
Synopsis
Synopsis
0
CEP/COS
0
JDMF
0
EU WC
0
KDMF
0
NDC API
0
VMF
0
Listed Suppliers
0
Canada
0
Australia
0
South Africa
0
Listed Dossiers
DRUG PRODUCT COMPOSITIONS
0
US Patents
0
US Exclusivities
0
Health Canada Patents
US Medicaid
NA
Annual Reports
NA
Regulatory FDF Prices
NA
0
API
0
FDF
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
0
News #PharmaBuzz
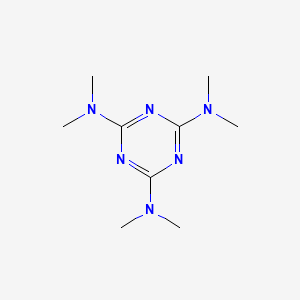

1. Hemel
2. Hexalen
3. Hexamethylmelamine
4. Hexastat
5. Hexinawas
1. 645-05-6
2. Hexamethylmelamine
3. Hexalen
4. Hexastat
5. Hemel
6. 2,4,6-tris(dimethylamino)-1,3,5-triazine
7. Melamine, Hexamethyl-
8. N2,n2,n4,n4,n6,n6-hexamethyl-1,3,5-triazine-2,4,6-triamine
9. Altretaminum
10. Altretamina
11. Altretaminum [inn-latin]
12. Ent 50852
13. Nc 195
14. 2,4,6-tris(dimethylamino)-s-triazine
15. Nci-c50259
16. Hmm
17. Altretamine (hexalen)
18. Nsc 13875
19. Htm
20. Hxm
21. 2-n,2-n,4-n,4-n,6-n,6-n-hexamethyl-1,3,5-triazine-2,4,6-triamine
22. S-triazine, 2,4,6-tris(dimethylamino)-
23. 1,3,5-triazine-2,4,6-triamine, N,n,n',n',n'',n''-hexamethyl-
24. Nsc-13875
25. Mls000069621
26. N,n,n',n',n'',n''-hexamethyl-1,3,5-triazine-2,4,6-triamine
27. Nsc13875
28. Q8bih59o7h
29. Smr000058181
30. 645-05-6 (free Base)
