
Synopsis
Synopsis
0
JDMF
0
KDMF
0
VMF
0
Australia
Annual Reports
NA
0
FDF
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
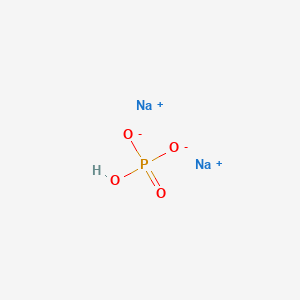

1. Dibasic Sodium Phosphate, Anhydrous
2. Disodium Acid Phosphate
3. Disodium Hydrogen Phosphate Anhydrous
4. Monosodium Dihydrogen Phosphate
5. Neutral Sodium Hydrogen Phosphate
6. Phosphoric Acid, Disodium Salt
7. Phosphoric Acid, Disodium Salt, 32p-labeled
8. Phosphoric Acid, Disodium Salt, Anhydrous
9. Phosphoric Acid, Disodium Salt, Dodecahydrate
10. Phosphoric Acid, Disodium Salt, Heptahydrate
11. Phosphoric Acid, Monosodium Salt
12. Phosphoric Acid, Monosodium Salt, Anhydrous
13. Phosphoric Acid, Sodium (2:3) Salt
14. Phosphoric Acid, Sodium Salt
15. Phosphoric Acid, Trisodium Salt
16. Phosphoric Acid, Trisodium Salt , 32p-labeled
17. Phosphoric Acid, Trisodium Salt , Dodecahydrate
18. Sodium Biphosphate
19. Sodium Dihydrogen Orthophosphate
20. Sodium Dihydrogen Phosphate
21. Sodium Hydrophosphate
22. Sodium Phosphate
23. Sodium Phosphate Monobasic Anhydrous
24. Sodium Phosphate, Dibasic
25. Sodium Phosphate, Dibasic (anhydrous)
26. Sodium Phosphate, Disodium Salt
27. Sodium Phosphate, Monobasic
28. Sodium Phosphate, Monobasic Anhydrous
29. Sodium Phosphate, Tribasic
30. Sodium Phosphate, Tribasic, Dodecahydrate
31. Trisodium Phosphate
32. Trisodium Phosphate Dodecahydrate
1. 7558-79-4
2. Disodium Phosphate
3. Sodium Phosphate Dibasic
4. Disodium Hydrogenorthophosphate
5. Dibasic Sodium Phosphate
6. Acetest
7. Sodium Phosphate, Dibasic
8. Disodium Acid Phosphate
9. Disodium Hydrogenphosphate
10. Soda Phosphate
11. Phosphoric Acid, Disodium Salt
12. Exsiccated Sodium Phosphate
13. Disodium Orthophosphate
14. Sodium Hydrogenphosphate
15. Sodium Hydrogen Phosphate
16. Sodium Monohydrogen Phosphate
17. Fema No. 2398
18. Secondary Sodium Phosphate
19. Disodium;hydrogen Phosphate
20. Disodium Acid Orthophosphate
21. Sec-sodium Phosphate
22. Disodium Phosphate, Anhydrous
23. Disodium Monohydrogen Phosphate
24. Disodium Hydrogen Phosphate, Anhydrous
25. Hydrogen Disodium Phosphate
26. Sodium Phosphate, Dibasic, Anhydrous
27. Sodium Phosphate, Dibasic (anhydrous)
28. Sodium Phosphate, Dibasic Anhydrous
29. Sodium Phosphate Dibasic Anhydrous
30. Na2hpo4
31. Phosphoric Acid, Sodium Salt (1:2)
32. Disodium Hydrogen Monophosphate
33. 22ado53m6f
34. Disodium Hydrogen Orthophosphate
35. Ins No.339(ii)
36. Dsp
37. Chebi:34683
38. Ins-339(ii)
39. Dibasic Sodium Phosphate Anhydrous
40. Dibasic Sodium Phosphate, Anhydrous
41. E-339(ii)
42. Mfcd00003496
43. Phosphoric Acid, Disodium Salt, Anhydrous
44. Phosphate Of Soda
45. Monohydrogen Sodium Phosphate
46. Caswell No. 778
47. Disodium Hydrophosphate
48. Fema Number 2398
49. Fleet Enema
50. Natriumphosphat [german]
51. Sodium Phosphate (nahpo4)
52. Sodium Phosphate, Exsiccated
53. Ccris 5931
54. Hsdb 376
55. Sodium Orthophosphate, Secondary
56. Sodium Acid Phosphate, Anhydrous
57. Einecs 231-448-7
58. Potassium Phosphate Monobasic/sodium Phosphate Dibasic
59. Epa Pesticide Chemical Code 076403
60. Sodium Monohydrogen Phosphate (2:1:1)
61. Unii-22ado53m6f
62. Disodium Phospahte
63. Sodiumphosphatedibasic
64. Disodiumhydrogenphosphate
65. Sodium Hydrogen-phosphate
66. Disodium Hydrogen-phosphate
67. Buffer Salt, Ph 6.87
68. Buffer Salt, Ph 7.42
69. Ec 231-448-7
70. Sorensen's Sodium Phosphate
71. Di-sodium Hydrogen Phosphate
72. Phosphoric Acid Disodium Salt
