
Synopsis
Synopsis
0
CEP/COS
0
JDMF
0
EU WC
0
KDMF
0
NDC API
0
VMF
0
Listed Suppliers
0
EDQM
0
USP
0
JP
0
Others
0
FDA Orange Book
0
Europe
0
Australia
0
South Africa
DRUG PRODUCT COMPOSITIONS
0
US Patents
0
US Exclusivities
0
Health Canada Patents
US Medicaid
NA
Annual Reports
NA
Regulatory FDF Prices
NA
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
0
News #PharmaBuzz
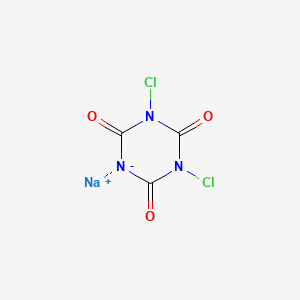

1. 1,3-dichloro-1,3,5-triazine-2,4,6(1h,3h,5h)-trione
2. Actichlor
3. Chlorcyanurate
4. Chlordesine
5. Chlordezine
6. Dichloroisocyanuric Acid
7. Dikon
8. Dikonit
9. Neoaquasept
10. Potassium Dichloro-s-triazinetrione
11. Presept
12. Sodium Dichloro-s-triazine-2,4,6(1h,3h,5h)-trione
13. Troclosene
14. Troclosene, Potassium Salt
15. Troclosene, Sodium Salt
16. Troclosene, Sodium Salt, Dihydrate
1. 2893-78-9
2. Dichloroisocyanuric Acid Sodium Salt
3. Troclosene Sodium
4. Sodium 3,5-dichloro-2,4,6-trioxo-1,3,5-triazinan-1-ide
5. Sodium Dichloro-s-triazinetrione
6. Dichloroisocyanuric Acid, Sodium Salt
7. 1260366-40-2
8. Sdic
9. Sodium Dichlorocyanurate
10. Sodium Troclosene
11. Ncgc00164091-01
12. Dsstox_cid_4994
13. Dsstox_rid_77616
14. Dsstox_gsid_24994
15. Actisept
16. Clearon
17. 07m9u9u0lk
18. Neochlor 55
19. Cas-2893-78-9
20. 1,3,5-triazine-2,4,6(1h,3h,5h)-trione, 1,3-dichloro-, Sodium Salt
21. Dichloro-s-triazinetrione Sodium Salt
22. Nadcc
23. Dichlorosan
24. 1,3,5-triazine-2,4,6(1h,3h,5h)-trione, 1,3-dichloro-, Sodium Salt (1:1)