31. Chebi:24564
32. Ncgc00015100-02
33. Dsstox_cid_2579
34. Dsstox_rid_76641
35. Dsstox_gsid_22579
36. Ent-50852;rb-1515;wr-95704
37. Altretamina [inn-spanish]
38. Cas-645-05-6
39. Hexalen (tn)
40. Ccris 5492
41. Sr-01000000117
42. Hexalen [antineoplastic]
43. Altretamine (usp/inn)
44. Einecs 211-428-4
45. Unii-q8bih59o7h
46. Brn 0195058
47. Ai3-50852
48. Hsdb 7559
49. Altretamine, Solid
50. Altretamine [usan:usp:inn:ban]
51. Kb-913
52. Spectrum_001308
53. Altretamine [mi]
54. Opera_id_1095
55. Prestwick0_000946
56. Prestwick1_000946
57. Prestwick2_000946
58. Prestwick3_000946
59. Spectrum2_000907
60. Spectrum3_000951
61. Spectrum4_001064
62. Spectrum5_001092
63. Altretamine [inn]
64. Lopac-a-8723
65. Chemdiv2_005185
66. Altretamine [hsdb]
67. Altretamine [usan]
68. 2,4, 6-tris(dimethylamino)-1,3,5-triazine
69. A 8723
70. Altretamine [vandf]
71. N~2~,n~2~,n~4~,n~4~,n~6~,n~6~-hexamethyl-1,3,5-triazine-2,4,6-triamine
72. Cambridge Id 5148727
73. Cid_2123
74. Schembl4206
75. Altretamine [mart.]
76. Chembl1455
77. Lopac0_000083
78. Altretamine [usp-rs]
79. Altretamine [who-dd]
80. Bspbio_000912
81. Kbiogr_001388
82. Kbioss_001788
83. Zinc905
84. Mls001076123
85. Divk1c_000772
86. Spectrum1503065
87. Spbio_000754
88. Spbio_003071
89. Bpbio1_001004
90. Gtpl7112
91. Dtxsid4022579
92. Bdbm37631
93. Hms502g14
94. Kbio1_000772
95. Kbio2_001788
96. Kbio2_004356
97. Kbio2_006924
98. Kbio3_002042
99. Altretamine [orange Book]
100. Ninds_000772
101. Bcpp000413
102. Hms1383l15
103. Hms1570n14
104. Hms1922c05
105. Hms2090g17
106. Hms2092h16
107. Hms2097n14
108. Hms2234f09
109. Hms3259i20
110. Hms3260a08
111. Hms3372h17
112. Hms3654h11
113. Hms3714n14
114. Pharmakon1600-01503065
115. Altretamine [usp Monograph]
116. 1,3,5-triazine-2,4,6-triamine, N,n,n',n',n',n'-hexamethyl-
117. Bcp27675
118. Ccg-2238
119. Hy-b0181
120. 2,6-tris(dimethylamino)-s-triazine
121. Tox21_110085
122. Tox21_202784
123. Tox21_500083
124. Ent-50852
125. Mfcd00549245
126. Nsc758231
127. S1278
128. Stk749184
129. S-triazine,4,6-tris(dimethylamino)-
130. Akos001729401
131. Tox21_110085_1
132. 1,3,5-tris(dimethylamino)-s-triazine
133. Bcp9000278
134. Cs-2061
135. Db00488
136. Ks-5247
137. Lp00083
138. Nc00595
139. Nsc-758231
140. Rb-1515
141. Sb73671
142. Sdccgsbi-0050071.p004
143. Idi1_000772
144. Idi1_003900
145. Ncgc00015100-01
146. Ncgc00015100-03
147. Ncgc00015100-04
148. Ncgc00015100-05
149. Ncgc00015100-06
150. Ncgc00015100-07
151. Ncgc00015100-08
152. Ncgc00015100-09
153. Ncgc00015100-10
154. Ncgc00015100-11
155. Ncgc00015100-12
156. Ncgc00015100-14
157. Ncgc00015100-15
158. Ncgc00015100-23
159. Ncgc00021216-03
160. Ncgc00021216-04
161. Ncgc00021216-05
162. Ncgc00021216-06
163. Ncgc00021216-07
164. Ncgc00260330-01
165. Ncgc00260768-01
166. Ac-12006
167. Cas-654-05-6
168. Nci60_000871
169. Wr-95704
170. 2,6-tris(dimethylamino)-1,3,5-triazine
171. Sbi-0050071.p003
172. Db-054676
173. Ab00052308
174. Eu-0100083
175. Ft-0651499
176. Sw102053-5
177. C76020
178. D02841
179. Ab00052308-16
180. Ab00052308-17
181. Ab00052308-18
182. Ab00052308_19
183. Ab00052308_20
184. 645a056
185. A834788
186. Q415242
187. Wln: T6n Cn Enj Bn1&1 Dn1&1 Fn1&1
188. 2,4,6-tris(dimethylamino)-1,3,5-triazine, 96%
189. J-523408
190. Sr-01000000117-2
191. Sr-01000000117-4
192. Sr-01000000117-6
193. Brd-k67043667-001-04-1
194. Brd-k67043667-001-15-7
195. Z57931197
196. N2,n2,n4,n4,n6,n6-hexamethyl-1,3,5-trazne-2,4,6-tramne
197. 1,5-triazine-2,4,6-triamine, N,n,n',n',n'',n''-hexamethyl-
198. Altretamine, United States Pharmacopeia (usp) Reference Standard
199. N-[4,6-bis(dimethylamino)-1,3,5-triazin-2-yl]-n,n-dimethylamine
200. 1,3,5-triazine-2,4,6-triamine, N,n,n',n',n'', N''-hexamethyl-
| Molecular Weight | 210.28 g/mol |
|---|---|
| Molecular Formula | C9H18N6 |
| XLogP3 | 2.7 |
| Hydrogen Bond Donor Count | 0 |
| Hydrogen Bond Acceptor Count | 6 |
| Rotatable Bond Count | 3 |
| Exact Mass | 210.15929460 g/mol |
| Monoisotopic Mass | 210.15929460 g/mol |
| Topological Polar Surface Area | 48.4 Ų |
| Heavy Atom Count | 15 |
| Formal Charge | 0 |
| Complexity | 148 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
| 1 of 2 | |
|---|---|
| Drug Name | Hexalen |
| PubMed Health | Altretamine (By mouth) |
| Drug Classes | Antineoplastic Agent |
| Drug Label | HEXALEN (altretamine) capsules, is a synthetic cytotoxic antineoplastic s-triazine derivative. HEXALEN capsules contain 50mg of altretamine for oral administration. Inert ingredients include lactose, anhydrous and calcium stearate. Altretamine,... |
| Active Ingredient | Altretamine |
| Dosage Form | Capsule |
| Route | Oral |
