
Synopsis
Synopsis
0
VMF
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
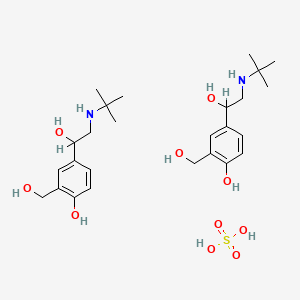

1. 2-t-butylamino-1-(4-hydroxy-3-hydroxy-3-hydroxymethyl)phenylethanol
2. Albuterol
3. Proventil
4. Salbutamol
5. Sultanol
6. Ventolin
1. Salbutamol Sulfate
2. 51022-70-9
3. Salbutamol Hemisulfate
4. Proventil
5. Venetlin
6. Loftan
7. Albuterol Hemisulfate
8. Bronchospray
9. Ventolin
10. Accuneb
11. Proventil-hfa
12. Salbutamol Sulphate
13. Sch 13949w Sulfate
14. Proair
15. Ventolin Rotacaps
16. Proair Respiclick
17. Vospire Er
18. Sultanol
19. Ventilastin
20. Ecovent
21. Salbumol
22. Vospire
23. Albuterol Sulfate(2:1)
24. Albuterol Sulphate
25. 36519-31-0
26. Buventol
27. Salamol
28. Salbutamol (as Sulfate)
29. Sch 13949w Sulphate
30. Sch-13949w Sulphate
31. Sulbutamol (as Sulphate)
32. Sch-13949w Sulfate
33. 4-[2-(tert-butylamino)-1-hydroxyethyl]-2-(hydroxymethyl)phenol;sulfuric Acid
34. R03ac02
35. R03cc02
36. Aerolin
37. Airomir
38. 021sef3731
39. Nsc-289928
40. Aerotec
41. Aloprol
42. Amocasin
43. Broncodil
44. Dipulmin
45. Fartolin
46. Inspiryl
47. Emican
48. Proventil (tn)
49. Epaq
50. Broncho Inhalat
51. Huma-salmol
52. Ventolin Hfa
53. Dl-salbutamol Sulfate
54. (+-)-salbutamol Sulfate
55. 2-(tert-butylamino)-1-(4-hydroxy-3-hydroxymethylphenyl)ethanol Hemisulfate
56. 5-[2-(tert-butylamino)-1-hydroxyethyl]-2-hydroxybenzyl Alcohol Hemisulfate
57. Albuterol Sulfate [usan]
58. Proair Hfa
59. Ah-3365
60. Einecs 256-916-8
61. Salbutamol Hemisulfate Salt
62. Nsc 289928
63. Accuvent
64. Pediavent
65. Torpex
66. Volare
67. Albuterol Sulfate [usan:usp]
68. Ventolin Nebules
69. Ventolin Sulfate
70. Unii-021sef3731
71. Salbutamol Diskus
72. Salbutamoli Sulfas
73. Buventol Easyhaler
74. Proventil Repetabs
75. Pulvinal Salbutamol
76. Sulbutamol Sulphate
77. Proventil Repetabs, Solution, Syrup, And Tablets
78. Ventolin (tn)
79. Accuneb (tn)
80. Salbutamol Easyhaler
81. Ventolin Easy-breathe
82. Albuterol Sulfate,(s)
83. Albuterol Sulfate (usp)
84. Albuterol Sulfate- Bio-x
85. 1-(4-hydroxy-3-hydroxymethylphenyl)-2-(tert-butylamino)ethanol Sulfate
86. Salbutamol Sulfate (jp17)
87. Tbs-7
88. Schembl33279
89. Alpha(sup 1)-((tert-butylamino)methyl)-4-hydroxy-m-xylene-alpha,alpha'-diol Sulfate (2:1) (salt)
90. Albuterol Sulfate [mi]
91. Chebi:2550
92. (+-)-ysulfate(2:1)(salt)
93. Chembl1441059
94. Salbutamol Sulfate [jan]
95. Albuterol Sulfate [vandf]
96. Dtxsid10881180
97. Hms3263k18
98. Hms3266d17
99. Torpex (albuterol Sulfate)
100. Albuterol Sulfate [usp-rs]
101. Salbutamol Sulfate [mart.]
102. Bis((tert-butyl)(beta,3,4-trihydroxyphenethyl)ammonium) Sulphate
103. Salbutamol Sulfate [who-dd]
104. Salbutamol Sulfate [who-ip]
105. Tox21_501098
106. Mfcd00055200
107. Salbutamol Hemisulfate Salt, >=98%
108. Akos015994719
109. Albuterol Sulfate [green Book]
110. Albuterol Sulfate [orange Book]
111. Ccg-222402
112. Ks-5056
113. Lp01098
114. Tq-1016
115. Albuterol Sulfate [usp Impurity]
116. Salbutamol Sulfate [ep Impurity]
117. Salbutamol Sulphate (albuterol Sulphate)
118. Albuterol Sulfate [usp Monograph]
119. Duoneb Component Albuterol Sulfate
120. Ncgc00094370-01
121. Ncgc00261783-01
122. Salbutamol Sulfate [ep Monograph]
123. 1,3-benzenedimethanol, Alpha(sup 1)-(((1,1-dimethylethyl)amino)methyl)-4-hydroxy-, Sulfate (2:1) (salt)