73. Sodium Hydrogen Orthophosphate
74. Sodium Phosphate, Unspecified
75. Sodium Phosphate [fhfi]
76. Dtxsid1026039
77. Disodium Phosphate [hsdb]
78. Disodium Phosphate [inci]
79. Sodium Phosphate,dibasic,anhydrous
80. Bcp13559
81. Sodium Hydrogen Phosphate, Anhydrous
82. Sodium Phosphate Dibasic, Acs Grade
83. Disodium Hydrogen Phosphate Submicron
84. Disodium Phosphate (fragrance Grade)
85. Akos015902440
86. Akos015950661
87. Disodium Phosphate (industrial Grade)
88. Ccg-266159
89. Db14502
90. Sodium Phosphate, Dibasic [mi]
91. Disodium Phosphate [ep Monograph]
92. Sodium Phosphate Dibasic [who-dd]
93. Sodium Phosphate Dibasic, Biochemical Grade
94. B7293
95. Ft-0625321
96. Sodium Phosphate Dibasic, Trace Metals Grade
97. A937532
98. Q418448
99. Sodium Phosphate, Dibasic, Anhydrous [ii]
100. Sodium Phosphate,dibasic,anhydrous [vandf]
101. Phosphoric-32p Acid,disodium Salt (8ci,9ci)
102. Disodium Hydrogen Orthophosphate;sodium Hydrogen Phosphate
103. Sodium Phosphate, Dibasic Anhydrous [orange Book]
104. Sodium Phosphate, Dibasic, Anhydrous [orange Book]
105. Visicol Component Sodium Phosphate, Dibasic Anhydrous
106. Osmoprep Component Sodium Phosphate, Dibasic Anhydrous
107. Sodium Phosphate, Dibasic Anhydrous Component Of Visicol
108. Sodium Phosphate, Dibasic Anhydrous Component Of Osmoprep
| Molecular Weight | 141.959 g/mol |
|---|---|
| Molecular Formula | HNa2O4P |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 4 |
| Rotatable Bond Count | 0 |
| Exact Mass | 141.94078407 g/mol |
| Monoisotopic Mass | 141.94078407 g/mol |
| Topological Polar Surface Area | 83.4 Ų |
| Heavy Atom Count | 7 |
| Formal Charge | 0 |
| Complexity | 46.5 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 3 |
Cathartics
National Library of Medicine's Medical Subject Headings online file (MeSH, 1999)
Sodium Phosphates Injection, USP, ... is indicated as a source of phosphorus, for addition to large volume intravenous fluids, to prevent or correct hypophosphatemia in patients with restricted or no oral intake. It is also useful as an additive for preparing specific parenteral fluid formulas when the needs of the patient cannot be met by standard electrolyte or nutrient solutions. /Included in US product label/
US Natl Inst Health; DailyMed. Current Medication Information for Sodium Phosphates (Sodium Phosphate) Injection (June 2006). Available from, as of March 20, 2009: https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?id=1758
Visicol tablets are indicated for cleansing of the colon as a preparation for colonoscopy in adults 18 years of age or older. /Included in US product label/
US Natl Inst Health; DailyMed. Current Medication Information for VISICOL (sodium phosphate, monobasic, monohydrate and sodium phosphate, dibasic anhydrous) tablet (November 2008). Available from, as of March 20, 2009: https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?id=1758
Although sodium and/or potassium phosphates have been used in the treatment of hypercalcemia, USP medical advisory panels do not recommend this use since these medications have been replaced by safer and more effective agents. /Phosphates/
Thomson/Micromedex. Drug Information for the Health Care Professional. Volume 1, Greenwood Village, CO. 2006., p. 2453
For more Therapeutic Uses (Complete) data for DISODIUM HYDROGEN PHOSPHATE (14 total), please visit the HSDB record page.