| Strength | 50mg |
| Market Status | Prescription |
| Company | Eisai |
| 2 of 2 | |
|---|---|
| Drug Name | Hexalen |
| PubMed Health | Altretamine (By mouth) |
| Drug Classes | Antineoplastic Agent |
| Drug Label | HEXALEN (altretamine) capsules, is a synthetic cytotoxic antineoplastic s-triazine derivative. HEXALEN capsules contain 50mg of altretamine for oral administration. Inert ingredients include lactose, anhydrous and calcium stearate. Altretamine,... |
| Active Ingredient | Altretamine |
| Dosage Form | Capsule |
| Route | Oral |
| Strength | 50mg |
| Market Status | Prescription |
| Company | Eisai |
Antineoplastic
O'Neil, M.J. (ed.). The Merck Index - An Encyclopedia of Chemicals, Drugs, and Biologicals. Whitehouse Station, NJ: Merck and Co., Inc., 2006., p. 57
Altretamine is indicated for use as a single agent in the palliative treatment of patients with persistent or recurrent epithelial ovarian cancer following first-line therapy with a cisplatin- and/or alkylating agent-based combination. /Included in US product label/
Thomson/Micromedex. Drug Information for the Health Care Professional. Volume 1, Greenwood Village, CO. 2007.
Altretamine, in combination therapy, is considered reasonable medical therapy at some point in the management of small cell lung carcinoma (Evidence rating: IA). /Not included in US product label/
Thomson/Micromedex. Drug Information for the Health Care Professional. Volume 1, Greenwood Village, CO. 2007.
Secondary malignancies are potential delayed effects of many antineoplastic agents, although it is not clear whether the effect is related to their mutagenic or immunosuppressive action. The effect of dose and duration of therapy is also unknown, although risk seems to increase with long-term use. Although information is limited, available data seem to indicate that the carcinogenic risk is greatest with the alkylating agents.
Thomson/Micromedex. Drug Information for the Health Care Professional. Volume 1, Greenwood Village, CO. 2007.
The bone marrow depressant effects of altretamine may result in an increased incidence of microbial infection, delayed healing, and gingival bleeding. Dental work, whenever possible, should be completed prior to initiation of therapy or deferred until blood counts have returned to normal. Patients should be instructed in proper oral hygiene during treatment, including caution in use of regular toothbrushes, dental floss, and toothpicks.
Thomson/Micromedex. Drug Information for the Health Care Professional. Volume 1, Greenwood Village, CO. 2007.
Adverse events ... indicating the need for medical attention /occurring/ at an incidence more frequent: Anemia (unusual tiredness); leukopenia (fever or chills; cough or hoarseness; lower back or side pain; painful or difficult urination); neurotoxicity, including central nervous system (CNS) effects (anxiety; clumsiness; confusion; dizziness; mental depression; weakness; seizures); neurotoxicity, including peripheral neuropathy (numbness in arms or legs); thrombocytopenia (unusual bleeding or bruising; black, tarry stools; blood in urine or stools; pinpoint red spots on skin). Incidence /occurring/ rarely Hepatotoxicity; skin rash or itching.
Thomson/Micromedex. Drug Information for the Health Care Professional. Volume 1, Greenwood Village, CO. 2007.
/Altretamine is/ contraindicated in patients with known sensitivity to altretamine.
Thomson/Micromedex. Drug Information for the Health Care Professional. Volume 1, Greenwood Village, CO. 2007.
For more Drug Warnings (Complete) data for ALTRETAMINE (11 total), please visit the HSDB record page.
For use as a single agent in the palliative treatment of patients with persistent or recurrent ovarian cancer following first-line therapy with a cisplatin and/or alkylating agent-based combination.
Altretamine is a novel antineoplastic agent. The precise mechanism by which altretamine exerts its cytotoxic effect is unknown, although a number of theoretical possibilities have been studied. Structurally, altretamine resembles the alkylating agent triethylenemelamine, yet in vitro tests for alkylating activity of altretamine and its metabolitics have been negative. Altretamine has been demonstrated to be efficacious for certain ovarian tumors resistant to classical alkylating agents. Metabolism of altretamine is a requirement of cytotoxicity. Synthetic monohydroxymethylmelamines, and products of altretamine metabolism, in vitro and in vivo, can form covalent adducts with tissue macromolecules including DNA, but the relevance of these reactions to antitumor activity is unknown.