25. Dichloroisocyanuric Acid Sodium
26. Schembl2485330
27. 3,5-dichloro-2-hydroxy-4,6-s-triazinedione Sodium Salt
28. Chembl3182790
29. Dtxsid3024994
30. Troclosene Sodium [hsdb]
31. Sodium Dichloroisocyanurate Powder
32. Sodium Dichloroisocyanurate, 96%
33. Troclosene Sodium [mart.]
34. Troclosene Sodium [who-dd]
35. Tox21_112083
36. Tox21_201601
37. Tox21_303262
38. Mfcd00006036
39. Akos024363213
40. Ncgc00257133-01
41. Ncgc00259150-01
42. Sodium Dichloroisocyanurate [jan]
43. Sodium Dichloroisocyanurate(sdic Or Nadcc)
44. D1003
45. Ft-0624714
46. J-017330
47. F0001-0906
48. Sodium3,5-dichloro-2,4,6-trioxo-1,3,5-triazinan-1-ide
49. Sodium;1,3-dichloro-1,3-diaza-5-azanidacyclohexane-2,4,6-trione
| Molecular Weight | 219.94 g/mol |
|---|---|
| Molecular Formula | C3Cl2N3NaO3 |
| Hydrogen Bond Donor Count | 0 |
| Hydrogen Bond Acceptor Count | 4 |
| Rotatable Bond Count | 0 |
| Exact Mass | 218.9214405 g/mol |
| Monoisotopic Mass | 218.9214405 g/mol |
| Topological Polar Surface Area | 58.7 Ų |
| Heavy Atom Count | 12 |
| Formal Charge | 0 |
| Complexity | 225 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 2 |
The microbiologic effectiveness of sodium dichloroisocyanurate (NaDCC) tablets used on a routine basis at the household level by a vulnerable population /was assessed/. In a 4-mo trial in Dhaka, Bangladesh, one half of the 100 participating households received NaDCC tablets and instructions on how to use the same; the other one half received a placebo and the same instructions. Monthly samples of stored drinking water from intervention households were significantly lower in thermotolerant coliforms (TTCs) than those of control households (geometric mean, 2.8 [95% CI: 2.2, 3.6] versus 604.1 [95% CI: 463.2, 787.9]; P < 0.0001). While 61.7% (116/188) of samples from the intervention households met World Health Organization (WHO) guidelines for 0 TTCs in drinking water, none of the 191 samples from control households met such a benchmark. Residual free chlorine in water samples suggested that householders consistently used the intervention, but 11.7% of samples exceeded the WHO guideline value of 5.0 mg/L, underscoring the need to ensure that tablet dose and vessel size are compatible.
PMID:17255252 Clasen T et al; Am J Trop Med Hyg 76 (1): 187-92 (2007).
3. 3 = moderately toxic: probable oral lethal dose (human) 0.5-5 g/kg, between 1 oz and 1 pint for 70 kg person (150 lb). /Trichloroisocyanuric acid/
Gosselin, R.E., H.C. Hodge, R.P. Smith, and M.N. Gleason. Clinical Toxicology of Commercial Products. 4th ed. Baltimore: Williams and Wilkins, 1976., p. II-77
Antifungal Agents
Substances that destroy fungi by suppressing their ability to grow or reproduce. They differ from FUNGICIDES, INDUSTRIAL because they defend against fungi present in human or animal tissues. (See all compounds classified as Antifungal Agents.)
Antiviral Agents
Agents used in the prophylaxis or therapy of VIRUS DISEASES. Some of the ways they may act include preventing viral replication by inhibiting viral DNA polymerase; binding to specific cell-surface receptors and inhibiting viral penetration or uncoating; inhibiting viral protein synthesis; or blocking late stages of virus assembly. (See all compounds classified as Antiviral Agents.)
Disinfectants
Substances used on inanimate objects that destroy harmful microorganisms or inhibit their activity. Disinfectants are classed as complete, destroying SPORES as well as vegetative forms of microorganisms, or incomplete, destroying only vegetative forms of the organisms. They are distinguished from ANTISEPTICS, which are local anti-infective agents used on humans and other animals. (From Hawley's Condensed Chemical Dictionary, 11th ed) (See all compounds classified as Disinfectants.)
Metabolism studies in both the rat and dog, following administration of (14)C-sodium isocyanurate by the oral and iv routes, demonstrated rapid absorption, distribution, and excretion of unmetabolized isocyanurate. ... At the 5 mg/kg dose, excretion was largely via the urine with about 5% in the feces. At the 500 mg/kg oral dose, 55-70% (rats) or 27-86% (dogs) was excreted in the feces and the remainder in the urine. /Sodium isocyanurate/
USEPA/Office of Pesticide Programs; Reregistration Eligibility Decision Document - Chlorinated Isocyanurates p.17 H-750BW (September, 1992). Available from, as of September 24, 2009: https://www.epa.gov/oppsrrd1/REDs/old_reds/chlorinated_isocyanurates.pdf
Both s-triazine-2,4,6(1H,3H,5H)-trione, 1,3,5-trichloro- and s-triazine-2,4,6(1H,3H,5H)-trione, 1,3-dichloro-, sodium salt are unstable in the body (particularly the stomach) because the available chlorine is rapidly reduced. Cyanuric acid (or its monosodium salt) is the degradation product from both products.