/BOXED WARNING/ There have been rare, but serious reports of acute phosphate nephropathy in patients who received oral sodium phosphate products for colon cleansing prior to colonoscopy. Some cases have resulted in permanent impairment of renal function and some patients required long-term dialysis. While some cases have occurred in patients without identifiable risk factors, patients at increased risk of acute phosphate nephropathy may include those with increased age, hypovolemia, increased bowel transit time (such as bowel obstruction), active colitis, or baseline kidney disease, and those using medicines that affect renal perfusion or function (such as diuretics, angiotensin converting enzyme (ACE) inhibitors, angiotensin receptor blockers (ARBs), and possibly nonsteroidal anti-inflammatory drugs (NSAIDs)).
US Natl Inst Health; DailyMed. Current Medication Information for Osmoprep (sodium phosphate, monobasic, monohydrate, sodium phosphate, dibasic anhydrous) tablet (Updated: March 2013). Available from, as of April 24, 2015: https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=b46c0b17-c49b-4791-913f-3e6e1fdbe88e
FDA has become aware of reports of acute phosphate nephropathy, a type of acute kidney injury, associated with the use of oral sodium phosphate products (OSP) for bowel cleansing prior to colonoscopy or other procedures. These products include the prescription products, Visicol and OsmoPrep, and OSPs available over-the-counter without a prescription as laxatives (e.g., Fleet Phospho-soda). In some cases when used for bowel cleansing, these serious adverse events have occurred in patients without identifiable factors that would put them at risk for developing acute kidney injury. We cannot rule out, however, that some of these patients were dehydrated prior to ingestion of OSPs or they did not drink sufficient fluids after ingesting OSP. Acute phosphate nephropathy is a form of acute kidney injury that is associated with deposits of calcium-phosphate crystals in the renal tubules that may result in permanent renal function impairment. Acute phosphate nephropathy is a rare, serious adverse event that has been associated with the use of OSPs. The occurrence of these events was previously described in an Information for Healthcare Professionals sheet and an FDA Science Paper issued in May 2006. Additional cases of acute phosphate nephropathy have been reported to FDA and described in the literature since these were issued. Individuals who appear to have an increased risk of acute phosphate nephropathy following the use of OSPs include persons: who are over age 55; who are hypovolemic or have decreased intravascular volume; who have baseline kidney disease, bowel obstruction, or active colitis; and who are using medications that affect renal perfusion or function (such as diuretics, angiotensin converting enzyme [ACE] inhibitors, angiotensin receptor blockers [ARBs], and possibly nonsteroidal anti-inflammatory drugs [NSAIDs]). As a result of new safety information received, FDA is requiring the manufacturer of Visicol and OsmoPrep, the two OSPs available by prescription only, to add a Boxed Warning to the labeling for these products. FDA is also requiring that the manufacturer develop and implement a risk evaluation and mitigation strategy (REMS), which will include a Medication Guide, to ensure that the benefits of these products outweigh the risk of acute phosphate nephropathy, and to conduct a postmarketing clinical trial to further assess the risk of acute kidney injury with use of these products.
FDA/CDER; FDA Alert: Oral Sodium Phosphate (OSP) Products for Bowel Cleansing (marketed as Visicol and OsmoPrep, and oral sodium phosphate products available without a prescription) (12/11/2008). Available from: https://www.fda.gov/cder/drug/infopage/OSP_solution/default.htm as of March 20,2009.
This phosphate should not be confused with tribasic sodium phosphate which is very alkaline and has caustic action.
Osol, A. (ed.). Remington's Pharmaceutical Sciences. 16th ed. Easton, Pennsylvania: Mack Publishing Co., 1980., p. 745
Oral administration is safer, but careful monitoring of serum electrolyte levels and renal function is necessary. Nausea, vomiting, and diarrhea may occur and may be dose dependent. Concomitant use of antacids containing aluminum and/or magnesium should be avoided, because they may bind phosphate and prevent it absorption (calcium antacids also may bind phosphate, and it is assumed that these agents are not given to hypercalcemic patients). /Monobasic or dibasic sodium or potassium phosphate/
American Medical Association, Department of Drugs. Drug Evaluations. 6th ed. Chicago, Ill: American Medical Association, 1986., p. 897
For more Drug Warnings (Complete) data for DISODIUM HYDROGEN PHOSPHATE (57 total), please visit the HSDB record page.