124. 1,3-benzenedimethanol, Alpha1-(((1,1-dimethylethyl)amino)methyl)-4-hydroxy-, Sulfate (2:1) (salt)
125. Ba164388
126. Salbutamoli Sulfas [who-ip Latin]
127. Combivent Component Albuterol Sulfate
128. Albuterol Sulfate Component Of Duoneb
129. Eu-0101098
130. Ft-0650809
131. S0531
132. Salbutamol Sulfate 100 Microg/ml In Methanol
133. A51088
134. Albuterol Sulfate Component Of Combivent
135. D00683
136. S 5013
137. 022s709
138. A828402
139. Q27231480
140. Salbutamol Hemisulfate Salt, Vetranal(tm), Analytical Standard
141. Salbutamol Sulfate, British Pharmacopoeia (bp) Reference Standard
142. Salbutamol Sulfate, European Pharmacopoeia (ep) Reference Standard
143. Albuterol Sulfate, United States Pharmacopeia (usp) Reference Standard
144. Albuterol Sulfate, Pharmaceutical Secondary Standard; Certified Reference Material
145. Salbutamol Sulfate For System Suitability, European Pharmacopoeia (ep) Reference Standard
146. Salbutamol Sulphate (albuterol Sulphate) 1.0 Mg/ml In Methanol (as Free Base)
147. (+/-)-2-tert-butylamino-1-(4-hydroxy-3-hydroxymethylphenyl)ethanol Sulfate Salt (2:1)
148. .alpha.(sup 1)-((tert-butylamino)methyl)-4-hydroxy-m-xylene-.alpha.,.alpha.'-diol Sulfate (2:1) (salt)
149. .alpha.(sup 1)-((tert-butylamino)methyl)-4-hydroxy-m-xylene-.alpha.,.alpha.'-diol Sulphate (2:1) (salt)
150. 1,3-benzenedimethanol,.alpha.(sup 1)-(((1,1 Dimethylethyl)amino)methyl)-4-hydroxy-, Sulfate (2:1) (salt)
151. 1,3-benzenedimethanol,.alpha.(sup 1)-(((1,1 Dimethylethyl)amino)methyl)-4-hydroxy-, Sulphate (2:1) (salt)
152. 1,3-benzenedimethanol,.alpha.1-(((1,1-dimethylethyl)amino)methyl)-4-hydroxy-, Sulfate (2:1) (salt)
| Molecular Weight | 576.7 g/mol |
|---|---|
| Molecular Formula | C26H44N2O10S |
| Hydrogen Bond Donor Count | 10 |
| Hydrogen Bond Acceptor Count | 12 |
| Rotatable Bond Count | 10 |
| Exact Mass | 576.27166678 g/mol |
| Monoisotopic Mass | 576.27166678 g/mol |
| Topological Polar Surface Area | 228 Ų |
| Heavy Atom Count | 39 |
| Formal Charge | 0 |
| Complexity | 309 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 2 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 3 |
| 1 of 12 | |
|---|---|
| Drug Name | Accuneb |
| PubMed Health | Albuterol (By breathing) |
| Drug Classes | Bronchodilator |
| Drug Label | Albuterol Sulfate Inhalation Solution is a relatively selective beta2-adrenergic bronchodilator (see CLINICAL PHARMACOLOGY section below). Albuterol sulfate, the racemic form of albuterol, has the chemical name 1-[(tert-Butylamino)methyl]-4-hydroxy... |
| Active Ingredient | Albuterol sulfate |
| Dosage Form | Solution |
| Route | Inhalation |
| Strength | eq 0.021% base; eq 0.042% base |
| Market Status | Prescription |
| Company | Mylan Speclt |
| 2 of 12 | |
|---|---|
| Drug Name | Albuterol sulfate |
| PubMed Health | Albuterol |
| Drug Classes | Bronchodilator, Cardiovascular Agent |
| Drug Label | The active component of VENTOLIN HFA is albuterol sulfate, USP, the racemic form of albuterol and a relatively selective beta2-adrenergic bronchodilator. Albuterol sulfate has the chemical name 1-[(tert-butylamino)methyl]-4-hydroxy-m-xylene-,... |
| Active Ingredient | Albuterol sulfate |
| Dosage Form | Tablet, extended release; Tablet; Syrup; Solution |
