
Synopsis
Synopsis
0
USDMF
0
CEP/COS
0
JDMF
0
EU WC
0
KDMF
0
NDC API
0
VMF
0
FDA Orange Book

0
Europe

0
Canada

0
Australia

0
Listed Dossiers
DRUG PRODUCT COMPOSITIONS
0
US Patents
0
US Exclusivities
0
Health Canada Patents
US Medicaid
NA
Annual Reports
NA
Regulatory FDF Prices
NA
0
FDF
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
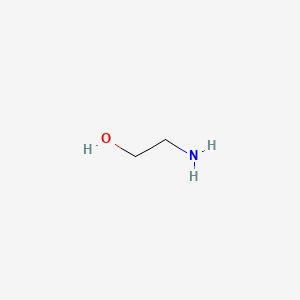

1. 2 Aminoethanol
2. 2-aminoethanol
3. Colamine
4. Monoethanolamine
1. 2-aminoethanol
2. Monoethanolamine
3. 141-43-5
4. Colamine
5. Aminoethanol
6. 2-hydroxyethylamine
7. Glycinol
8. Olamine
9. 2-aminoethan-1-ol
10. 2-amino-1-ethanol
11. Ethanol, 2-amino-
12. Ethylolamine
13. Beta-hydroxyethylamine
14. 2-hydroxyethanamine
15. 1-amino-2-hydroxyethane
16. 2-aminoethyl Alcohol
17. Beta-aminoethyl Alcohol
18. Aethanolamin
19. 2-ethanolamine
20. Thiofaco M-50
21. Beta-aminoethanol
22. 2-amino-ethanol
23. Mea (alcohol)
24. Beta-ethanolamine
25. Kolamin [czech]
26. Usaf Ek-1597
27. Kolamin
28. Monoaethanolamin
29. Aethanolamin [german]
30. Caswell No. 426
31. Etanolamina [italian]
32. 2-aminoaethanol
33. 2-amino Ethanol
34. Monoaethanolamin [german]
35. 2-aminoaethanol [german]
36. 2-aminoetanolo [italian]
37. Glycinol (monoethanolamine)
38. Un2491
39. Ccris 6260
40. Hsdb 531
41. Ai3-24219
42. Olamine [inn]
43. Epa Pesticide Chemical Code 011601
44. Acid Orange 86
45. Ethanol Amine
46. Chebi:16000
47. Mfcd00008183
48. C.i. Acid Orange 108
49. Chembl104943
50. 12220-07-4
51. Ethanol, 2-amino-, Homopolymer
52. Envision Conditioner Pdd 9020
53. 5kv86114pt
54. Ethanol-amine
55. Un 2491
56. Monoethanolamine (nf)
57. Monoethanolamine [nf]
58. Ncgc00090789-02
59. 12220-09-6
60. 2-aminoethanol (ethanolamine)
61. 2-aminoethanol Reagent Acs Grade
62. Etanolamina
63. Ethanolamine Or Ethanolamine Solutions [un2491] [corrosive]
64. 2-aminoetanolo
65. Monoethanol Amine
66. 26778-51-8
67. Einecs 205-483-3
68. Ethanolamin
69. Ethanolarnine
70. Hydoxyethylamine
71. B-aminoethanol
72. B-ethanolamine
73. Unii-5kv86114pt
74. H-glycinol
75. 2 -aminoethanol
76. B-hydroxyethylamine
77. Ethanol, 2-amino
78. 2-hydroxylethylamine
79. H-gly-ol
80. .beta.-aminoethanol
81. .beta.-ethanolamine
82. B-aminoethyl Alcohol
83. Ethanolamine Solution
84. 2-hydroxy-ethylamine
85. 2-hydroxyethyl Amine
86. 2-hydroxyethyl-amine
87. 2-amino-ethan-1-ol
88. 2-hydroxy- Ethylamine
89. Ethanolamine Or Ethanolamine Solutions [un2491] [corrosive]
90. 2-hydroxy-1-ethylamine
91. .beta.-hydroxyethylamine
92. Ethanolamine, >=98%
93. Ethanolamine, >=99%
94. Ethanolamine [mi]
95. Nh2ch2ch2oh
96. .beta.-aminoethyl Alcohol
97. Dsstox_cid_2000
98. Ethanolamine, Ar, 99%
99. Bmse000276
100. Epitope Id:120354
101. Nh2c2h4oh
102. Ec 205-483-3
103. Ethanolamine [hsdb]
104. Ethanolamine [inci]
105. 2-aminoethanol, Redistilled
106. 2-aminoethanol,hydrochloride
107. Dsstox_rid_76452
108. Mea-lci; Mea 90
109. Dsstox_gsid_22000
110. Ethanolamine: 2-aminoethanol
111. Monoethanolamine [ii]
112. Bidd:er0367
113. Ethanolamine [who-dd]
114. Ethanolamine, Lr, >=99%
115. Bdbm7973
116. Dtxsid6022000
117. Ethanolamine, Analytical Standard
118. Monoethanolamine [usp-rs]
119. Ethanol, 2-amino- (8ci,9ci)
120. Ethanolamine [ep Monograph]
121. Str00417
122. Zinc8214617
123. Tox21_400020
124. C0594
125. Stl199164
126. Ethanolamine Or Ethanolamine Solutions
127. Ethanolamine, For Synthesis, 99.0%
128. Akos000245055
129. Db03994
130. 1h-indole-5-carbothioicacidamide
131. Ethanolamine, Acs Reagent, >=99.0%
132. Ethanolamine, Reagentplus(r), >=99%
133. Ethanolamine, Usp, 98.0-100.5%
134. Beta-aminoethyl Alcohol Beta-ethanolamine
135. Ncgc00090789-01
136. Ncgc00090789-03
137. Bp-21017
138. Bp-24368
139. Bp-31055
140. Cas-141-43-5
141. Trolamine Impurity A [ep Impurity]
