
Synopsis
Synopsis
0
CEP/COS
0
VMF
0
Australia
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
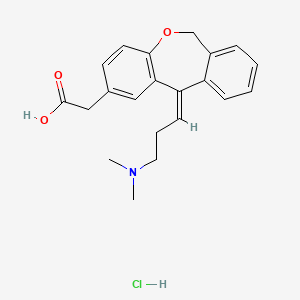

1. 11-(3-(dimethylamino)propylidene)-6,11-dihydrodibenz(b,e)oxepin-2-acetic Acid
2. 4679, Kw
3. Hydrochloride, Olopatadine
4. Kw 4679
5. Kw 4943a
6. Kw-4679
7. Kw-4943a
8. Kw4943a
9. Olopatadine
10. Patanol
1. 140462-76-6
2. Olopatadine Hcl
3. Allelock
4. Pataday
5. Patanase
6. Alo4943a
7. Pazeo
8. Kw4679
9. Kw-4679
10. Olopatadine (hydrochloride)
11. Alo-4943a
12. (z)-2-(11-(3-(dimethylamino)propylidene)-6,11-dihydrodibenzo[b,e]oxepin-2-yl)acetic Acid Hydrochloride
13. Olopatadine (as Hydrochloride)
14. 2xg66w44kf
15. Dsstox_cid_26486
16. Dsstox_rid_81657
17. Dsstox_gsid_46486
18. Dibenz(b,e)oxepin-2-acetic Acid, 11-(3-(dimethylamino)propylidene)-6,11-dihydro-, Hydrochloride, (z)-
19. Olopine
20. Patadine
21. Olopax
22. Chembl1719
23. Kw 4679
24. Cas-140462-76-6
25. Opatanol (tn)
26. Unii-2xg66w44kf
27. Sr-01000763388
28. Alo 4943a
29. Olopatadine Hydrochloride [usan]
30. Allelock (tn)
31. Patanol (tn)
32. De 114
33. Mfcd00875716
34. Olopatadine Hydrochloride [usan:usp]
35. Olopatadine Hydrochlorde
36. O-ppds
37. Ncgc00164623-01
38. Schembl23213
39. Mls001401465
40. Dtxsid0046486
41. Chebi:31933
42. Hy-b0426a
43. Olopatadine Hydrochloride- Bio-x
44. Al4943a
45. Alo4943a;kw4679
46. 2-[(11z)-11-[3-(dimethylamino)propylidene]-6h-benzo[c][1]benzoxepin-2-yl]acetic Acid;hydrochloride
47. Dibenz[b,e]oxepin-2-acetic Acid, 11-[3-(dimethylamino)propylidene]-6,11-dihydro-, Hydrochloride (1:1), (11z)-
48. Ex-a1347
49. Olopatadine Hydrochloride (opatanol)
50. Tox21_112242
51. S2494
52. Olopatadine Hydrochloride [mi]
53. Akos015895232
54. Akos025149118
55. Olopatadine Hydrochloride (jp17/usp)
56. Tox21_112242_1
57. Bcp9001022
58. Ccg-101141
59. H06o056
60. Ks-1228
61. Nc00391
62. Olopatadine Hydrochloride [jan]
63. Ncgc00263532-01
64. Olopatadine Hydrochloride [mart.]
65. Olopatadine Hydrochloride [vandf]
66. 11-((z)-3-(dimethylamino)propylidene)-6,11-dihydrodibenz(b,e)oxepin-2-acetic Acid Hydrochloride
67. 11-((z)-3-(dimethylamino)propylidene)-6,11-dihydrodibenz(b,e)oxepin-2-acetic Acid, Hydrochloride
68. Bo164171
69. Dibenz(b,e)oxepin-2-acetic Acid, 11-(3-dimethylamino)propylidene)-6,11-dihydro-, Hydrochloride, (z)-
70. Olopatadine Hydrochloride [usp-rs]
71. Olopatadine Hydrochloride [who-dd]
72. Smr000469220
73. Olopatadine Hydrochloride, >=98% (hplc)
74. Olopatadine Hydrochloride [ema Epar]
75. O0361
76. C72607
77. D01192
78. Olopatadine Hydrochloride [orange Book]
79. Olopatadine Hydrochloride [usp Monograph]
80. A807652
81. Ryaltris Component Olopatadine Hydrochloride
82. Q-201511
83. Sr-01000763388-4
84. Olopatadine Hydrochloride Component Of Ryaltris
85. Q27255766
86. Olopatadine Hydrochloride, United States Pharmacopeia (usp) Reference Standard
87. (z)-11-[3-(dimethylamino)propylidene]-6,11-dihydro-dibenz[b,e]oxepin-2-acetic Acid Hydrochloride
88. (z)-2-(11-(3-(dimethylamino)propylidene)-6,11-dihydrodibenzo[b,e]oxepin-2-yl)aceticacidhydrochloride
| Molecular Weight | 373.9 g/mol |
|---|---|
| Molecular Formula | C21H24ClNO3 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 4 |
| Rotatable Bond Count | 5 |
| Exact Mass | 373.1444713 g/mol |
| Monoisotopic Mass | 373.1444713 g/mol |
| Topological Polar Surface Area | 49.8 Ų |
| Heavy Atom Count | 26 |
| Formal Charge | 0 |
| Complexity | 488 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 1 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 2 |
| 1 of 8 | |
|---|---|
| Drug Name | Olopatadine hydrochloride |
| Drug Label | Olopatadine Hydrochloride Nasal Solution (Nasal Spray), 665 micrograms (mcg) is a metered-spray solution for intranasal administration. Olopatadine hydrochloride, the active component of Olopatadine Hydrochloride Nasal Solution (Nasal Spray), is a wh... |
| Active Ingredient | Olopatadine hydrochloride |
| Dosage Form | Solution/drops; Spray, metered; Solution |
| Route | Ophthalmic; Nasal; ophthalmic |
| Strength | 0.665mg/spray; 0.2% base; 0.1%; eq 0.1% base; 0.2% |
| Market Status | Tentative Approval; Prescription |
| Company | Wockhardt; Apotex; Sandoz; Watson Labs; Barr |
| 2 of 8 | |
|---|---|
| Drug Name | Pataday |
| Drug Label | PATADAY (olopatadine hydrochloride ophthalmic solution) 0.2% is a sterile ophthalmic solution containing olopatadine for topical administration to the eyes.Olopatadine hydrochloride is a white, crystalline, water-soluble powder with a molecular we... |
| Active Ingredient | Olopatadine hydrochloride |
| Dosage Form | Solution/drops |
| Route | Ophthalmic |
| Strength | eq 0.2% base |
| Market Status | Prescription |
| Company | Alcon Pharms |
| 3 of 8 | |
|---|---|
| Drug Name | Patanase |
| PubMed Health | Olopatadine (Into the nose) |
| Drug Classes | Nasal Agent |
| Drug Label | PATANASE (olopatadine hydrochloride) Nasal Spray, 665 micrograms (mcg) is a metered-spray solution for intranasal administration. Olopatadine hydrochloride, the active component of PATANASE Nasal Spray, is a white, water-soluble crystalline powder. T... |
| Active Ingredient | Olopatadine hydrochloride |
| Dosage Form | Spray, metered |
| Route | Nasal |
| Strength | 0.665mg/spray |
| Market Status | Prescription |
| Company | Alcon Pharms |
| 4 of 8 | |
|---|---|
| Drug Name | Patanol |
| PubMed Health | Olopatadine |
| Drug Classes | Nasal Agent, Ophthalmologic Agent |
| Drug Label | PATANOL (olopatadine hydrochloride ophthalmic solution) 0.1% is a sterile ophthalmic solution containing olopatadine, a relatively selective H1-receptor antagonist and inhibitor of histamine release from the mast cell for topical administration to... |
| Active Ingredient | Olopatadine hydrochloride |
| Dosage Form | Solution/drops |
| Route | Ophthalmic |
| Strength | eq 0.1% base |
| Market Status | Prescription |
| Company | Alcon |
| 5 of 8 | |
|---|---|
| Drug Name | Olopatadine hydrochloride |
| Drug Label | Olopatadine Hydrochloride Nasal Solution (Nasal Spray), 665 micrograms (mcg) is a metered-spray solution for intranasal administration. Olopatadine hydrochloride, the active component of Olopatadine Hydrochloride Nasal Solution (Nasal Spray), is a wh... |
| Active Ingredient | Olopatadine hydrochloride |
| Dosage Form | Solution/drops; Spray, metered; Solution |
| Route | Ophthalmic; Nasal; ophthalmic |
| Strength | 0.665mg/spray; 0.2% base; 0.1%; eq 0.1% base; 0.2% |
| Market Status | Tentative Approval; Prescription |
| Company | Wockhardt; Apotex; Sandoz; Watson Labs; Barr |
| 6 of 8 | |
|---|---|
| Drug Name | Pataday |
| Drug Label | PATADAY (olopatadine hydrochloride ophthalmic solution) 0.2% is a sterile ophthalmic solution containing olopatadine for topical administration to the eyes.Olopatadine hydrochloride is a white, crystalline, water-soluble powder with a molecular we... |
| Active Ingredient | Olopatadine hydrochloride |
| Dosage Form | Solution/drops |
| Route | Ophthalmic |
| Strength | eq 0.2% base |
| Market Status | Prescription |
| Company | Alcon Pharms |
| 7 of 8 | |
|---|---|
| Drug Name | Patanase |
| PubMed Health | Olopatadine (Into the nose) |
| Drug Classes | Nasal Agent |
| Drug Label | PATANASE (olopatadine hydrochloride) Nasal Spray, 665 micrograms (mcg) is a metered-spray solution for intranasal administration. Olopatadine hydrochloride, the active component of PATANASE Nasal Spray, is a white, water-soluble crystalline powder. T... |
| Active Ingredient | Olopatadine hydrochloride |
| Dosage Form | Spray, metered |
| Route | Nasal |
| Strength | 0.665mg/spray |
| Market Status | Prescription |
| Company | Alcon Pharms |
| 8 of 8 | |
|---|---|
| Drug Name | Patanol |
| PubMed Health | Olopatadine |
| Drug Classes | Nasal Agent, Ophthalmologic Agent |
| Drug Label | PATANOL (olopatadine hydrochloride ophthalmic solution) 0.1% is a sterile ophthalmic solution containing olopatadine, a relatively selective H1-receptor antagonist and inhibitor of histamine release from the mast cell for topical administration to... |
| Active Ingredient | Olopatadine hydrochloride |
| Dosage Form | Solution/drops |
| Route | Ophthalmic |
| Strength | eq 0.1% base |
| Market Status | Prescription |
| Company | Alcon |
Treatment of ocular signs and symptoms of seasonal allergic conjunctivitis.
Anti-Inflammatory Agents, Non-Steroidal
Anti-inflammatory agents that are non-steroidal in nature. In addition to anti-inflammatory actions, they have analgesic, antipyretic, and platelet-inhibitory actions. They act by blocking the synthesis of prostaglandins by inhibiting cyclooxygenase, which converts arachidonic acid to cyclic endoperoxides, precursors of prostaglandins. Inhibition of prostaglandin synthesis accounts for their analgesic, antipyretic, and platelet-inhibitory actions; other mechanisms may contribute to their anti-inflammatory effects. (See all compounds classified as Anti-Inflammatory Agents, Non-Steroidal.)
Histamine H1 Antagonists, Non-Sedating
A class of non-sedating drugs that bind to but do not activate histamine receptors (DRUG INVERSE AGONISM), thereby blocking the actions of histamine or histamine agonists. These antihistamines represent a heterogenous group of compounds with differing chemical structures, adverse effects, distribution, and metabolism. Compared to the early (first generation) antihistamines, these non-sedating antihistamines have greater receptor specificity, lower penetration of BLOOD-BRAIN BARRIER, and are less likely to cause drowsiness or psychomotor impairment. (See all compounds classified as Histamine H1 Antagonists, Non-Sedating.)
Anti-Allergic Agents
Agents that are used to treat allergic reactions. Most of these drugs act by preventing the release of inflammatory mediators or inhibiting the actions of released mediators on their target cells. (From AMA Drug Evaluations Annual, 1994, p475) (See all compounds classified as Anti-Allergic Agents.)
S01GX09
 Seqens is an integrated global leader in pharmaceutical solutions & specialty ingredients, & custom-made solutions to our customers.
Seqens is an integrated global leader in pharmaceutical solutions & specialty ingredients, & custom-made solutions to our customers.
 Click Us!
Click Us! 
GDUFA
DMF Review : Reviewed
Rev. Date : 2013-02-07
Pay. Date : 2013-01-02
DMF Number : 23908
Submission : 2010-06-18
Status : Active
Type : II