Antineoplastic Agents, Alkylating
A class of drugs that differs from other alkylating agents used clinically in that they are monofunctional and thus unable to cross-link cellular macromolecules. Among their common properties are a requirement for metabolic activation to intermediates with antitumor efficacy and the presence in their chemical structures of N-methyl groups, that after metabolism, can covalently modify cellular DNA. The precise mechanisms by which each of these drugs acts to kill tumor cells are not completely understood. (From AMA, Drug Evaluations Annual, 1994, p2026) (See all compounds classified as Antineoplastic Agents, Alkylating.)
L - Antineoplastic and immunomodulating agents
L01 - Antineoplastic agents
L01X - Other antineoplastic agents
L01XX - Other antineoplastic agents
L01XX03 - Altretamine
Route of Elimination
Human urinary metabolites were Ndemethylated homologues of altretamine with <1% unmetabolized altretamine excreted at 24 hours.
Rapidly and well-absorbed following oral administration; however, because of rapid hepatic metabolism, peak plasma concentrations are variable.
Thomson/Micromedex. Drug Information for the Health Care Professional. Volume 1, Greenwood Village, CO. 2007.
... The inter-hand intrapatient variability of the bioavailability of altretamine after oral administration represents an important drawback for effective clinical use of this drug. The variability appears to be mostly related to the first-pass effect and therefore may be overcome by intravenous administration of the drug. Attempts to administer the drug intravenously have not been successful in the past...
PMID:7656502 Damia G et al; Clin Pharmacokinet 28 (6): 439-48 (1995)
Because it is highly lipid-soluble, altretamine is distributed to tissues with a high lipid component (e.g., omentum and subcutaneous tissues).
Thomson/Micromedex. Drug Information for the Health Care Professional. Volume 1, Greenwood Village, CO. 2007.
Protein binding: Free fractions: Altretamine: 6%; Pentamethylmelamine: 25%; Tetramethylmelamine: 50%.
Thomson/Micromedex. Drug Information for the Health Care Professional. Volume 1, Greenwood Village, CO. 2007.
For more Absorption, Distribution and Excretion (Complete) data for ALTRETAMINE (10 total), please visit the HSDB record page.
Biotransformation /is/ Hepatic. Metabolism is required for activity. Altretamine undergoes rapid and extensive demethylation, catalyzed by cytochrome P450 enzymes.
Thomson/Micromedex. Drug Information for the Health Care Professional. Volume 1, Greenwood Village, CO. 2007.
The covalent binding of hexamethylmelamine (HMM) and its metabolites was studied in liver, tumor, blood, kidney, spleen, lung, brain, heart, and small intestine after a single ip injection of 2,4,6-14C-hexamethylmelamine (50 mg/kg) to C57Bl/6J female mice bearing 20-day-old M5076/73A ovarian cancer. ... HMM metabolism was also studied. Tissue distribution of pentamethylmelamine (PMM), 2,2,4,6-tetramethylmelamine (TMM), and 2,4,6-trimethylmelamine (TriMM) was determined at the three times considered. At 2 hr the drug was already extensively metabolized, TriMM being the major metabolite among those determined.
PMID:6411375 Garattini E, Colombo T et al; Cancer Chemother Pharmacol 11 (1): 51-5 (1983)
... In all animal species, including humans, altretamine undergoes oxidative N-demethylation with the formation of hydroxymethyl derivatives as intermediates. Hydroxymethylmelamines are believed to be responsible for the cytotoxic and antitumour activity of the drug. ...
PMID:7656502 Damia G et al; Clin Pharmacokinet 28 (6): 439-48 (1995)
In the rat 40% of a dose of 25 mg/kg of hexamethylmelamine or pentamethylmelamine was excreted in the urine as metabolites, more than 95% of which were N2N4-dimethylmelamine and monomethylmelamine. Biliary excretion of hexamethylmelamine or pentamethylmelamine and their N-demethylated metabolites accounted for less than 2% of the administered dose. Only 3% was excreted with the feces, suggesting that there is intestinal reabsorption of a portion of the methylmelamines passing into the bile. Conjugates of methylmelamines with glucuronic acid or sulphate were found only in minute quantities in the urine or bile of rats treated with hexamethylmelamine or pentamethylmelamine. However a conjugation product of pentamethylmelamine, of as yet unknown nature, is a major metabolite after pentamethylmelamine treatment.