| Route | Inhalation; Oral |
| Strength | eq 4mg base; eq 0.083% base; eq 2mg base; eq 0.021% base; eq 0.5% base; eq 2mg base/5ml; eq 0.042% base; eq 8mg base |
| Market Status | Prescription |
| Company | Watson Labs; Amneal Pharms; Vistapharm; Teva Pharms; Hi Tech Pharma; Ritedose; Actavis Mid Atlantic; Mutual Pharm; Teva; Vintage; Bausch And Lomb; Landela Pharm; Nephron; Mylan Speclt; Mylan |
| 3 of 12 | |
|---|---|
| Drug Name | Proair hfa |
| Active Ingredient | Albuterol sulfate |
| Dosage Form | Aerosol, metered |
| Route | Inhalation |
| Strength | eq 0.09mg base/inh |
| Market Status | Prescription |
| Company | Teva Branded Pharm |
| 4 of 12 | |
|---|---|
| Drug Name | Proventil-hfa |
| Active Ingredient | Albuterol sulfate |
| Dosage Form | Aerosol, metered |
| Route | Inhalation |
| Strength | eq 0.09mg base/inh |
| Market Status | Prescription |
| Company | 3m |
| 5 of 12 | |
|---|---|
| Drug Name | Ventolin hfa |
| Drug Label | Albuterol extended-release tablets contain albuterol sulfate, the racemic form of albuterol and a relatively selective beta2-adrenergic bronchodilator, in an extended-release formulation. Albuterol sulfate has the chemical name () 1-[(tert-butyla... |
| Active Ingredient | Albuterol sulfate |
| Dosage Form | Aerosol, metered |
| Route | Inhalation |
| Strength | eq 0.09mg base/inh |
| Market Status | Prescription |
| Company | Glaxo Grp |
| 6 of 12 | |
|---|---|
| Drug Name | Vospire er |
| Drug Label | The active ingredient of PROAIR HFA (albuterol sulfate) Inhalation Aerosol is albuterol sulfate, a racemic salt, of albuterol. Albuterol sulfate has the chemical name 1-[(tert-butylamino) methyl]-4-hydroxy-m-xylene-,'-diol sulfate (2:1) (salt),... |
| Active Ingredient | Albuterol sulfate |
| Dosage Form | Tablet, extended release |
| Route | Oral |
| Strength | eq 4mg base; eq 8mg base |
| Market Status | Prescription |
| Company | Dava Pharms |
| 7 of 12 | |
|---|---|
| Drug Name | Accuneb |
| PubMed Health | Albuterol (By breathing) |
| Drug Classes | Bronchodilator |
| Drug Label | Albuterol Sulfate Inhalation Solution is a relatively selective beta2-adrenergic bronchodilator (see CLINICAL PHARMACOLOGY section below). Albuterol sulfate, the racemic form of albuterol, has the chemical name 1-[(tert-Butylamino)methyl]-4-hydroxy... |
| Active Ingredient | Albuterol sulfate |
| Dosage Form | Solution |
| Route | Inhalation |
| Strength | eq 0.021% base; eq 0.042% base |
| Market Status | Prescription |
| Company | Mylan Speclt |
| 8 of 12 | |
|---|---|
| Drug Name | Albuterol sulfate |
| PubMed Health | Albuterol |
| Drug Classes | Bronchodilator, Cardiovascular Agent |
| Drug Label | The active component of VENTOLIN HFA is albuterol sulfate, USP, the racemic form of albuterol and a relatively selective beta2-adrenergic bronchodilator. Albuterol sulfate has the chemical name 1-[(tert-butylamino)methyl]-4-hydroxy-m-xylene-,... |
| Active Ingredient | Albuterol sulfate |
| Dosage Form | Tablet, extended release; Tablet; Syrup; Solution |
| Route | Inhalation; Oral |
| Strength | eq 4mg base; eq 0.083% base; eq 2mg base; eq 0.021% base; eq 0.5% base; eq 2mg base/5ml; eq 0.042% base; eq 8mg base |
| Market Status | Prescription |