NDC Package Code : 51014-7733
Start Marketing Date : 2010-04-26
End Marketing Date : 2026-12-31
Dosage Form (Strength) : POWDER (1kg/kg)
Marketing Category : BULK INGREDIENT

| Available Reg Filing : ASMF, BR, CA |

 Bhavna Laboratories is an API & Intermediate manufacturer focusing on the muscle relaxant & ophthalmic segments.
Bhavna Laboratories is an API & Intermediate manufacturer focusing on the muscle relaxant & ophthalmic segments.
 LGM Pharma accelerates & optimizes the new product pathway from early development through commercialization.
LGM Pharma accelerates & optimizes the new product pathway from early development through commercialization.
 Tenatra connects Indian manufacturers with global buyers through active partners in Germany, Switzerland, Belgium, Spain & Turkey.
Tenatra connects Indian manufacturers with global buyers through active partners in Germany, Switzerland, Belgium, Spain & Turkey.
 Curia - Accelerating therapies from discovery to commercialization with flexible, scalable CDMO solutions.
Curia - Accelerating therapies from discovery to commercialization with flexible, scalable CDMO solutions.

GDUFA
DMF Review : Reviewed
Rev. Date : 2016-03-02
Pay. Date : 2016-02-12
DMF Number : 19526
Submission : 2006-06-14
Status : Active
Type : II

Registration Number : 305MF10136
Registrant's Address : Parque Tecnologico-Parcela 105 Boecillo (Valladolid) Spain
Initial Date of Registration : 2023-12-19
Latest Date of Registration :

NDC Package Code : 65089-0041
Start Marketing Date : 1996-12-18
End Marketing Date : 2026-12-31
Dosage Form (Strength) : POWDER (1kg/kg)
Marketing Category : BULK INGREDIENT

Registrant Name : Goodwills Co., Ltd.
Registration Date : 2021-04-28
Registration Number : 20210428-209-J-960
Manufacturer Name : Curia Spain SAU
Manufacturer Address : Parque Tecnológico Boecillo, Parcelas 2y 3, Boecillo, 47151 Valladolid, Spain
 Curia - Accelerating therapies from discovery to commercialization with flexible, scalable CDMO solutions.
Curia - Accelerating therapies from discovery to commercialization with flexible, scalable CDMO solutions.