The estimated fatal dose of sodium phosphates is 50 g.
Dreisbach, R.H. Handbook of Poisoning. 12th ed. Norwalk, CT: Appleton and Lange, 1987., p. 212
Used to treat constipation or to clean the bowel before a colonoscopy.
Sodium phosphate inceases fecal water content to increase mobility through the large intestine.
Absorption
Tmax for phosphate absorption with orally administered liquid sodium phosphate is 1-3h.
... Phosphates (dibasic and monobasic sodium phosphate) are slowly and incompletely absorbed. /Dibasic and Monobasic Sodium phosphate/
Gosselin, R.E., H.C. Hodge, R.P. Smith, and M.N. Gleason. Clinical Toxicology of Commercial Products. 4th ed. Baltimore: Williams and Wilkins, 1976., p. II-83
Net phosphorus absorption may occur in the small intestine in some species but is primarily a function of the colon in horses. /Phosphorus/
Booth, N.H., L.E. McDonald (eds.). Veterinary Pharmacology and Therapeutics. 5th ed. Ames, Iowa: Iowa State University Press, 1982., p. 640
Elimination: Renal (90%) and fecal (10%). /Phosphates/
Thomson/Micromedex. Drug Information for the Health Care Professional. Volume 1, Greenwood Village, CO. 2006., p. 2453
Ingested phosphates are absorbed from the gastrointestinal tract. However, the presence of large amounts of calcium or aluminum may lead to formation of insoluble phosphate and reduce the net absorption. Vitamin D stimulates phosphate absorption. /Phosphates/
Thomson/Micromedex. Drug Information for the Health Care Professional. Volume 1, Greenwood Village, CO. 2006., p. 2453
For more Absorption, Distribution and Excretion (Complete) data for DISODIUM HYDROGEN PHOSPHATE (9 total), please visit the HSDB record page.
Sodium phosphate is thought to work by increasing the amount of solute present in the intestinal lumen thereby creating an osmotic gradient which draws water into the lumen.
At the renal distal tubule, the secretion of hydrogen by the tubular cell in exchange for sodium in the tubular urine converts dibasic phosphate salts to monobasic phosphate salts. Therefore, large amounts of acid can be excreted without lowering the pH of the urine to a degree that would block hydrogen transport by a high concentration gradient between the tubular cell and luminal fluid. /Phosphates/
Thomson/Micromedex. Drug Information for the Health Care Professional. Volume 1, Greenwood Village, CO. 2006., p. 2453
DRUG PRODUCT COMPOSITIONS

Related Excipient Companies

Excipients by Applications
Global Sales Information

Market Place

Patents & EXCLUSIVITIES
REF. STANDARDS & IMPURITIES

ABOUT THIS PAGE
46
PharmaCompass offers a list of Disodium Hydrogen Phosphate API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, Price,and more, enabling you to easily find the right Disodium Hydrogen Phosphate manufacturer or Disodium Hydrogen Phosphate supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Disodium Hydrogen Phosphate manufacturer or Disodium Hydrogen Phosphate supplier.
PharmaCompass also assists you with knowing the Disodium Hydrogen Phosphate API Price utilized in the formulation of products. Disodium Hydrogen Phosphate API Price is not always fixed or binding as the Disodium Hydrogen Phosphate Price is obtained through a variety of data sources. The Disodium Hydrogen Phosphate Price can also vary due to multiple factors, including market conditions, regulatory modifications, or negotiated pricing deals.
A 04273_SIAL manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of 04273_SIAL, including repackagers and relabelers. The FDA regulates 04273_SIAL manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. 04273_SIAL API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
click here to find a list of 04273_SIAL manufacturers with USDMF, JDMF, KDMF, CEP, GMP, COA and API Price related information on PhamaCompass.
A 04273_SIAL supplier is an individual or a company that provides 04273_SIAL active pharmaceutical ingredient (API) or 04273_SIAL finished formulations upon request. The 04273_SIAL suppliers may include 04273_SIAL API manufacturers, exporters, distributors and traders.
click here to find a list of 04273_SIAL suppliers with USDMF, JDMF, KDMF, CEP, GMP, COA and API Price related information on PharmaCompass.
