheres to the Good Manufacturing Practices (GMP) regulations of the importing country. When exporting Alunex APIs or Alunex finished pharmaceutical products to another nation, regulatory agencies frequently require a Alunex WC (written confirmation) as part of the regulatory process.
click here to find a list of Alunex suppliers with Written Confirmation (WC) on PharmaCompass.
National Drug Code is a comprehensive database maintained by the FDA that contains information on all drugs marketed in the US. This directory includes information about finished drug products, unfinished drug products, and compounded drug products, including those containing Alunex as an active pharmaceutical ingredient (API).
The FDA updates the NDC directory daily. The NDC numbers for Alunex API and other APIs are published in this directory by the FDA.
The NDC unfinished drugs database includes product listing information submitted for all unfinished drugs, such as active pharmaceutical ingredients (APIs), drugs intended for further processing and bulk drug substances for compounding.
Pharmaceutical companies that manufacture Alunex as an active pharmaceutical ingredient (API) must furnish the FDA with an updated record of all drugs that they produce, prepare, propagate, compound, or process for commercial distribution in the US at their facilities.
The NDC directory also contains data on finished compounded human drug products that contain Alunex and are produced by outsourcing facilities. While these outsourcing facilities are not mandated to assign a Alunex NDC to their finished compounded human drug products, they may choose to do so.
click here to find a list of Alunex suppliers with NDC on PharmaCompass.
Alunex Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Alunex GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Alunex GMP manufacturer or Alunex GMP API supplier for your needs.
A Alunex CoA (Certificate of Analysis) is a formal document that attests to Alunex's compliance with Alunex specifications and serves as a tool for batch-level quality control.
Alunex CoA mostly includes findings from lab analyses of a specific batch. For each Alunex CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Alunex may be tested according to a variety of international standards, such as European Pharmacopoeia (Alunex EP), Alunex JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Alunex USP).