GDUFA
DMF Review : Reviewed
Rev. Date : 2013-06-25
Pay. Date : 2012-12-24
DMF Number : 25044
Submission : 2011-06-13
Status : Active
Type : II
 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
 Seqens is an integrated global leader in pharmaceutical solutions & specialty ingredients, & custom-made solutions to our customers.
Seqens is an integrated global leader in pharmaceutical solutions & specialty ingredients, & custom-made solutions to our customers.
GDUFA
DMF Review : Complete
Rev. Date : 2013-02-07
Pay. Date : 2013-01-02
DMF Number : 23908
Submission : 2010-06-18
Status : Active
Type : II
 Curia - Accelerating therapies from discovery to commercialization with flexible, scalable CDMO solutions.
Curia - Accelerating therapies from discovery to commercialization with flexible, scalable CDMO solutions.
GDUFA
DMF Review : Complete
Rev. Date : 2013-06-25
Pay. Date : 2012-12-24
DMF Number : 25044
Submission : 2011-06-13
Status : Active
Type : II
 Curia - Accelerating therapies from discovery to commercialization with flexible, scalable CDMO solutions.
Curia - Accelerating therapies from discovery to commercialization with flexible, scalable CDMO solutions.
GDUFA
DMF Review : Complete
Rev. Date : 2016-03-02
Pay. Date : 2016-02-12
DMF Number : 19526
Submission : 2006-06-14
Status : Active
Type : II
GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 23240
Submission : 2009-10-30
Status : Inactive
Type : II

GDUFA
DMF Review : Complete
Rev. Date : 2014-06-23
Pay. Date : 2014-06-17
DMF Number : 24106
Submission : 2010-08-27
Status : Active
Type : II

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 11801
Submission : 1996-01-10
Status : Inactive
Type : II

GDUFA
DMF Review : Complete
Rev. Date : 2015-09-23
Pay. Date : 2015-09-14
DMF Number : 22532
Submission : 2009-02-12
Status : Active
Type : II

GDUFA
DMF Review : Complete
Rev. Date : 2014-05-28
Pay. Date : 2013-12-17
DMF Number : 24624
Submission : 2011-02-08
Status : Active
Type : II

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 24408
Submission : 2010-12-17
Status : Inactive
Type : II

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 19417
Submission : 2006-05-09
Status : Inactive
Type : II

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results] Curia - Accelerating therapies from discovery to commercialization with flexible, scalable CDMO solutions.
Curia - Accelerating therapies from discovery to commercialization with flexible, scalable CDMO solutions.
Registration Number : 305MF10136
Registrant's Address : Parque Tecnologico-Parcela 105 Boecillo (Valladolid) Spain
Initial Date of Registration : 2023-12-19
Latest Date of Registration : 2023-12-19
Registration Number : 304MF10045
Registrant's Address : 4th Floor, OIA House, 470, Cardinal Gracious Road, Andheri East Mumbai 400099, Mahara...
Initial Date of Registration : 2022-02-24
Latest Date of Registration : 2022-02-24

Olopatadine Hydrochloride (Olopatadine HCl)
Registration Number : 303MF10171
Registrant's Address : No. 69 Xingguang Avenue, Renhe Town, Yubei District, Chongqing, China
Initial Date of Registration : 2021-12-02
Latest Date of Registration : 2021-12-02

Registration Number : 223MF10009
Registrant's Address : Viale Milano 26 36075 Montecchio Maggiore Vicenza, Italy
Initial Date of Registration : 2011-01-21
Latest Date of Registration : 2023-07-12

Registration Number : 222MF10253
Registrant's Address : 110, MAGOKDONG-RO, GANGSEO-GU, SEOUL, KOREA
Initial Date of Registration : 2010-10-28
Latest Date of Registration : 2010-10-28

Japanese Pharmacopoeia Olopatadine Hydrochloride (For Manufacturing Use Only)
Registration Number : 226MF10140
Registrant's Address : 530 Chokeiji, Takaoka City, Toyama Prefecture
Initial Date of Registration : 2014-07-31
Latest Date of Registration : 2016-06-06

Registration Number : 224MF10094
Registrant's Address : Plot No: C-24, Sanath Nagar Industrial Estate, Sanath Nagar, Hyderabad, Telangana, In...
Initial Date of Registration : 2012-04-27
Latest Date of Registration : 2021-06-01

Registration Number : 225MF10202
Registrant's Address : 2-27-1 Shinkawa, Chuo-ku, Tokyo
Initial Date of Registration : 2013-11-11
Latest Date of Registration : 2013-11-11

Registration Number : 222MF10280
Registrant's Address : 2-7-1 Nihonbashi, Chuo-ku, Tokyo
Initial Date of Registration : 2010-12-24
Latest Date of Registration : 2012-05-22

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]Date of Issue : 2025-10-07
Valid Till : 2028-10-06
Written Confirmation Number : WC-0119
Address of the Firm : Plot No. 35, 36, 38, 39, 40, 49, 50 & 51, Phase-IV, IDA, Jeedimetla, Medchal-Mal...

Olopatadine Hydrochloride IH/USP/IP
Date of Issue : 2025-08-08
Valid Till : 2028-07-07
Written Confirmation Number : WC-0055
Address of the Firm : Plot No. 19 & 20/2, MIDC, Dhatav, Taluka: Roha, District-Raigad, Maharashtra, In...

Date of Issue : 2025-10-24
Valid Till : 2027-10-23
Written Confirmation Number : WC-0625
Address of the Firm : PLOT NO.: В-29/1, MIDC MAHAD, VILLAGE BIRWADI,MAHAD - 402301, Taluka: MAHAD, Di...

Date of Issue : 2025-08-05
Valid Till : 2028-07-15
Written Confirmation Number : WC-0152
Address of the Firm : Kilo Lab, R-92-93,15 Floor, T.T.C. M.I.D.C., Thane Belapur Road, Navi Mumbai., R...

Olopatadine Hydrochloride IP/JP/USP
Date of Issue : 2022-08-04
Valid Till : 2025-07-22
Written Confirmation Number : WC-0341
Address of the Firm : Plot No. J-46/57, MIDC Taloja Panvel, Raigad-410208, Maharashtra, India

Olopatadine Hydrochloride IH/USP
Date of Issue : 2025-07-07
Valid Till : 2028-07-14
Written Confirmation Number : WC-0021
Address of the Firm : Sy. Nos, 317,320,321,322,323,604 & 605, Pincode-502329, Rudraram (Village), Pata...

Date of Issue : 2022-07-27
Valid Till : 2025-07-26
Written Confirmation Number : WC-0531
Address of the Firm : Plot No. D-90/3 TTC IND Area, MIDC Turbhe, Navi Mumbai- 400705,Taluka: Pawvane, ...

Olopatadine Hydrochloride IH/USP
Date of Issue : 2022-06-08
Valid Till : 2025-07-25
Written Confirmation Number : WC-0159nA2
Address of the Firm : A-7/A-8, M.I.D.C Industrial Area, Ahmednagar-414111, Maharashtra, India

Olopatadine Hydrochloride USP/IH
Date of Issue : 2025-07-14
Valid Till : 2028-02-06
Written Confirmation Number : WC-0012
Address of the Firm : B-1/8,MIDC, Lote Parshuram, Tal., Dist. Ratnagiri, Khed - 415722, Taluka: Khed, ...