| Company | Watson Labs; Amneal Pharms; Vistapharm; Teva Pharms; Hi Tech Pharma; Ritedose; Actavis Mid Atlantic; Mutual Pharm; Teva; Vintage; Bausch And Lomb; Landela Pharm; Nephron; Mylan Speclt; Mylan |
| 9 of 12 | |
|---|---|
| Drug Name | Proair hfa |
| Active Ingredient | Albuterol sulfate |
| Dosage Form | Aerosol, metered |
| Route | Inhalation |
| Strength | eq 0.09mg base/inh |
| Market Status | Prescription |
| Company | Teva Branded Pharm |
| 10 of 12 | |
|---|---|
| Drug Name | Proventil-hfa |
| Active Ingredient | Albuterol sulfate |
| Dosage Form | Aerosol, metered |
| Route | Inhalation |
| Strength | eq 0.09mg base/inh |
| Market Status | Prescription |
| Company | 3m |
| 11 of 12 | |
|---|---|
| Drug Name | Ventolin hfa |
| Drug Label | Albuterol extended-release tablets contain albuterol sulfate, the racemic form of albuterol and a relatively selective beta2-adrenergic bronchodilator, in an extended-release formulation. Albuterol sulfate has the chemical name () 1-[(tert-butyla... |
| Active Ingredient | Albuterol sulfate |
| Dosage Form | Aerosol, metered |
| Route | Inhalation |
| Strength | eq 0.09mg base/inh |
| Market Status | Prescription |
| Company | Glaxo Grp |
| 12 of 12 | |
|---|---|
| Drug Name | Vospire er |
| Drug Label | The active ingredient of PROAIR HFA (albuterol sulfate) Inhalation Aerosol is albuterol sulfate, a racemic salt, of albuterol. Albuterol sulfate has the chemical name 1-[(tert-butylamino) methyl]-4-hydroxy-m-xylene-,'-diol sulfate (2:1) (salt),... |
| Active Ingredient | Albuterol sulfate |
| Dosage Form | Tablet, extended release |
| Route | Oral |
| Strength | eq 4mg base; eq 8mg base |
| Market Status | Prescription |
| Company | Dava Pharms |
Adrenergic beta-2 Receptor Agonists
Compounds bind to and activate ADRENERGIC BETA-2 RECEPTORS. (See all compounds classified as Adrenergic beta-2 Receptor Agonists.)
Tocolytic Agents
Drugs that prevent preterm labor and immature birth by suppressing uterine contractions (TOCOLYSIS). Agents used to delay premature uterine activity include magnesium sulfate, beta-mimetics, oxytocin antagonists, calcium channel inhibitors, and adrenergic beta-receptor agonists. The use of intravenous alcohol as a tocolytic is now obsolete. (See all compounds classified as Tocolytic Agents.)
Bronchodilator Agents
Agents that cause an increase in the expansion of a bronchus or bronchial tubes. (See all compounds classified as Bronchodilator Agents.)
 Supriya Lifesciences is a cGMP-compliant API manufacturing organization with a leadership position across key & niche products.
Supriya Lifesciences is a cGMP-compliant API manufacturing organization with a leadership position across key & niche products.
 LGM Pharma accelerates & optimizes the new product pathway from early development through commercialization.
LGM Pharma accelerates & optimizes the new product pathway from early development through commercialization.
 Vamsi Labs is one of the major manufacturers of Anti-asthmatic, Anti-migraine & Anti-psychotic APIs.
Vamsi Labs is one of the major manufacturers of Anti-asthmatic, Anti-migraine & Anti-psychotic APIs.
GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 40579
Submission : 2024-09-30
Status : Inactive
Type : II