142. Ethanolamine, Saj First Grade, >=99.0%
143. Ifosfamide Impurity D [ep Impurity]
144. A0297
145. Ethanolamine, Jis Special Grade, >=99.0%
146. Ft-0777922
147. En300-19392
148. 2-aminoethanol 1000 Microg/ml In Acetonitrile
149. C00189
150. D05074
151. P20022
152. A902308
153. Q410387
154. Ethanolamine, Purified By Redistillation, >=99.5%
155. J-508043
156. 587ce5aa-008a-469c-ab39-82cc4d0ca779
157. Ethanolamine; 2-aminoethanol; 2-aminoethyl Alcohol; Mea
158. F2190-0367
159. Ethanolamine, Puriss. P.a., Acs Reagent, >=99.0% (gc/nt)
160. Ethanolamine, Liquid, Bioreagent, Suitable For Cell Culture, >=98%
161. Trolamine Impurity A, European Pharmacopoeia (ep) Reference Standard
162. Ethanolamine Solution, For Peptide Synthesis, ~74% (t), ~70% In Methanol
163. Monoethanolamine, United States Pharmacopeia (usp) Reference Standard
164. Monoethanolamine, Pharmaceutical Secondary Standard; Certified Reference Material
165. Ethanolamine, Pharmagrade, Usp/nf, Manufactured Under Appropriate Gmp Controls For Pharma Or Biopharmaceutical Production
| Molecular Weight | 61.08 g/mol |
|---|---|
| Molecular Formula | C2H7NO |
| XLogP3 | -1.3 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 2 |
| Rotatable Bond Count | 1 |
| Exact Mass | 61.052763847 g/mol |
| Monoisotopic Mass | 61.052763847 g/mol |
| Topological Polar Surface Area | 46.2 Ų |
| Heavy Atom Count | 4 |
| Formal Charge | 0 |
| Complexity | 10 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
The principal route of exposure is through skin, with some exposure occurring by inhalation of vapor and aerosols. Monoethanolamine (MEA), diethanolamine (DEA), and triethanolamine (TEA) in water penetrate rat skin at the rate of 2.9 x 10(-3), 4.36 x 10(-3) and 18 x 10(-3) cm/hr, respectively. MEA, DEA, and TEA are water-soluble ammonia derivatives, with pHs of 9-11 in water and pHa values of 9.3, 8.8, and 7.7, respectively.
PMID:8956558 Knaak JB et al; Rev Environ Contam Toxicol 149: 1-86 (1997)
The excretion rate in men was found to vary between 4.8 and 22.9 mg/day with a mean of 0.162 mg/kg /body weight/. 11 women were observed to excrete larger amounts, varying between 7.7 and 34.9 mg/day with a mean excretion rate of 0.492 mg/kg/day. The excretion rates in animals were approximately, for cats, 0.47 mg/kg/day; for rats, 1.46 mg/kg/day; and for rabbits, 1.0 mg/kg/day. From 6-47% of monoethanolamine administered to rats can be recovered in the urine.
Bingham, E.; Cohrssen, B.; Powell, C.H.; Patty's Toxicology Volumes 1-9 5th ed. John Wiley & Sons. New York, N.Y. (2001)., p. V4 782
Persistence of low levels of radioactivity in dog whole blood was obtained after admin of (14)C-labeled ethanolamine. Excretion of radioactivity as % of dose in dog urine was 11. After 24 hr total blood radioactivity as % of dose was 1.69.
Rhodes C, Case DE; Xenobiotica 7 (1-2): 112 (1977)
/Ethanolamine/ is a normal urine constituent in man, excreted at a rate of 5-23 mg/day ... 40 percent of an administered dose is deaminated and excreted as urea.
Zenz, C., O.B. Dickerson, E.P. Horvath. Occupational Medicine. 3rd ed. St. Louis, MO., 1994, p. 707
For more Absorption, Distribution and Excretion (Complete) data for 2-AMINOETHANOL (10 total), please visit the HSDB record page.