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results] Curia - Accelerating therapies from discovery to commercialization with flexible, scalable CDMO solutions.
Curia - Accelerating therapies from discovery to commercialization with flexible, scalable CDMO solutions.
Registrant Name : Goodwills Co., Ltd.
Registration Date : 2021-04-28
Registration Number : 20210428-209-J-960
Manufacturer Name : Curia Spain SAU
Manufacturer Address : Parque Tecnológico Boecillo, Parcelas 2y 3, Boecillo, 47151 Valladolid, Spain
Registrant Name : Kolon Life Science Co., Ltd.
Registration Date : 2024-09-11
Registration Number : 20240911-209-J-1704
Manufacturer Name : Aurore Pharmaceuticals Priva...
Manufacturer Address : Plot No. 35, 36, 38 to 40, 49 to 51 Phase IV, IDA, Jeedimetla, Hyderabad Telangana Je...

Registrant Name : Medipoem Co., Ltd.
Registration Date : 2026-04-14
Registration Number : 20251229-209-J-2080(1)
Manufacturer Name : Chongqing Huapont Shengchem ...
Manufacturer Address : No. 6, Huanan 1st Road, Yanjia Avenue, Changshou District, Chongqing 401221, China

Registrant Name : Ace Biopharm Co., Ltd.
Registration Date : 2025-12-29
Registration Number : 20251229-209-J-2080
Manufacturer Name : Chongqing Huapont Shengchem ...
Manufacturer Address : No. 6, Huanan 1st Road, Yanjia Avenue, Changshou District, Chongqing 401221, China

Registrant Name : Ace Biopharm Co., Ltd.
Registration Date : 2021-03-30
Registration Number : 20210330-209-J-913
Manufacturer Name : FIS-Fabbrica Italiana Sintet...
Manufacturer Address : Viale Milano, 26 Alte di Montecchio Maggiore, Vicenza, Italy

Registrant Name : Next Bio Co., Ltd.
Registration Date : 2026-04-14
Registration Number : 20260414-209-J-2149
Manufacturer Name : Flax Laboratories Private Li...
Manufacturer Address : Plot No. B-29/1, MIDC Mahad, Village-Birwadi, Mahad-402301 Tal-Mahad, Dist-Raigad, Ma...

Registrant Name : Chong Kun Dang Co., Ltd.
Registration Date : 2021-04-22
Registration Number : 20181116-209-J-150(2)
Manufacturer Name : Indoco Remedies Limited (KIL...
Manufacturer Address : R-92/93, 1st floor, TTC, MIDC, Thane Belapur Road, Rabale, Navi Mumbai, Thane 400701....

Registrant Name : Bansen Co., Ltd.
Registration Date : 2022-11-28
Registration Number : 20181116-209-J-150(3)
Manufacturer Name : Indoco Remedies Limited
Manufacturer Address : Kilo Lab, R-92/93, 1st floor TTC MIDC area, Thane-Belapur Road, Rabale, Navi Mumbia 4...

Registrant Name : Novartis Korea
Registration Date : 2021-05-20
Registration Number : 20210503-209-J-967(A)
Manufacturer Name : Kyowa Pharma Chemical Co., L...
Manufacturer Address : 530 Chokeiji, Takaoka, Toyama 933-8511, Japan

Registrant Name : Korea Kyowa Kirin Co., Ltd.
Registration Date : 2021-05-03
Registration Number : 20210503-209-J-967
Manufacturer Name : Kyowa Pharma Chemical Co., L...
Manufacturer Address : 530 Chokeiji, Takaoka, Toyama 933-8511, Japan

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results] Seqens is an integrated global leader in pharmaceutical solutions & specialty ingredients, & custom-made solutions to our customers.
Seqens is an integrated global leader in pharmaceutical solutions & specialty ingredients, & custom-made solutions to our customers.
NDC Package Code : 51014-7733
Start Marketing Date : 2010-04-26
End Marketing Date : 2026-12-31
Dosage Form (Strength) : POWDER (1kg/kg)
Marketing Category : BULK INGREDIENT
 Curia - Accelerating therapies from discovery to commercialization with flexible, scalable CDMO solutions.
Curia - Accelerating therapies from discovery to commercialization with flexible, scalable CDMO solutions.
NDC Package Code : 65089-0041
Start Marketing Date : 1996-12-18
End Marketing Date : 2026-12-31
Dosage Form (Strength) : POWDER (1kg/kg)
Marketing Category : BULK INGREDIENT
NDC Package Code : 73377-336
Start Marketing Date : 2025-10-28
End Marketing Date : 2026-12-31
Dosage Form (Strength) : POWDER (1g/g)
Marketing Category : BULK INGREDIENT FOR HUMAN P...

NDC Package Code : 52133-0036
Start Marketing Date : 1996-12-18
End Marketing Date : 2026-12-31
Dosage Form (Strength) : POWDER (1kg/kg)
Marketing Category : BULK INGREDIENT

NDC Package Code : 48087-0165
Start Marketing Date : 2023-09-05
End Marketing Date : 2026-12-31
Dosage Form (Strength) : POWDER (1kg/kg)
Marketing Category : BULK INGREDIENT

NDC Package Code : 55545-0464
Start Marketing Date : 2015-05-02
End Marketing Date : 2026-12-31
Dosage Form (Strength) : POWDER (1kg/kg)
Marketing Category : BULK INGREDIENT

NDC Package Code : 12079-2002
Start Marketing Date : 2013-10-15
End Marketing Date : 2026-12-31
Dosage Form (Strength) : POWDER (1kg/kg)
Marketing Category : BULK INGREDIENT

NDC Package Code : 53296-0078
Start Marketing Date : 2010-05-27
End Marketing Date : 2026-12-31
Dosage Form (Strength) : POWDER (75kg/75kg)
Marketing Category : BULK INGREDIENT

NDC Package Code : 14501-0050
Start Marketing Date : 2010-03-25
End Marketing Date : 2026-12-31
Dosage Form (Strength) : POWDER (1kg/kg)
Marketing Category : BULK INGREDIENT

NDC Package Code : 62756-041
Start Marketing Date : 2025-03-31
End Marketing Date : 2026-12-31
Dosage Form (Strength) : POWDER (5kg/5kg)
Marketing Category : BULK INGREDIENT

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results] Seqens is an integrated global leader in pharmaceutical solutions & specialty ingredients, & custom-made solutions to our customers.
Seqens is an integrated global leader in pharmaceutical solutions & specialty ingredients, & custom-made solutions to our customers.
About the Company : Founded in 2003, Seqens has evolved into a global leader in pharmaceutical solutions and specialty ingredients. The company supports customers in the development, scale-up, and man...
 Bhavna Laboratories is an API & Intermediate manufacturer focusing on the muscle relaxant & ophthalmic segments.
Bhavna Laboratories is an API & Intermediate manufacturer focusing on the muscle relaxant & ophthalmic segments.
About the Company : Bhavna Laboratories Pvt. Ltd., founded in 1986, is an Indian manufacturer of APIs and intermediates. The company is approved by the FDA of India and holds cGMP certification for it...
 LGM Pharma accelerates & optimizes the new product pathway from early development through commercialization.
LGM Pharma accelerates & optimizes the new product pathway from early development through commercialization.
About the Company : LGM Pharma is a global leader in sourcing APIs, including hard-to-find drug substances, for pharmaceutical and biotech industries. LGM also operates as a full-service drug product ...
About the Company : Jai Radhe Sales, founded in 1999, is a global distributor specializing in high-quality pharmaceutical ingredients from India. It offers complete sourcing solutions, technical and r...
About the Company : HRV Pharma is a global manufacturer, seller, and exporter of APIs, intermediates, pellets, food-grade chemicals, food additives, and food ingredients. The company provides sourcing...
 Tenatra connects Indian manufacturers with global buyers through active partners in Germany, Switzerland, Belgium, Spain & Turkey.
Tenatra connects Indian manufacturers with global buyers through active partners in Germany, Switzerland, Belgium, Spain & Turkey.
About the Company : Tenatra International was established as a proprietorship firm in 1999. It got off to a very good start, supporting clients in the United States, Mexico and Europe. As business opp...
About the Company : Zeon Pharma Industries India Pvt. Ltd. is an ISO 9001:2015, cGMP, and WHO-GMP certified company with a dedicated manufacturing facility for Bulk Drugs (APIs), phytochemicals, herba...
About the Company : Shamrock Pharmachemi Pvt Ltd. is a globally recognized API leader with 26+ years in human and veterinary pharmaceuticals. Operating in 40+ countries, it owns two Gujarat facilities...
About the Company : BIOTECHNICA DWC LLC has carved a niche for itself in providing value added compliance, regulatory qualification, project management and GDP guidance services to pharma companies al...