Certificate Number : CEP 2024-328 - Rev 00
Issue Date : 2025-11-17
Type : Chemical
Substance Number : 687
Status : Valid

Date of Issue : 2025-11-12
Valid Till : 2028-07-02
Written Confirmation Number : WC-0155
Address of the Firm :

NDC Package Code : 66412-0770
Start Marketing Date : 2025-06-12
End Marketing Date : 2026-12-31
Dosage Form (Strength) : POWDER (1g/g)
Marketing Category : BULK INGREDIENT FOR HUMAN PRESCRIPTION COMPOUNDING

Registrant Name : Huseong Bio Co., Ltd.
Registration Date : 2026-01-23
Registration Number : 20260123-209-J-2116
Manufacturer Name : Vamsi Labs Ltd.
Manufacturer Address : Plot No. A-14, A-15, A-31, A-32 & A-33, MIDC Area, Chincholi, Tal- Mohol, Dist Solapur 413255, Maharashtra State, India

 Aarti Pharmalabs is a partner of choice for pharmaceutical companies for APIs & Intermediates. Largest Indian producer of Caffeine.
Aarti Pharmalabs is a partner of choice for pharmaceutical companies for APIs & Intermediates. Largest Indian producer of Caffeine.
Product Not Available For Sales

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 16104
Submission : 2002-08-16
Status : Active
Type : II

Date of Issue : 2025-06-16
Valid Till : 2028-06-16
Written Confirmation Number : WC-0036
Address of the Firm :

NDC Package Code : 58032-0002
Start Marketing Date : 2017-11-27
End Marketing Date : 2026-12-31
Dosage Form (Strength) : POWDER (1kg/kg)
Marketing Category : BULK INGREDIENT

Registrant Name : Aging Life Science Co., Ltd.
Registration Date : 2021-05-11
Registration Number : 20210511-209-J-985
Manufacturer Name : Neuland Laboratories Limited
Manufacturer Address : Unit 1, Sy. No. 347, 473, 474, 490/2, Bonthapalli Village, Veerabhadraswamy Temple road, Gummadidala Mandal, Sangareddy District 502313, Telangana State, India

 Tenatra connects Indian manufacturers with global buyers through active partners in Germany, Switzerland, Belgium, Spain & Turkey.
Tenatra connects Indian manufacturers with global buyers through active partners in Germany, Switzerland, Belgium, Spain & Turkey.
 TAPI, a leading global supplier of APIs, provides over 350 products and customized CDMO solutions for every stage of development.
TAPI, a leading global supplier of APIs, provides over 350 products and customized CDMO solutions for every stage of development.
 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
 Supriya Lifesciences is a cGMP-compliant API manufacturing organization with a leadership position across key & niche products.
Supriya Lifesciences is a cGMP-compliant API manufacturing organization with a leadership position across key & niche products.
GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 30708
Submission : 2016-06-27
Status : Active
Type : II
 Vamsi Labs is one of the major manufacturers of Anti-asthmatic, Anti-migraine & Anti-psychotic APIs.
Vamsi Labs is one of the major manufacturers of Anti-asthmatic, Anti-migraine & Anti-psychotic APIs.
GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 42593
Submission : 2025-11-22
Status : Active
Type : II
 Vamsi Labs is one of the major manufacturers of Anti-asthmatic, Anti-migraine & Anti-psychotic APIs.
Vamsi Labs is one of the major manufacturers of Anti-asthmatic, Anti-migraine & Anti-psychotic APIs.
GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 40579
Submission : 2024-09-30
Status : Inactive
Type : II
GDUFA
DMF Review : Complete
Rev. Date : 2013-09-26
Pay. Date : 2013-08-29
DMF Number : 16371
Submission : 2003-01-20
Status : Active
Type : II
GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 16104
Submission : 2002-08-16
Status : Active
Type : II
GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 6524
Submission : 1986-08-07
Status : Inactive
Type : II

GDUFA
DMF Review : Complete
Rev. Date : 2015-10-07
Pay. Date : 2015-09-24
DMF Number : 6522
Submission : 1986-08-07
Status : Active
Type : II

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 6885
Submission : 1987-01-20
Status : Active
Type : II

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 5885
Submission : 1985-06-14
Status : Inactive
Type : II

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 6143
Submission : 1985-01-04
Status : Inactive
Type : II

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]DRUG PRODUCT COMPOSITIONS

Related Excipient Companies

Excipients by Applications
Global Sales Information

Company : Astrazeneca
Albuterol Sulfate/Budesonide
Drug Cost (USD) : 925,019
Year : 2023
Prescribers : 1487
Prescriptions : 1753

Portfolio PDF
Product Web Link
Virtual Booth
Digital Content
Website
Corporate PDF
Company : AHP
Albuterol Sulfate
Drug Cost (USD) : 52,018
Year : 2023
Prescribers : 1463
Prescriptions : 2172

Portfolio PDF
Product Web Link
Virtual Booth
Digital Content
Website
Corporate PDF
Company : Cipla USA, Inc.
Albuterol Sulfate
Drug Cost (USD) : 65,279,635
Year : 2023
Prescribers : 1079948
Prescriptions : 2239841