Ethanolamine can be used as a source of carbon and nitrogen by phylogenetically diverse bacteria. Ethanolamine-ammonia lyase, the enzyme that breaks ethanolamine into acetaldehyde and ammonia, is encoded by the gene tandem eutBC. Despite extensive studies of ethanolamine utilization in Salmonella enterica serovar Typhimurium, much remains to be learned about EutBC structure and catalytic mechanism, about the evolutionary origin of ethanolamine utilization, and about regulatory links between the metabolism of ethanolamine itself and the ethanolamine-ammonia lyase cofactor adenosylcobalamin. We used computational analysis of sequences, structures, genome contexts, and phylogenies of ethanolamine-ammonia lyases to address these questions and to evaluate recent data-mining studies that have suggested an association between bacterial food poisoning and the diol utilization pathways. We found that EutBC evolution included recruitment of a TIM barrel and a Rossmann fold domain and their fusion to N-terminal alpha-helical domains to give EutB and EutC, respectively. This fusion was followed by recruitment and occasional loss of auxiliary ethanolamine utilization genes in Firmicutes and by several horizontal transfers, most notably from the firmicute stem to the Enterobacteriaceae and from Alphaproteobacteria to Actinobacteria. We identified a conserved DNA motif that likely represents the EutR-binding site and is shared by the ethanolamine and cobalamin operons in several enterobacterial species, suggesting a mechanism for coupling the biosyntheses of apoenzyme and cofactor in these species. Finally, we found that the food poisoning phenotype is associated with the structural components of metabolosome more strongly than with ethanolamine utilization genes or with paralogous propanediol utilization genes per se.
PMID:19783625 Full text: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2786565 Tsoy O et al; J Bacteriol 191 (23): 7157-64 (2009)
Forty percent of (15)N-labeled ethanolamine appears as urea within 24 hr when it is given to rabbits, suggesting that it is deaminated. In rat liver homogenates, ethanolamine undergoes demethylation yielding formaldehyde.
Bingham, E.; Cohrssen, B.; Powell, C.H.; Patty's Toxicology Volumes 1-9 5th ed. John Wiley & Sons. New York, N.Y. (2001)., p. V4 782
Ethanolamine is a normal intermediate in the metabolism of some animal species, having a part in the formation of phospholipids and choline.
American Conference of Governmental Industrial Hygienists. Documentation of the TLVs and BEIs with Other World Wide Occupational Exposure Values. 7th Ed. CD-ROM Cincinnati, OH 45240-1634 2013., p. 2
The distribution and metabolism of topical (14)C ethanolamine was studied in vivo, using athymic nude mice, human skin grafted onto athymic nude mice, and in vitro, using excised pig skin. Ethanolamine was the only radioactive phospholipid base detected in the human skin grafts, in the mouse skin, and in the pig skin. Ethanolamine that penetrated human skin grafts or mouse skin was extensively metabolized in the animal. The liver is a major site for metabolism of ethanolamine, containing over 24% of the applied radioactive dose. The kidneys, lungs, brain, and the heart contained 2.53, 0.55, 0.27, and 0.15% of the dose, respectively. Hepatic, human skin graft, and mouse skin proteins were also highly radioactive. Over 18% of the topical radioactive dose oxidized to (14)CO2 and 4.6% was excreted in the urine over 24 hr. Urea, glycine, serine, choline, and uric acid were the urinary metabolites of ethanolamine.
Klain GJ et al; Fundam Appl Toxicol 5 (6 Pt 2): S127-33 (1985)
For more Metabolism/Metabolites (Complete) data for 2-AMINOETHANOL (10 total), please visit the HSDB record page.
The half-life of the persistent low level of radioactivity in the blood /of dogs administered 14C-ethanolamine/ was 19 days.
Snyder, R. (ed.). Ethel Browning's Toxicity and Metabolism of Industrial Solvents. 2nd ed. Volume II: Nitrogen and Phosphorus Solvents. Amsterdam-New York-Oxford: Elsevier, 1990., p. 426
Labeled MEA was administered to dogs. ... After 24 hr ... the half-life was 19 days.
Christian M, ed; J American College of Toxicology 2 (7): 183-226 (1983)