PMID:6814076 Colombo T, Broggini M et al; Xenobiotica 12 (5): 315-21 (1982)
4.7-10.2 hours
Elimination /half-life/-Beta-phase: Range, 4.7 to 10.2 hours.
Thomson/Micromedex. Drug Information for the Health Care Professional. Volume 1, Greenwood Village, CO. 2007.
The precise mechanism by which altretamine exerts its cytotoxic effect is unknown although it is classified as an alkylating anti-neoplastic agent. Through this mechanism, the drug is metabolized into alkylating agents by N-demethylation. These alkylating species consequently damage tumor cells.
The exact mechanism of action is unknown. Although altretamine structurally resembles an alkylating agent, it has not been found to have alkylating activity in vitro. There is some evidence that it may inhibit DNA and RNA synthesis.
Thomson/Micromedex. Drug Information for the Health Care Professional. Volume 1, Greenwood Village, CO. 2007.
The hexamethylmelamine analogue trimelamol (tris-hydroxymethyl[trimethyl]melamine) and its equicytotoxic stable analogues CB 7547, CB 7639 and CB 7669 have been used to clarify the mechanism of action for the N-(hydroxymethyl)melamines as antitumour agents. Two main mechanisms have been proposed and explored: (i) formation of a reactive iminium species forming covalent adducts with DNA; and (ii) local formaldehyde release leading to cytotoxic damage. 32P-postlabelling and thermal denaturation experiments showed these compounds to be interactive with cytosine and guanine. Trimelamol gave rise to DNA-interstrand crosslinks in naked plasmid DNA and in cultured cell lines, whereas the analogues failed to do so under a variety of experimental conditions. Along with our observations that cell lines with acquired resistance to the N-(hydroxymethyl)melamines showed no significant cross-resistance to classical bifunctional alkylating agents, DNA crosslinking may play only a minor role in their mechanism of action. In cultured cell lines treatment with formaldehyde, trimelamol and CB 7639 gave rise to high levels of DNA-protein crosslinks with a gradual disappearance over a 24 hr period. Along with ... earlier observations that resistance to trimelamol coincides with cross-resistance to formaldehyde, /investigators/ conclude that formaldehyde-release may be an important factor in their cytotoxicity. Further, the cytotoxicity of trimelamol or formaldehyde towards human ovarian cancer cells was not influenced by glutathione depletion. /N-(hydroxymethyl)melamines/
PMID:7763301 Coley H, Brooks N et al; Biochem Pharmacol 49 (9): 1203-12 (1995)
Related Excipient Companies

Excipients by Applications
REF. STANDARDS & IMPURITIES

ABOUT THIS PAGE
87
PharmaCompass offers a list of Altretamine API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Altretamine manufacturer or Altretamine supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Altretamine manufacturer or Altretamine supplier.
A Altretamine manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Altretamine, including repackagers and relabelers. The FDA regulates Altretamine manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Altretamine API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
A Altretamine supplier is an individual or a company that provides Altretamine active pharmaceutical ingredient (API) or Altretamine finished formulations upon request. The Altretamine suppliers may include Altretamine API manufacturers, exporters, distributors and traders.
click here to find a list of Altretamine suppliers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PharmaCompass.
A Altretamine DMF (Drug Master File) is a document detailing the whole manufacturing process of Altretamine active pharmaceutical ingredient (API) in detail. Different forms of Altretamine DMFs exist exist since differing nations have different regulations, such as Altretamine USDMF, ASMF (EDMF), JDMF, CDMF, etc.
A Altretamine DMF submitted to regulatory agencies in the US is known as a USDMF. Altretamine USDMF includes data on Altretamine's chemical properties, information on the facilities and procedures used, and details about packaging and storage. The Altretamine USDMF is kept confidential to protect the manufacturer’s intellectual property.
click here to find a list of Altretamine suppliers with USDMF on PharmaCompass.
Altretamine Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Altretamine GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Altretamine GMP manufacturer or Altretamine GMP API supplier for your needs.
A Altretamine CoA (Certificate of Analysis) is a formal document that attests to Altretamine's compliance with Altretamine specifications and serves as a tool for batch-level quality control.
Altretamine CoA mostly includes findings from lab analyses of a specific batch. For each Altretamine CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Altretamine may be tested according to a variety of international standards, such as European Pharmacopoeia (Altretamine EP), Altretamine JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Altretamine USP).