Portfolio PDF
Product Web Link
Virtual Booth
Digital Content
Website
Corporate PDF
Company : Civica, Inc.
Albuterol Sulfate
Drug Cost (USD) : 3,631
Year : 2023
Prescribers : 106
Prescriptions : 189

Portfolio PDF
Product Web Link
Virtual Booth
Digital Content
Website
Corporate PDF
Company : Exelan Pharmace
Albuterol Sulfate
Drug Cost (USD) : 102,508
Year : 2023
Prescribers : 6788
Prescriptions : 10489

Portfolio PDF
Product Web Link
Virtual Booth
Digital Content
Website
Corporate PDF
Company : Lupin Pharmaceu
Albuterol Sulfate
Drug Cost (USD) : 136,827,437
Year : 2023
Prescribers : 1698934
Prescriptions : 4162423

Portfolio PDF
Product Web Link
Virtual Booth
Digital Content
Website
Corporate PDF
Company : Perrigo/Padagis
Albuterol Sulfate
Drug Cost (USD) : 563,049
Year : 2023
Prescribers : 7693
Prescriptions : 18043

Portfolio PDF
Product Web Link
Virtual Booth
Digital Content
Website
Corporate PDF
Company : Prasco Labs
Albuterol Sulfate
Drug Cost (USD) : 196,941,823
Year : 2023
Prescribers : 1667423
Prescriptions : 4067376

Portfolio PDF
Product Web Link
Virtual Booth
Digital Content
Website
Corporate PDF
Company : Sandoz
Albuterol Sulfate
Drug Cost (USD) : 27,414,219
Year : 2023
Prescribers : 576133
Prescriptions : 1184113

Portfolio PDF
Product Web Link
Virtual Booth
Digital Content
Website
Corporate PDF
Company : Teva USA
Albuterol Sulfate
Drug Cost (USD) : 129,646,097
Year : 2023
Prescribers : 1783712
Prescriptions : 4025156

Portfolio PDF
Product Web Link
Virtual Booth
Digital Content
Website
Corporate PDF
 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]RX/OTC/DISCN : DISCN
Dosage Form : TABLET, EXTENDED RELEASE;ORAL
Dosage Strength : EQ 4MG BASE
Price Per Pack :
Published in :
Country : USA
RX/OTC/DISCN : DISCN
RX/OTC/DISCN : DISCN
Dosage Form : TABLET, EXTENDED RELEASE;ORAL
Dosage Strength : EQ 8MG BASE
Price Per Pack :
Published in :
Country : USA
RX/OTC/DISCN : DISCN
Dosage Form :
Dosage Strength : The Scir 200 Ml 0.024%
Price Per Pack (Euro) : 6.9
Published in :
Country : Italy
RX/OTC/DISCN : Class C

Dosage Form :
Dosage Strength : 30 Cpr Eff 2 Mg
Price Per Pack (Euro) : 6.9
Published in :
Country : Italy
RX/OTC/DISCN : Class C

Dosage Form :
Dosage Strength : 60 Erog 200 Mcg Groun Inhal
Price Per Pack (Euro) : 13
Published in :
Country : Italy
RX/OTC/DISCN : Class C

Dosage Form : AEROSOL, METERED;INHALATION
Dosage Strength : EQ 0.09MG BASE/INH
Price Per Pack :
Published in :
Country : USA
RX/OTC/DISCN : RX

GLAXO ALLEN SpA
Dosage Form :
Dosage Strength : 30 Cpr 4 Mg Prolonged Release
Price Per Pack (Euro) : 12.75
Published in :
Country : Italy
RX/OTC/DISCN : Class C

Portfolio PDF
Product Web Link
Virtual Booth
Digital Content
Website
Corporate PDF
GLAXO ALLEN SpA
Dosage Form :
Dosage Strength : 30 Cpr 8 Mg Prolonged Release
Price Per Pack (Euro) : 15.3
Published in :
Country : Italy
RX/OTC/DISCN : Class C

Portfolio PDF
Product Web Link
Virtual Booth
Digital Content
Website
Corporate PDF
VALEAS IND.CHIM.FARMAC. SpA
Dosage Form :
Dosage Strength : The Scir 120 Ml
Price Per Pack (Euro) : 6.4
Published in :
Country : Italy
RX/OTC/DISCN : Class C