EPA/Office of Pollution Prevention and Toxics; High Production Volume Information System (HPVIS) on 1,3,5-Triazine-2,4,6(1H,3H,5H)-trione, 1,3,5-trichloro- (87-90-1). Available from, as of September 24, 2009: https://www.epa.gov/hpvis/index.html
Administration of (14C)sodium isocyanurate by the oral and iv routes demonstrated rapid absorption, distribution, and excretion of unmetabolized isocyanurate. The elimination half-life in rats was 32-43 min following 5 mg/kg iv or oral administration. The half-life was 122-148 min after oral dosing at 500 mg/kg.
USEPA/Office of Pesticide Programs; Reregistration Eligibility Decision Document - Chlorinated Isocyanurates p.17 H-750BW (September, 1992). Available from, as of September 24, 2009: https://www.epa.gov/oppsrrd1/REDs/old_reds/chlorinated_isocyanurates.pdf

API/FDF Prices: Book a Demo to explore the features and consider upgrading later
API Imports and Exports
| Importing Country | Total Quantity (KGS) |
Average Price (USD/KGS) |
Number of Transactions |
|---|
Upgrade, download data, analyse, strategize, subscribe with us
Market Place

ABOUT THIS PAGE
28
PharmaCompass offers a list of Sodium Dichloroisocyanurate API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, Price,and more, enabling you to easily find the right Sodium Dichloroisocyanurate manufacturer or Sodium Dichloroisocyanurate supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Sodium Dichloroisocyanurate manufacturer or Sodium Dichloroisocyanurate supplier.
PharmaCompass also assists you with knowing the Sodium Dichloroisocyanurate API Price utilized in the formulation of products. Sodium Dichloroisocyanurate API Price is not always fixed or binding as the Sodium Dichloroisocyanurate Price is obtained through a variety of data sources. The Sodium Dichloroisocyanurate Price can also vary due to multiple factors, including market conditions, regulatory modifications, or negotiated pricing deals.
A 07M9U9U0LK manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of 07M9U9U0LK, including repackagers and relabelers. The FDA regulates 07M9U9U0LK manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. 07M9U9U0LK API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
A 07M9U9U0LK supplier is an individual or a company that provides 07M9U9U0LK active pharmaceutical ingredient (API) or 07M9U9U0LK finished formulations upon request. The 07M9U9U0LK suppliers may include 07M9U9U0LK API manufacturers, exporters, distributors and traders.
click here to find a list of 07M9U9U0LK suppliers with USDMF, JDMF, KDMF, CEP, GMP, COA and API Price related information on PharmaCompass.
A 07M9U9U0LK DMF (Drug Master File) is a document detailing the whole manufacturing process of 07M9U9U0LK active pharmaceutical ingredient (API) in detail. Different forms of 07M9U9U0LK DMFs exist exist since differing nations have different regulations, such as 07M9U9U0LK USDMF, ASMF (EDMF), JDMF, CDMF, etc.
A 07M9U9U0LK DMF submitted to regulatory agencies in the US is known as a USDMF. 07M9U9U0LK USDMF includes data on 07M9U9U0LK's chemical properties, information on the facilities and procedures used, and details about packaging and storage. The 07M9U9U0LK USDMF is kept confidential to protect the manufacturer’s intellectual property.
click here to find a list of 07M9U9U0LK suppliers with USDMF on PharmaCompass.
07M9U9U0LK Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of 07M9U9U0LK GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, API price, and more, enabling you to easily find the right 07M9U9U0LK GMP manufacturer or 07M9U9U0LK GMP API supplier for your needs.
A 07M9U9U0LK CoA (Certificate of Analysis) is a formal document that attests to 07M9U9U0LK's compliance with 07M9U9U0LK specifications and serves as a tool for batch-level quality control.
07M9U9U0LK CoA mostly includes findings from lab analyses of a specific batch. For each 07M9U9U0LK CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
07M9U9U0LK may be tested according to a variety of international standards, such as European Pharmacopoeia (07M9U9U0LK EP), 07M9U9U0LK JP (Japanese Pharmacopeia) and the US Pharmacopoeia (07M9U9U0LK USP).