About the Company : Dalian Richon Chem. Co., Ltd is a high-tech enterprise professionally involved in research and development, manufacturing process and sales service of pharmaceutical intermediates ...

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]Details:
Olopatadine HCl, a miscellaneous product targeting the Histamine H1 receptor, shows promise in relieving itchy eyes.
Lead Product(s): Olopatadine Hydrochloride,Inapplicable
Therapeutic Area: Ophthalmology Brand Name: Pataday-Generic
Study Phase: Approved FDFProduct Type: Miscellaneous
Sponsor: Undisclosed
Deal Size: Inapplicable Upfront Cash: Inapplicable
Deal Type: Inapplicable January 14, 2026
Lead Product(s) : Olopatadine Hydrochloride,Inapplicable
Therapeutic Area : Ophthalmology
Highest Development Status : Approved FDF
Partner/Sponsor/Collaborator : Undisclosed
Deal Size : Inapplicable
Deal Type : Inapplicable
Dr. Reddy’s Launches Olopatadine Hydrochloride Ophthalmic Solution in the U.S.
Details : Olopatadine HCl, a miscellaneous product targeting the Histamine H1 receptor, shows promise in relieving itchy eyes.
Product Name : Pataday-Generic
Product Type : Miscellaneous
Upfront Cash : Inapplicable
January 14, 2026
Details:
Dr. Reddy’s has launched of over-the-counter Olopatadine Hydrochloride Ophthalmic Solution USP, 0.2% and 0.1%, the storebrand equivalents of Pataday® Once Daily Relief and Pataday® Twice Daily Relief, in the U.S. market, as approved by the USFDA.
Lead Product(s): Olopatadine Hydrochloride,Inapplicable
Therapeutic Area: Ophthalmology Brand Name: Pataday-Generic
Study Phase: Approved FDFProduct Type: Miscellaneous
Sponsor: Undisclosed
Deal Size: Inapplicable Upfront Cash: Inapplicable
Deal Type: Inapplicable September 17, 2020
Lead Product(s) : Olopatadine Hydrochloride,Inapplicable
Therapeutic Area : Ophthalmology
Highest Development Status : Approved FDF
Partner/Sponsor/Collaborator : Undisclosed
Deal Size : Inapplicable
Deal Type : Inapplicable
Details : Dr. Reddy’s has launched of over-the-counter Olopatadine Hydrochloride Ophthalmic Solution USP, 0.2% and 0.1%, the storebrand equivalents of Pataday® Once Daily Relief and Pataday® Twice Daily Relief, in the U.S. market, as approved by the USFDA.
Product Name : Pataday-Generic
Product Type : Miscellaneous
Upfront Cash : Inapplicable
September 17, 2020
 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Details:
Olopatadine HCl, a histamine H1 receptor antagonist, shows promise in treating allergic rhinitis.
Lead Product(s): Olopatadine Hydrochloride,Mometasone Furoate
Therapeutic Area: Immunology Brand Name: Ryaltris
Study Phase: Approved FDFProduct Type: Miscellaneous
Sponsor: Undisclosed
Deal Size: Inapplicable Upfront Cash: Inapplicable
Deal Type: Inapplicable November 11, 2025

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Lead Product(s) : Olopatadine Hydrochloride,Mometasone Furoate
Therapeutic Area : Immunology
Highest Development Status : Approved FDF
Partner/Sponsor/Collaborator : Undisclosed
Deal Size : Inapplicable
Deal Type : Inapplicable
Glenmark Specialty Gets NMPA Approval for RYALTRIS Nasal Spray
Details : Olopatadine HCl, a histamine H1 receptor antagonist, shows promise in treating allergic rhinitis.
Product Name : Ryaltris
Product Type : Miscellaneous
Upfront Cash : Inapplicable
November 11, 2025

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Details:
Olopatadine HCl, a miscellaneous product targeting the Histamine H1 receptor, shows promising results in treating Allergic Rhinitis.
Lead Product(s): Olopatadine Hydrochloride,Mometasone Furoate
Therapeutic Area: Immunology Brand Name: Ryaltris
Study Phase: Approved FDFProduct Type: Miscellaneous
Sponsor: Undisclosed
Deal Size: Inapplicable Upfront Cash: Inapplicable
Deal Type: Inapplicable November 11, 2025

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Lead Product(s) : Olopatadine Hydrochloride,Mometasone Furoate
Therapeutic Area : Immunology
Highest Development Status : Approved FDF
Partner/Sponsor/Collaborator : Undisclosed
Deal Size : Inapplicable
Deal Type : Inapplicable
Glenmark Specialty Gets NMPA Approval for RYALTRIS Nasal Spray
Details : Olopatadine HCl, a miscellaneous product targeting the Histamine H1 receptor, shows promising results in treating Allergic Rhinitis.
Product Name : Ryaltris
Product Type : Miscellaneous
Upfront Cash : Inapplicable
November 11, 2025

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Details:
Olopatadine HCl is a mast cell stabilizer and a histamine H1 antagonist. It is indicated for the treatment of ocular itching associated with allergic conjunctivitis.
Lead Product(s): Olopatadine Hydrochloride,Inapplicable
Therapeutic Area: Ophthalmology Brand Name: Pataday-Generic
Study Phase: Approved FDFProduct Type: Miscellaneous
Sponsor: Undisclosed
Deal Size: Inapplicable Upfront Cash: Inapplicable
Deal Type: Inapplicable June 16, 2025

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Lead Product(s) : Olopatadine Hydrochloride,Inapplicable
Therapeutic Area : Ophthalmology
Highest Development Status : Approved FDF
Partner/Sponsor/Collaborator : Undisclosed
Deal Size : Inapplicable
Deal Type : Inapplicable
Glenmark USA Launches OTC Olopatadine Hydrochloride Ophthalmic Solution 0.2%
Details : Olopatadine HCl is a mast cell stabilizer and a histamine H1 antagonist. It is indicated for the treatment of ocular itching associated with allergic conjunctivitis.
Product Name : Pataday-Generic
Product Type : Miscellaneous
Upfront Cash : Inapplicable
June 16, 2025

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Details:
Pataday-Generic (olopatadine hydrochloride) solution is a mast cell stabilizer indicated for the treatment of ocular itching associated with allergic conjunctivitis
Lead Product(s): Olopatadine Hydrochloride,Inapplicable
Therapeutic Area: Ophthalmology Brand Name: Pataday-Generic
Study Phase: Approved FDFProduct Type: Miscellaneous
Sponsor: Undisclosed
Deal Size: Inapplicable Upfront Cash: Inapplicable
Deal Type: Inapplicable March 20, 2025

