Synopsis
Synopsis
0
CEP/COS
0
JDMF
0
EU WC
0
KDMF
0
NDC API
0
VMF
0
Listed Suppliers
0
EDQM
0
USP
0
JP
0
Others
0
FDF Dossiers
0
FDA Orange Book
0
Europe
0
Canada
0
Australia
0
South Africa
0
Listed Dossiers
DRUG PRODUCT COMPOSITIONS
0
US Patents
0
US Exclusivities
0
Health Canada Patents
US Medicaid
NA
Annual Reports
NA
Regulatory FDF Prices
NA
0
API
0
FDF
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
0
News #PharmaBuzz
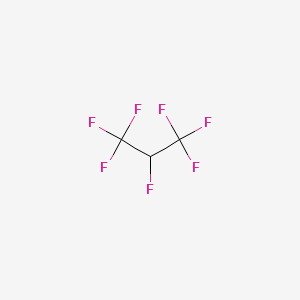

1. 2-hydroheptafluoropropane
2. 2h-heptafluoropropane
3. Apaflurane
4. Hfa 227
5. Hfa-227
6. Hydrofluoroalkane
1. 431-89-0
2. Apaflurane
3. 2h-heptafluoropropane
4. Hfc-227ea
5. 2-hydroheptafluoropropane
6. Solkane 227
7. Propane, 1,1,1,2,3,3,3-heptafluoro-
8. Hfa-227
9. Hfc 227
10. Hfa 227ea
11. Hfc 227ea
12. R40p36gdk6
13. Hydrofluorocarbon 227ea
14. 1,1,1,2,3,3,3-heptafluoropropane (fc 227ea)
15. R-227
16. Khladon 227
17. 2-hydroperfluoropropane
18. Apaflurane [inn:ban]
19. 2h-perfluoropropane
20. Ccris 7786
21. Hfa 227
22. Einecs 207-079-2
23. Fm 200
24. Unii-r40p36gdk6
25. Hsdb 7830
26. Hfc-227
27. Apaflurane [ii]
28. Apaflurane (inn/ban)
29. Cf3chfcf3
30. Apaflurane [inn]
31. Ec 207-079-2
32. Apaflurane [mart.]
33. Schembl19860
34. Chembl2104472
35. Dtxsid4042048
36. Zinc8214485
37. Mfcd00043834
38. 1,1,1,2,3,3,3 Heptafluoropropane
39. Akos006229361
40. 1,1,1,2,3,3,3-heptafluoro-propane
41. Hydrofluorocarbon 227ea [inci]
42. Db-051054
43. 1,1,1,2,3,3,3-heptakis(fluoranyl)propane
44. 2-propanyl, 1,1,1,2,3,3,3-heptafluoro-
45. D10216
46. A826208
47. Q2683759
| Molecular Weight | 170.03 g/mol |
|---|---|
| Molecular Formula | C3HF7 |
| XLogP3 | 2.9 |
| Hydrogen Bond Donor Count | 0 |
| Hydrogen Bond Acceptor Count | 7 |
| Rotatable Bond Count | 0 |
| Exact Mass | 169.99664717 g/mol |
| Monoisotopic Mass | 169.99664717 g/mol |
| Topological Polar Surface Area | 0 Ų |
| Heavy Atom Count | 10 |
| Formal Charge | 0 |
| Complexity | 93 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
Oral absorption is rapid but less complete than pulmonary absorption. Skin absorption is insignificant, except in patients with skin breakdown (e.g., burns, ulcers, severe ichthyosis). Increased metabolic rate can lead to greater inhalational absorption as well. Peak blood levels occur soon after inhalation but occur in 1 to 2 hours after oral administration. The chemicals distribute to tissues with high blood flow (e.g., brain, heart, liver, kidney) and then to adipose tissue, where the highest chemical concentrations are typically found. Halogenated hydrocarbons are metabolized in the liver by cytochrome P-450 oxidation. Partial glutathione conjugation may occur. Halogenated solvents can be excreted unchanged through the lungs. Elimination half-lives can be increased because of either prolonged exposure or hepatic dysfunction. Prolonged exposure allows more chemical to be stored in the adipose tissue, which serves as a source of continued release. /Halogenated Hydrocarbons - Halogenated Solvents/
Dart, R.C. (ed). Medical Toxicology. Third Edition, Lippincott Williams & Wilkins. Philadelphia, PA. 2004., p. 1339