API/FDF Prices: Book a Demo to explore the features and consider upgrading later
API Imports and Exports
| Importing Country | Total Quantity (KGS) |
Average Price (USD/KGS) |
Number of Transactions |
|---|
Upgrade, download data, analyse, strategize, subscribe with us
Market Place

REF. STANDARDS & IMPURITIES

ANALYTICAL

ABOUT THIS PAGE
85
PharmaCompass offers a list of Ethanolamine API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, Price,and more, enabling you to easily find the right Ethanolamine manufacturer or Ethanolamine supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Ethanolamine manufacturer or Ethanolamine supplier.
PharmaCompass also assists you with knowing the Ethanolamine API Price utilized in the formulation of products. Ethanolamine API Price is not always fixed or binding as the Ethanolamine Price is obtained through a variety of data sources. The Ethanolamine Price can also vary due to multiple factors, including market conditions, regulatory modifications, or negotiated pricing deals.
A 07969_FLUKA manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of 07969_FLUKA, including repackagers and relabelers. The FDA regulates 07969_FLUKA manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. 07969_FLUKA API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
click here to find a list of 07969_FLUKA manufacturers with USDMF, JDMF, KDMF, CEP, GMP, COA and API Price related information on PhamaCompass.
A 07969_FLUKA supplier is an individual or a company that provides 07969_FLUKA active pharmaceutical ingredient (API) or 07969_FLUKA finished formulations upon request. The 07969_FLUKA suppliers may include 07969_FLUKA API manufacturers, exporters, distributors and traders.
click here to find a list of 07969_FLUKA suppliers with USDMF, JDMF, KDMF, CEP, GMP, COA and API Price related information on PharmaCompass.
07969_FLUKA Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of 07969_FLUKA GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, API price, and more, enabling you to easily find the right 07969_FLUKA GMP manufacturer or 07969_FLUKA GMP API supplier for your needs.
A 07969_FLUKA CoA (Certificate of Analysis) is a formal document that attests to 07969_FLUKA's compliance with 07969_FLUKA specifications and serves as a tool for batch-level quality control.
07969_FLUKA CoA mostly includes findings from lab analyses of a specific batch. For each 07969_FLUKA CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
07969_FLUKA may be tested according to a variety of international standards, such as European Pharmacopoeia (07969_FLUKA EP), 07969_FLUKA JP (Japanese Pharmacopeia) and the US Pharmacopoeia (07969_FLUKA USP).