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Lead Product(s) : Olopatadine Hydrochloride,Inapplicable
Therapeutic Area : Ophthalmology
Highest Development Status : Approved FDF
Partner/Sponsor/Collaborator : Undisclosed
Deal Size : Inapplicable
Deal Type : Inapplicable
Glenmark Secures ANDA for Olopatadine Hydrochloride Ophthalmic Solution
Details : Pataday-Generic (olopatadine hydrochloride) solution is a mast cell stabilizer indicated for the treatment of ocular itching associated with allergic conjunctivitis
Product Name : Pataday-Generic
Product Type : Miscellaneous
Upfront Cash : Inapplicable
March 20, 2025

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Details:
Olopatadine HCl solution is a mast cell stabilizer, works by H1R antagonist, indicated for the treatment of ocular itching associated with allergic conjunctivitis.
Lead Product(s): Olopatadine Hydrochloride,Inapplicable
Therapeutic Area: Ophthalmology Brand Name: Pataday-Generic
Study Phase: Approved FDFProduct Type: Miscellaneous
Sponsor: Undisclosed
Deal Size: Inapplicable Upfront Cash: Inapplicable
Deal Type: Inapplicable December 04, 2024

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Lead Product(s) : Olopatadine Hydrochloride,Inapplicable
Therapeutic Area : Ophthalmology
Highest Development Status : Approved FDF
Partner/Sponsor/Collaborator : Undisclosed
Deal Size : Inapplicable
Deal Type : Inapplicable
Alembic Pharma Gets Tentative USFDA Nod for Olopatadine Drug
Details : Olopatadine HCl solution is a mast cell stabilizer, works by H1R antagonist, indicated for the treatment of ocular itching associated with allergic conjunctivitis.
Product Name : Pataday-Generic
Product Type : Miscellaneous
Upfront Cash : Inapplicable
December 04, 2024

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Details:
Pataday-Generic (olopatadine hydrochloride) is a solution that is a mast cell stabilizer indicated for the treatment of ocular itching associated with allergic conjunctivitis.
Lead Product(s): Olopatadine Hydrochloride,Inapplicable
Therapeutic Area: Ophthalmology Brand Name: Pataday-Generic
Study Phase: Approved FDFProduct Type: Miscellaneous
Sponsor: Undisclosed
Deal Size: Inapplicable Upfront Cash: Inapplicable
Deal Type: Inapplicable August 19, 2024

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Lead Product(s) : Olopatadine Hydrochloride,Inapplicable
Therapeutic Area : Ophthalmology
Highest Development Status : Approved FDF
Partner/Sponsor/Collaborator : Undisclosed
Deal Size : Inapplicable
Deal Type : Inapplicable
Glenmark Launches OTC Olopatadine Hydrochloride Eye Drops in USA
Details : Pataday-Generic (olopatadine hydrochloride) is a solution that is a mast cell stabilizer indicated for the treatment of ocular itching associated with allergic conjunctivitis.
Product Name : Pataday-Generic
Product Type : Miscellaneous
Upfront Cash : Inapplicable
August 19, 2024

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Details:
Azelastine HCl is a Other Small Molecule drug candidate, which is currently being evaluated in phase II clinical studies for the treatment of Rhinitis, Allergic, Seasonal.
Lead Product(s): Azelastine Hydrochloride,Mometasone Furoate,Olopatadine Hydrochloride
Therapeutic Area: Immunology Brand Name: Undisclosed
Study Phase: Phase IIProduct Type: Miscellaneous
Sponsor: Undisclosed
Deal Size: Inapplicable Upfront Cash: Inapplicable
Deal Type: Inapplicable November 13, 2023

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Lead Product(s) : Azelastine Hydrochloride,Mometasone Furoate,Olopatadine Hydrochloride
Therapeutic Area : Immunology
Highest Development Status : Phase II
Partner/Sponsor/Collaborator : Undisclosed
Deal Size : Inapplicable
Deal Type : Inapplicable
Azelastine Allergen Chamber - Onset of Action Study
Details : Azelastine HCl is a Other Small Molecule drug candidate, which is currently being evaluated in phase II clinical studies for the treatment of Rhinitis, Allergic, Seasonal.
Product Name : Undisclosed
Product Type : Miscellaneous
Upfront Cash : Inapplicable
November 13, 2023

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Details:
RYALTRIS® (olopatadine hydrochloride and mometasone furoate nasal spray) treats moderate to severe seasonal allergic rhinitis (SAR) and associated ocular symptoms in adults, adolescents, and children aged 6 years and older.
Lead Product(s): Olopatadine Hydrochloride,Mometasone Furoate
Therapeutic Area: Immunology Brand Name: Ryaltris
Study Phase: Approved FDFProduct Type: Miscellaneous
Sponsor: Glenmark Pharmaceuticals
Deal Size: Inapplicable Upfront Cash: Inapplicable
Deal Type: Inapplicable September 23, 2022

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Lead Product(s) : Olopatadine Hydrochloride,Mometasone Furoate
Therapeutic Area : Immunology
Highest Development Status : Approved FDF
Partner/Sponsor/Collaborator : Glenmark Pharmaceuticals
Deal Size : Inapplicable
Deal Type : Inapplicable
Bausch Health and Glenmark Announce the Approval of RYALTRIS® in Canada
Details : RYALTRIS® (olopatadine hydrochloride and mometasone furoate nasal spray) treats moderate to severe seasonal allergic rhinitis (SAR) and associated ocular symptoms in adults, adolescents, and children aged 6 years and older.
Product Name : Ryaltris
Product Type : Miscellaneous
Upfront Cash : Inapplicable
September 23, 2022

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results](3-(Dimethylamino)propyl)triphenylphosphonium brom...
CAS Number : 27710-82-3
End Use API : Olopatadine Hydrochloride
About The Company : Bhavna Laboratories Pvt. Ltd., founded in 1986, is an Indian manufacturer of APIs and intermediates. The company is approved by the FDA of India and holds cGMP ...
Isoxepac [(11-Oxo-6,11-dihydrodibenzo[b,e]oxepin-2...
CAS Number : 55453-87-7
End Use API : Olopatadine Hydrochloride
About The Company : Bhavna Laboratories Pvt. Ltd., founded in 1986, is an Indian manufacturer of APIs and intermediates. The company is approved by the FDA of India and holds cGMP ...
CAS Number : 2971-79-1
End Use API : Olopatadine Hydrochloride
About The Company : Tagoor Laboratories, established in 2018 and part of the Tagoor Group, provides APIs, advanced intermediates, and key starting materials for critical and high-g...
(3-(dimethylamino)propyl)triphenyl phosphonium bro...
CAS Number : 3934-20-1
End Use API : Olopatadine Hydrochloride
About The Company : Indian Drugs And Chemicals supplies quality products using modern technologies, supported by strong industry expertise. The company serves global markets with a...
[3-(dimethylamino)propyl]triphenylphosphonium brom...
CAS Number : 27710-82-3
End Use API : Olopatadine Hydrochloride
About The Company : Dalian Richon Chem. Co., Ltd is a high-tech enterprise professionally involved in research and development, manufacturing process and sales service of pharmaceu...

2-[[4-(Carboxymethyl)phenoxy]methyl]benzoic Acid
CAS Number : CAS-55453-89-9
End Use API : Olopatadine Hydrochloride
About The Company : Jinan Lead Pharmaceutical Technology Co., LTD., located in Jinan, Shandong Province, is mainly engaged in R&D, production and sales of pharmaceutical raw materi...