... The safety and pharmacokinetics of HFC 134a (1,1,1,2-tetrafluoroethane) and HFC 227 (1,1,1,2,3,3, 3-heptafluoropropane) were assessed in two separate double-blind studies. Each HFC (hydrofluorocarbon) was administered via whole-body exposure as a vapor to eight (four male and four female) healthy volunteers. Volunteers were exposed, once weekly for 1 hr, first to air and then to ascending concentrations of HFC (1000, 2000, 4000, and 8000 ppm), interspersed with a second air exposure and two CFC 12 (dichlorodifluoromethane) exposures (1000 and 4000 ppm). HFC 134a, HFC 227, and CFC 12 blood concentrations increased rapidly and in an exposure-concentration-dependent manner, although not strictly proportionally, and approached steady state. Maximum blood concentrations (C(max)) tended to be higher in males than females; in the HFC 227 study, these were statistically significantly (P < 0. 05) higher in males for each HFC 227 and CFC 12 exposure level. ... In the HFC 227 study, t(1/2)alpha (alpha elimination half-life) for both CFC 12 and HFC 227, at each exposure level, was short (< 9 min) and tended to be lower in males than females. For CFC 12 mean t(1/2)beta (beta elimination half-life) ranged from 23 to 43 min and for HFC 227 the mean range was 19-92 min. The values tended to be lower for females than males for HFC 227. For both CFC 12 and HFC 227, mean residence time (MRT) was statistically significantly lower (P < 0.05) in males than females and independent of exposure concentration. For CFC 12, MRT was a mean of 37 and 45 min for males and females, respectively, and for HFC 227 MRT was a mean of 36 and 42 min, respectively...
PMID:11029265 Emmen HH et al; Regul Toxicol Pharmacol 32 (1): 22-35 (2000)
The biotransformation of the aerosol propellant 1,1,1,2,3,3,3-heptafluoropropane (HFA-227) was investigated in rats in vivo and in rat and human liver microsomes. In the urine of rats exposed to 5000 ppm HFA-227 for 6 hr, very small amounts of hexafluoroacetone trihydrate were identified as an HFA-227 metabolite by 19F-NMR. Fluoride concentrations in the urine samples (0-48 hr after the end of the exposure) from exposed animals were not significantly different from those found in samples from nonexposed rats. In rat and human liver microsomes, fluoride and hexafluoroacetone trihydrate formation from HFA-227 was detected in very low levels only in liver microsomes from pyridine-treated rats and in two of eight human liver microsome samples, which exhibited the highest cytochrome P4502E1 activities. Because some aldehydes may covalently bind to proteins and the formation of fluorinated protein adducts has been implicated in immune-mediated hepatitis induced by halothane, the binding of hexafluoroacetone trihydrate to proteins was also investigated. Hexafluoroacetone trihydrate also gave only a very small resonance in fluorine NMR experiments when binding to human serum albumin was studied in comparison with the acylating agent S-ethyltrifluoroacetate. Moreover, no fluorine-containing products were formed by the reaction of hexafluoroacetone trihydrate with N alpha-acetyl-L-lysine, and hexafluoroacetone trihydrate was not metabolized to fluorine-containing metabolites or inorganic fluoride in rats. Comparative studies in human liver microsomes demonstrated that a halothane metabolite may covalently bind to proteins; in contrast, metabolism and covalent binding of HFA-227 could not be demonstrated. In summary, these data indicate that HFA-227 is biotransformed at very low rates to hexafluoroacetone trihydrate but irreversible binding of hexafluoroacetone trihydrate cannot be demonstrated, even with the application of very sensitive methods, and is considered unlikely, based on the combination of the results obtained.