Portfolio PDF
Product Web Link
Virtual Booth
Digital Content
Website
Corporate PDF
PROMEDICA Srl
Beclomethasone Dipropionate; Salbutamol Sulphate
Dosage Form : Beclomethasone+Salbutamol+1,60.8...
Dosage Strength : suspe nebul 10 vials 0.8 mg + 1.6 mg 2 ml
Price Per Pack (Euro) : 6.11
Published in :
Country : Italy
RX/OTC/DISCN : Class A

Portfolio PDF
Product Web Link
Virtual Booth
Digital Content
Website
Corporate PDF
 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]Main Therapeutic Indication : Respiratory Disorders
Currency : USD
2019 Revenue in Millions : 274
2018 Revenue in Millions : 397
Growth (%) : -31

Main Therapeutic Indication : Respiratory Disorders
Currency : USD
2020 Revenue in Millions : 1,088
2019 Revenue in Millions : 1,300
Growth (%) : -16

Main Therapeutic Indication : Respiratory Disorders
Currency : USD
2020 Revenue in Millions : 241
2019 Revenue in Millions : 274
Growth (%) : -12

Main Therapeutic Indication : Respiratory Disorders
Currency : USD
2021 Revenue in Millions : 180
2020 Revenue in Millions : 241
Growth (%) : -25

Main Therapeutic Indication : Respiratory Disorders
Currency : USD
2021 Revenue in Millions : 972
2020 Revenue in Millions : 1,075
Growth (%) : -9

Main Therapeutic Indication : Respiratory Diseases
Currency : USD
2022 Revenue in Millions : 951
2021 Revenue in Millions : 972
Growth (%) : -2

Main Therapeutic Indication : Respiratory Diseases
Currency : USD
2023 Revenue in Millions : 808
2022 Revenue in Millions : 951
Growth (%) : -3

Main Therapeutic Indication : Pulmonary/Respiratory Diseases
Currency : USD
2024 Revenue in Millions : 66
2023 Revenue in Millions : 2
Growth (%) : 3,200

Main Therapeutic Indication : Pulmonary/Respiratory Diseases
Currency : USD
2024 Revenue in Millions : 873
2023 Revenue in Millions : 808
Growth (%) : 8

Main Therapeutic Indication : Respiratory Disorders
Currency : USD
2014 Revenue in Millions : 11.40%
2013 Revenue in Millions :
Growth (%) :

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]Market Place

Reply
04 Mar 2026
Reply
13 Sep 2025
Reply
08 Jul 2025
Reply
01 Nov 2024
Reply
25 Sep 2024
Reply
20 Sep 2024
Reply
31 Aug 2024
Reply
17 Jul 2024
Reply
22 May 2024
Reply
10 Aug 2023
 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
Patents & EXCLUSIVITIES
REF. STANDARDS & IMPURITIES