CAS Number : CAS-55453-87-7
End Use API : Olopatadine Hydrochloride
About The Company : Jinan Lead Pharmaceutical Technology Co., LTD., located in Jinan, Shandong Province, is mainly engaged in R&D, production and sales of pharmaceutical raw materi...

[3-(Dimethylamino)propyl]triphenylphosphonium bro...
CAS Number : CAS-27710-82-3
End Use API : Olopatadine Hydrochloride
About The Company : Jinan Lead Pharmaceutical Technology Co., LTD., located in Jinan, Shandong Province, is mainly engaged in R&D, production and sales of pharmaceutical raw materi...

2-(11-Oxo-6,11-dihydrodibenzo[b,e]oxepin-2-yl)acet...
CAS Number : 55453-87-7
End Use API : Olopatadine Hydrochloride
About The Company : Established in 1997, the PRECISE Group is a respected pharmaceutical company in India with an annual turnover of USD 40 million. Backed by a skilled regulatory ...

6,11-Dihydro-1 1-oxodibenz[b,e] oxepin-2-acetic 55...
CAS Number : 55453-87-7
End Use API : Olopatadine Hydrochloride
About The Company : Progress is a company led by professionals, established by technocrats with a vision for global business expansion. Our headquarters are located in Vashi, Mumba...