PMID:8869827 Koster U et al; Drug Metab Dispos 24 (8): 906-10 (1996)
HFA 227 showed a biphasic elimination from the body after exposure (mean T1/2: approx. 6.5 minutes and 44.5 minutes).
Solvay; Solkane 227 Pharma and Solkane 134a Pharma: HFA Propellants for Medical Use, p.29. Available from, as of March 3, 2016: https://www.solvay.us/en/
ABOUT THIS PAGE
57
PharmaCompass offers a list of 1,1,1,2,3,3,3-Heptafluoropropane API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right 1,1,1,2,3,3,3-Heptafluoropropane manufacturer or 1,1,1,2,3,3,3-Heptafluoropropane supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred 1,1,1,2,3,3,3-Heptafluoropropane manufacturer or 1,1,1,2,3,3,3-Heptafluoropropane supplier.
A 1,1,1,2,3,3,3-Heptafluoropropane manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of 1,1,1,2,3,3,3-Heptafluoropropane, including repackagers and relabelers. The FDA regulates 1,1,1,2,3,3,3-Heptafluoropropane manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. 1,1,1,2,3,3,3-Heptafluoropropane API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
A 1,1,1,2,3,3,3-Heptafluoropropane supplier is an individual or a company that provides 1,1,1,2,3,3,3-Heptafluoropropane active pharmaceutical ingredient (API) or 1,1,1,2,3,3,3-Heptafluoropropane finished formulations upon request. The 1,1,1,2,3,3,3-Heptafluoropropane suppliers may include 1,1,1,2,3,3,3-Heptafluoropropane API manufacturers, exporters, distributors and traders.
click here to find a list of 1,1,1,2,3,3,3-Heptafluoropropane suppliers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PharmaCompass.
A 1,1,1,2,3,3,3-Heptafluoropropane DMF (Drug Master File) is a document detailing the whole manufacturing process of 1,1,1,2,3,3,3-Heptafluoropropane active pharmaceutical ingredient (API) in detail. Different forms of 1,1,1,2,3,3,3-Heptafluoropropane DMFs exist exist since differing nations have different regulations, such as 1,1,1,2,3,3,3-Heptafluoropropane USDMF, ASMF (EDMF), JDMF, CDMF, etc.
A 1,1,1,2,3,3,3-Heptafluoropropane DMF submitted to regulatory agencies in the US is known as a USDMF. 1,1,1,2,3,3,3-Heptafluoropropane USDMF includes data on 1,1,1,2,3,3,3-Heptafluoropropane's chemical properties, information on the facilities and procedures used, and details about packaging and storage. The 1,1,1,2,3,3,3-Heptafluoropropane USDMF is kept confidential to protect the manufacturer’s intellectual property.
click here to find a list of 1,1,1,2,3,3,3-Heptafluoropropane suppliers with USDMF on PharmaCompass.
1,1,1,2,3,3,3-Heptafluoropropane Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of 1,1,1,2,3,3,3-Heptafluoropropane GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right 1,1,1,2,3,3,3-Heptafluoropropane GMP manufacturer or 1,1,1,2,3,3,3-Heptafluoropropane GMP API supplier for your needs.
A 1,1,1,2,3,3,3-Heptafluoropropane CoA (Certificate of Analysis) is a formal document that attests to 1,1,1,2,3,3,3-Heptafluoropropane's compliance with 1,1,1,2,3,3,3-Heptafluoropropane specifications and serves as a tool for batch-level quality control.
1,1,1,2,3,3,3-Heptafluoropropane CoA mostly includes findings from lab analyses of a specific batch. For each 1,1,1,2,3,3,3-Heptafluoropropane CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
1,1,1,2,3,3,3-Heptafluoropropane may be tested according to a variety of international standards, such as European Pharmacopoeia (1,1,1,2,3,3,3-Heptafluoropropane EP), 1,1,1,2,3,3,3-Heptafluoropropane JP (Japanese Pharmacopeia) and the US Pharmacopoeia (1,1,1,2,3,3,3-Heptafluoropropane USP).