ABOUT THIS PAGE
34
PharmaCompass offers a list of Salbutamol Sulphate API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Salbutamol Sulphate manufacturer or Salbutamol Sulphate supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Salbutamol Sulphate manufacturer or Salbutamol Sulphate supplier.
A Aerolin manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Aerolin, including repackagers and relabelers. The FDA regulates Aerolin manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Aerolin API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
click here to find a list of Aerolin manufacturers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PhamaCompass.
A Aerolin supplier is an individual or a company that provides Aerolin active pharmaceutical ingredient (API) or Aerolin finished formulations upon request. The Aerolin suppliers may include Aerolin API manufacturers, exporters, distributors and traders.
click here to find a list of Aerolin suppliers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PharmaCompass.
A Aerolin DMF (Drug Master File) is a document detailing the whole manufacturing process of Aerolin active pharmaceutical ingredient (API) in detail. Different forms of Aerolin DMFs exist exist since differing nations have different regulations, such as Aerolin USDMF, ASMF (EDMF), JDMF, CDMF, etc.
A Aerolin DMF submitted to regulatory agencies in the US is known as a USDMF. Aerolin USDMF includes data on Aerolin's chemical properties, information on the facilities and procedures used, and details about packaging and storage. The Aerolin USDMF is kept confidential to protect the manufacturer’s intellectual property.
click here to find a list of Aerolin suppliers with USDMF on PharmaCompass.
The Pharmaceuticals and Medical Devices Agency (PMDA) established the Japan Drug Master File (JDMF), also known as the Master File (MF), to permit Japanese and foreign manufacturers of drug substances, intermediates, excipients, raw materials, and packaging materials (‘Products’) to voluntarily register confidential information about the production and management of their products in Japan.
The Aerolin Drug Master File in Japan (Aerolin JDMF) empowers Aerolin API manufacturers to present comprehensive information (e.g., production methods, data, etc.) to the review authority, i.e., PMDA (Pharmaceuticals & Medical Devices Agency).
PMDA reviews the Aerolin JDMF during the approval evaluation for pharmaceutical products. At the time of Aerolin JDMF registration, PMDA checks if the format is accurate, if the necessary items have been included (application), and if data has been attached.
click here to find a list of Aerolin suppliers with JDMF on PharmaCompass.
In Korea, the Ministry of Food and Drug Safety (MFDS) is in charge of regulating pharmaceutical products and services.
Pharmaceutical companies submit a Aerolin Drug Master File in Korea (Aerolin KDMF) to the MFDS, which includes comprehensive information about the production, processing, facilities, materials, packaging, and testing of Aerolin. The MFDS reviews the Aerolin KDMF as part of the drug registration process and uses the information provided in the Aerolin KDMF to evaluate the safety and efficacy of the drug.
After submitting a Aerolin KDMF to the MFDS, the registered manufacturer can provide importers or distributors with the registration number without revealing confidential information to Korean business partners. Applicants seeking to register their Aerolin API can apply through the Korea Drug Master File (KDMF).
click here to find a list of Aerolin suppliers with KDMF on PharmaCompass.
A Aerolin CEP of the European Pharmacopoeia monograph is often referred to as a Aerolin Certificate of Suitability (COS). The purpose of a Aerolin CEP is to show that the European Pharmacopoeia monograph adequately controls the purity of Aerolin EP produced by a given manufacturer. Suppliers of raw materials can prove the suitability of Aerolin to their clients by showing that a Aerolin CEP has been issued for it. The manufacturer submits a Aerolin CEP (COS) as part of the market authorization procedure, and it takes on the role of a Aerolin CEP holder for the record. Additionally, the data presented in the Aerolin CEP (COS) is managed confidentially and offers a centralized system acknowledged by numerous nations, exactly like the Aerolin DMF.
A Aerolin CEP (COS) is recognised by all 36 nations that make up the European Pharmacopoeia Convention. Aerolin CEPs may be accepted in nations that are not members of the Ph. Eur. at the discretion of the authorities there.
click here to find a list of Aerolin suppliers with CEP (COS) on PharmaCompass.
A Aerolin written confirmation (Aerolin WC) is an official document issued by a regulatory agency to a Aerolin manufacturer, verifying that the manufacturing facility of a Aerolin active pharmaceutical ingredient (API) adheres to the Good Manufacturing Practices (GMP) regulations of the importing country. When exporting Aerolin APIs or Aerolin finished pharmaceutical products to another nation, regulatory agencies frequently require a Aerolin WC (written confirmation) as part of the regulatory process.
click here to find a list of Aerolin suppliers with Written Confirmation (WC) on PharmaCompass.
National Drug Code is a comprehensive database maintained by the FDA that contains information on all drugs marketed in the US. This directory includes information about finished drug products, unfinished drug products, and compounded drug products, including those containing Aerolin as an active pharmaceutical ingredient (API).
The FDA updates the NDC directory daily. The NDC numbers for Aerolin API and other APIs are published in this directory by the FDA.
The NDC unfinished drugs database includes product listing information submitted for all unfinished drugs, such as active pharmaceutical ingredients (APIs), drugs intended for further processing and bulk drug substances for compounding.
Pharmaceutical companies that manufacture Aerolin as an active pharmaceutical ingredient (API) must furnish the FDA with an updated record of all drugs that they produce, prepare, propagate, compound, or process for commercial distribution in the US at their facilities.
The NDC directory also contains data on finished compounded human drug products that contain Aerolin and are produced by outsourcing facilities. While these outsourcing facilities are not mandated to assign a Aerolin NDC to their finished compounded human drug products, they may choose to do so.
click here to find a list of Aerolin suppliers with NDC on PharmaCompass.
Aerolin Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Aerolin GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Aerolin GMP manufacturer or Aerolin GMP API supplier for your needs.
A Aerolin CoA (Certificate of Analysis) is a formal document that attests to Aerolin's compliance with Aerolin specifications and serves as a tool for batch-level quality control.
Aerolin CoA mostly includes findings from lab analyses of a specific batch. For each Aerolin CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Aerolin may be tested according to a variety of international standards, such as European Pharmacopoeia (Aerolin EP), Aerolin JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Aerolin USP).