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]DRUG PRODUCT COMPOSITIONS

Related Excipient Companies

Excipients by Applications
Global Sales Information

Market Place

Patents & EXCLUSIVITIES
REF. STANDARDS & IMPURITIES

ANALYTICAL

ABOUT THIS PAGE
91
PharmaCompass offers a list of Olopatadine Hydrochloride API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Olopatadine Hydrochloride manufacturer or Olopatadine Hydrochloride supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Olopatadine Hydrochloride manufacturer or Olopatadine Hydrochloride supplier.
A 11-((Z)-3-(Dimethylamino)propylidene)-6,11-dihydrodibenz(b,e)oxepin-2-acetic acid, hydrochloride manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of 11-((Z)-3-(Dimethylamino)propylidene)-6,11-dihydrodibenz(b,e)oxepin-2-acetic acid, hydrochloride, including repackagers and relabelers. The FDA regulates 11-((Z)-3-(Dimethylamino)propylidene)-6,11-dihydrodibenz(b,e)oxepin-2-acetic acid, hydrochloride manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. 11-((Z)-3-(Dimethylamino)propylidene)-6,11-dihydrodibenz(b,e)oxepin-2-acetic acid, hydrochloride API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
click here to find a list of 11-((Z)-3-(Dimethylamino)propylidene)-6,11-dihydrodibenz(b,e)oxepin-2-acetic acid, hydrochloride manufacturers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PhamaCompass.
A 11-((Z)-3-(Dimethylamino)propylidene)-6,11-dihydrodibenz(b,e)oxepin-2-acetic acid, hydrochloride supplier is an individual or a company that provides 11-((Z)-3-(Dimethylamino)propylidene)-6,11-dihydrodibenz(b,e)oxepin-2-acetic acid, hydrochloride active pharmaceutical ingredient (API) or 11-((Z)-3-(Dimethylamino)propylidene)-6,11-dihydrodibenz(b,e)oxepin-2-acetic acid, hydrochloride finished formulations upon request. The 11-((Z)-3-(Dimethylamino)propylidene)-6,11-dihydrodibenz(b,e)oxepin-2-acetic acid, hydrochloride suppliers may include 11-((Z)-3-(Dimethylamino)propylidene)-6,11-dihydrodibenz(b,e)oxepin-2-acetic acid, hydrochloride API manufacturers, exporters, distributors and traders.
click here to find a list of 11-((Z)-3-(Dimethylamino)propylidene)-6,11-dihydrodibenz(b,e)oxepin-2-acetic acid, hydrochloride suppliers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PharmaCompass.
A 11-((Z)-3-(Dimethylamino)propylidene)-6,11-dihydrodibenz(b,e)oxepin-2-acetic acid, hydrochloride DMF (Drug Master File) is a document detailing the whole manufacturing process of 11-((Z)-3-(Dimethylamino)propylidene)-6,11-dihydrodibenz(b,e)oxepin-2-acetic acid, hydrochloride active pharmaceutical ingredient (API) in detail. Different forms of 11-((Z)-3-(Dimethylamino)propylidene)-6,11-dihydrodibenz(b,e)oxepin-2-acetic acid, hydrochloride DMFs exist exist since differing nations have different regulations, such as 11-((Z)-3-(Dimethylamino)propylidene)-6,11-dihydrodibenz(b,e)oxepin-2-acetic acid, hydrochloride USDMF, ASMF (EDMF), JDMF, CDMF, etc.
A 11-((Z)-3-(Dimethylamino)propylidene)-6,11-dihydrodibenz(b,e)oxepin-2-acetic acid, hydrochloride DMF submitted to regulatory agencies in the US is known as a USDMF. 11-((Z)-3-(Dimethylamino)propylidene)-6,11-dihydrodibenz(b,e)oxepin-2-acetic acid, hydrochloride USDMF includes data on 11-((Z)-3-(Dimethylamino)propylidene)-6,11-dihydrodibenz(b,e)oxepin-2-acetic acid, hydrochloride's chemical properties, information on the facilities and procedures used, and details about packaging and storage. The 11-((Z)-3-(Dimethylamino)propylidene)-6,11-dihydrodibenz(b,e)oxepin-2-acetic acid, hydrochloride USDMF is kept confidential to protect the manufacturer’s intellectual property.
click here to find a list of 11-((Z)-3-(Dimethylamino)propylidene)-6,11-dihydrodibenz(b,e)oxepin-2-acetic acid, hydrochloride suppliers with USDMF on PharmaCompass.
The Pharmaceuticals and Medical Devices Agency (PMDA) established the Japan Drug Master File (JDMF), also known as the Master File (MF), to permit Japanese and foreign manufacturers of drug substances, intermediates, excipients, raw materials, and packaging materials (‘Products’) to voluntarily register confidential information about the production and management of their products in Japan.
The 11-((Z)-3-(Dimethylamino)propylidene)-6,11-dihydrodibenz(b,e)oxepin-2-acetic acid, hydrochloride Drug Master File in Japan (11-((Z)-3-(Dimethylamino)propylidene)-6,11-dihydrodibenz(b,e)oxepin-2-acetic acid, hydrochloride JDMF) empowers 11-((Z)-3-(Dimethylamino)propylidene)-6,11-dihydrodibenz(b,e)oxepin-2-acetic acid, hydrochloride API manufacturers to present comprehensive information (e.g., production methods, data, etc.) to the review authority, i.e., PMDA (Pharmaceuticals & Medical Devices Agency).
PMDA reviews the 11-((Z)-3-(Dimethylamino)propylidene)-6,11-dihydrodibenz(b,e)oxepin-2-acetic acid, hydrochloride JDMF during the approval evaluation for pharmaceutical products. At the time of 11-((Z)-3-(Dimethylamino)propylidene)-6,11-dihydrodibenz(b,e)oxepin-2-acetic acid, hydrochloride JDMF registration, PMDA checks if the format is accurate, if the necessary items have been included (application), and if data has been attached.
click here to find a list of 11-((Z)-3-(Dimethylamino)propylidene)-6,11-dihydrodibenz(b,e)oxepin-2-acetic acid, hydrochloride suppliers with JDMF on PharmaCompass.
In Korea, the Ministry of Food and Drug Safety (MFDS) is in charge of regulating pharmaceutical products and services.
Pharmaceutical companies submit a 11-((Z)-3-(Dimethylamino)propylidene)-6,11-dihydrodibenz(b,e)oxepin-2-acetic acid, hydrochloride Drug Master File in Korea (11-((Z)-3-(Dimethylamino)propylidene)-6,11-dihydrodibenz(b,e)oxepin-2-acetic acid, hydrochloride KDMF) to the MFDS, which includes comprehensive information about the production, processing, facilities, materials, packaging, and testing of 11-((Z)-3-(Dimethylamino)propylidene)-6,11-dihydrodibenz(b,e)oxepin-2-acetic acid, hydrochloride. The MFDS reviews the 11-((Z)-3-(Dimethylamino)propylidene)-6,11-dihydrodibenz(b,e)oxepin-2-acetic acid, hydrochloride KDMF as part of the drug registration process and uses the information provided in the 11-((Z)-3-(Dimethylamino)propylidene)-6,11-dihydrodibenz(b,e)oxepin-2-acetic acid, hydrochloride KDMF to evaluate the safety and efficacy of the drug.
After submitting a 11-((Z)-3-(Dimethylamino)propylidene)-6,11-dihydrodibenz(b,e)oxepin-2-acetic acid, hydrochloride KDMF to the MFDS, the registered manufacturer can provide importers or distributors with the registration number without revealing confidential information to Korean business partners. Applicants seeking to register their 11-((Z)-3-(Dimethylamino)propylidene)-6,11-dihydrodibenz(b,e)oxepin-2-acetic acid, hydrochloride API can apply through the Korea Drug Master File (KDMF).
click here to find a list of 11-((Z)-3-(Dimethylamino)propylidene)-6,11-dihydrodibenz(b,e)oxepin-2-acetic acid, hydrochloride suppliers with KDMF on PharmaCompass.
A 11-((Z)-3-(Dimethylamino)propylidene)-6,11-dihydrodibenz(b,e)oxepin-2-acetic acid, hydrochloride written confirmation (11-((Z)-3-(Dimethylamino)propylidene)-6,11-dihydrodibenz(b,e)oxepin-2-acetic acid, hydrochloride WC) is an official document issued by a regulatory agency to a 11-((Z)-3-(Dimethylamino)propylidene)-6,11-dihydrodibenz(b,e)oxepin-2-acetic acid, hydrochloride manufacturer, verifying that the manufacturing facility of a 11-((Z)-3-(Dimethylamino)propylidene)-6,11-dihydrodibenz(b,e)oxepin-2-acetic acid, hydrochloride active pharmaceutical ingredient (API) adheres to the Good Manufacturing Practices (GMP) regulations of the importing country. When exporting 11-((Z)-3-(Dimethylamino)propylidene)-6,11-dihydrodibenz(b,e)oxepin-2-acetic acid, hydrochloride APIs or 11-((Z)-3-(Dimethylamino)propylidene)-6,11-dihydrodibenz(b,e)oxepin-2-acetic acid, hydrochloride finished pharmaceutical products to another nation, regulatory agencies frequently require a 11-((Z)-3-(Dimethylamino)propylidene)-6,11-dihydrodibenz(b,e)oxepin-2-acetic acid, hydrochloride WC (written confirmation) as part of the regulatory process.
click here to find a list of 11-((Z)-3-(Dimethylamino)propylidene)-6,11-dihydrodibenz(b,e)oxepin-2-acetic acid, hydrochloride suppliers with Written Confirmation (WC) on PharmaCompass.
National Drug Code is a comprehensive database maintained by the FDA that contains information on all drugs marketed in the US. This directory includes information about finished drug products, unfinished drug products, and compounded drug products, including those containing 11-((Z)-3-(Dimethylamino)propylidene)-6,11-dihydrodibenz(b,e)oxepin-2-acetic acid, hydrochloride as an active pharmaceutical ingredient (API).
The FDA updates the NDC directory daily. The NDC numbers for 11-((Z)-3-(Dimethylamino)propylidene)-6,11-dihydrodibenz(b,e)oxepin-2-acetic acid, hydrochloride API and other APIs are published in this directory by the FDA.
The NDC unfinished drugs database includes product listing information submitted for all unfinished drugs, such as active pharmaceutical ingredients (APIs), drugs intended for further processing and bulk drug substances for compounding.
Pharmaceutical companies that manufacture 11-((Z)-3-(Dimethylamino)propylidene)-6,11-dihydrodibenz(b,e)oxepin-2-acetic acid, hydrochloride as an active pharmaceutical ingredient (API) must furnish the FDA with an updated record of all drugs that they produce, prepare, propagate, compound, or process for commercial distribution in the US at their facilities.
The NDC directory also contains data on finished compounded human drug products that contain 11-((Z)-3-(Dimethylamino)propylidene)-6,11-dihydrodibenz(b,e)oxepin-2-acetic acid, hydrochloride and are produced by outsourcing facilities. While these outsourcing facilities are not mandated to assign a 11-((Z)-3-(Dimethylamino)propylidene)-6,11-dihydrodibenz(b,e)oxepin-2-acetic acid, hydrochloride NDC to their finished compounded human drug products, they may choose to do so.
click here to find a list of 11-((Z)-3-(Dimethylamino)propylidene)-6,11-dihydrodibenz(b,e)oxepin-2-acetic acid, hydrochloride suppliers with NDC on PharmaCompass.
11-((Z)-3-(Dimethylamino)propylidene)-6,11-dihydrodibenz(b,e)oxepin-2-acetic acid, hydrochloride Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of 11-((Z)-3-(Dimethylamino)propylidene)-6,11-dihydrodibenz(b,e)oxepin-2-acetic acid, hydrochloride GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right 11-((Z)-3-(Dimethylamino)propylidene)-6,11-dihydrodibenz(b,e)oxepin-2-acetic acid, hydrochloride GMP manufacturer or 11-((Z)-3-(Dimethylamino)propylidene)-6,11-dihydrodibenz(b,e)oxepin-2-acetic acid, hydrochloride GMP API supplier for your needs.
A 11-((Z)-3-(Dimethylamino)propylidene)-6,11-dihydrodibenz(b,e)oxepin-2-acetic acid, hydrochloride CoA (Certificate of Analysis) is a formal document that attests to 11-((Z)-3-(Dimethylamino)propylidene)-6,11-dihydrodibenz(b,e)oxepin-2-acetic acid, hydrochloride's compliance with 11-((Z)-3-(Dimethylamino)propylidene)-6,11-dihydrodibenz(b,e)oxepin-2-acetic acid, hydrochloride specifications and serves as a tool for batch-level quality control.
11-((Z)-3-(Dimethylamino)propylidene)-6,11-dihydrodibenz(b,e)oxepin-2-acetic acid, hydrochloride CoA mostly includes findings from lab analyses of a specific batch. For each 11-((Z)-3-(Dimethylamino)propylidene)-6,11-dihydrodibenz(b,e)oxepin-2-acetic acid, hydrochloride CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
11-((Z)-3-(Dimethylamino)propylidene)-6,11-dihydrodibenz(b,e)oxepin-2-acetic acid, hydrochloride may be tested according to a variety of international standards, such as European Pharmacopoeia (11-((Z)-3-(Dimethylamino)propylidene)-6,11-dihydrodibenz(b,e)oxepin-2-acetic acid, hydrochloride EP), 11-((Z)-3-(Dimethylamino)propylidene)-6,11-dihydrodibenz(b,e)oxepin-2-acetic acid, hydrochloride JP (Japanese Pharmacopeia) and the US Pharmacopoeia (11-((Z)-3-(Dimethylamino)propylidene)-6,11-dihydrodibenz(b,e)oxepin-2-acetic acid, hydrochloride USP).