A 04273_SIAL DMF (Drug Master File) is a document detailing the whole manufacturing process of 04273_SIAL active pharmaceutical ingredient (API) in detail. Different forms of 04273_SIAL DMFs exist exist since differing nations have different regulations, such as 04273_SIAL USDMF, ASMF (EDMF), JDMF, CDMF, etc.
A 04273_SIAL DMF submitted to regulatory agencies in the US is known as a USDMF. 04273_SIAL USDMF includes data on 04273_SIAL's chemical properties, information on the facilities and procedures used, and details about packaging and storage. The 04273_SIAL USDMF is kept confidential to protect the manufacturer’s intellectual property.
click here to find a list of 04273_SIAL suppliers with USDMF on PharmaCompass.
A 04273_SIAL CEP of the European Pharmacopoeia monograph is often referred to as a 04273_SIAL Certificate of Suitability (COS). The purpose of a 04273_SIAL CEP is to show that the European Pharmacopoeia monograph adequately controls the purity of 04273_SIAL EP produced by a given manufacturer. Suppliers of raw materials can prove the suitability of 04273_SIAL to their clients by showing that a 04273_SIAL CEP has been issued for it. The manufacturer submits a 04273_SIAL CEP (COS) as part of the market authorization procedure, and it takes on the role of a 04273_SIAL CEP holder for the record. Additionally, the data presented in the 04273_SIAL CEP (COS) is managed confidentially and offers a centralized system acknowledged by numerous nations, exactly like the 04273_SIAL DMF.
A 04273_SIAL CEP (COS) is recognised by all 36 nations that make up the European Pharmacopoeia Convention. 04273_SIAL CEPs may be accepted in nations that are not members of the Ph. Eur. at the discretion of the authorities there.
click here to find a list of 04273_SIAL suppliers with CEP (COS) on PharmaCompass.
A 04273_SIAL written confirmation (04273_SIAL WC) is an official document issued by a regulatory agency to a 04273_SIAL manufacturer, verifying that the manufacturing facility of a 04273_SIAL active pharmaceutical ingredient (API) adheres to the Good Manufacturing Practices (GMP) regulations of the importing country. When exporting 04273_SIAL APIs or 04273_SIAL finished pharmaceutical products to another nation, regulatory agencies frequently require a 04273_SIAL WC (written confirmation) as part of the regulatory process.
click here to find a list of 04273_SIAL suppliers with Written Confirmation (WC) on PharmaCompass.
National Drug Code is a comprehensive database maintained by the FDA that contains information on all drugs marketed in the US. This directory includes information about finished drug products, unfinished drug products, and compounded drug products, including those containing 04273_SIAL as an active pharmaceutical ingredient (API).
The FDA updates the NDC directory daily. The NDC numbers for 04273_SIAL API and other APIs are published in this directory by the FDA.
The NDC unfinished drugs database includes product listing information submitted for all unfinished drugs, such as active pharmaceutical ingredients (APIs), drugs intended for further processing and bulk drug substances for compounding.
Pharmaceutical companies that manufacture 04273_SIAL as an active pharmaceutical ingredient (API) must furnish the FDA with an updated record of all drugs that they produce, prepare, propagate, compound, or process for commercial distribution in the US at their facilities.
The NDC directory also contains data on finished compounded human drug products that contain 04273_SIAL and are produced by outsourcing facilities. While these outsourcing facilities are not mandated to assign a 04273_SIAL NDC to their finished compounded human drug products, they may choose to do so.
click here to find a list of 04273_SIAL suppliers with NDC on PharmaCompass.
04273_SIAL Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of 04273_SIAL GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, API price, and more, enabling you to easily find the right 04273_SIAL GMP manufacturer or 04273_SIAL GMP API supplier for your needs.
A 04273_SIAL CoA (Certificate of Analysis) is a formal document that attests to 04273_SIAL's compliance with 04273_SIAL specifications and serves as a tool for batch-level quality control.
04273_SIAL CoA mostly includes findings from lab analyses of a specific batch. For each 04273_SIAL CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
04273_SIAL may be tested according to a variety of international standards, such as European Pharmacopoeia (04273_SIAL EP), 04273_SIAL JP (Japanese Pharmacopeia) and the US Pharmacopoeia (04273_SIAL USP).

