



API Suppliers

US DMFs Filed
0

CEP/COS Certifications
0

JDMFs Filed
0
Other Certificates
0
Other Suppliers
0
0

USA (Orange Book)
0

Europe
0

Canada
0

Australia
0

South Africa
0
Uploaded Dossiers
0
U.S. Medicaid
Annual Reports
0
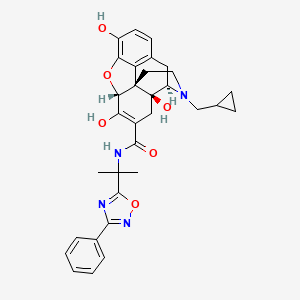

1. 17-(cyclopropylmethyl)-6,7-didehydro-4,5alpha-epoxy-3,6,14-trihydroxy-n-(2-(3-phenyl- 1,2,4-oxadiazol-5-yl)propan-2-yl)morphinan-7-carboxamide
2. S-297995
3. Symproic
1. 916072-89-4
2. S-297995
3. 03ksi6wlxh
4. S 297995
5. S-297,995
6. (4r,4as,7ar,12bs)-3-(cyclopropylmethyl)-4a,7,9-trihydroxy-n-[2-(3-phenyl-1,2,4-oxadiazol-5-yl)propan-2-yl]-1,2,4,5,7a,13-hexahydro-4,12-methanobenzofuro[3,2-e]isoquinoline-6-carboxamide
7. Naldemedine [usan]
8. Unii-03ksi6wlxh
9. Naldemedine [usan:inn]
10. Naldemedine [mi]
11. Naldemedine [inn]
12. Naldemedine (usan/inn)
13. Naldemedine [who-dd]
14. Gtpl9150
15. Schembl9880572
16. Chembl2105755
17. Dtxsid501030350
18. Bdbm50503604
19. Db11691
20. Compound 9k [pmid: 30446313]
21. Hy-19627
22. Morphinan-7-carboxamide, 17-(cyclopropylmethyl)-6,7-didehydro-4,5-epoxy-3,6,14- Trihydroxy-n-(1-methyl-1-(3-phenyl-1,2,4-oxadiazol-5-yl)ethyl)-,(5alpha)-
23. Cs-0016072
24. J3.573.009e
25. D10188
26. Q6960846
27. 17-(cyclopropylmethyl)-6,7-didehydro-4,5a-epoxy-3,6,14-trihydroxy-n-(2-(3-phenyl-1,2,4-oxadiazol-5-yl)propan-2-yl)morphinan-7-carboxamide
28. 17-(cyclopropylmethyl)-6,7-didehydro-4,5alpha-epoxy-3,6,14-trihydroxy-n-(2-(3-phenyl- 1,2,4-oxadiazol-5-yl)propan-2-yl)morphinan-7-carboxamide
| Molecular Weight | 570.6 g/mol |
|---|---|
| Molecular Formula | C32H34N4O6 |
| XLogP3 | 3.2 |
| Hydrogen Bond Donor Count | 4 |
| Hydrogen Bond Acceptor Count | 9 |
| Rotatable Bond Count | 6 |
| Exact Mass | 570.24783482 g/mol |
| Monoisotopic Mass | 570.24783482 g/mol |
| Topological Polar Surface Area | 141 Ų |
| Heavy Atom Count | 42 |
| Formal Charge | 0 |
| Complexity | 1140 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 4 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
| 1 of 1 | |
|---|---|
| Drug Name | SYMPROIC |
| Active Ingredient | NALDEMEDINE TOSYLATE |
| Company | SHIONOGI INC (Application Number: N208854. Patents: 9108975, RE46365, RE46375) |
For the treatment of opioid-induced constipation.
FDA Label
Rizmoic is indicated for the treatment of opioid-induced constipation (OIC) in adult patients who have previously been treated with a laxative.
Naldemedine is an opioid receptor antagonist with restricted movement across the blood brain barrier. This allows it to antagonize the periperal effects of opioid drugs such as constipation without interfering with the effects on the central nervous system.
A06AH05
A - Alimentary tract and metabolism
A06 - Drugs for constipation
A06A - Drugs for constipation
A06AH - Peripheral opioid receptor antagonists
A06AH05 - Naldemedine
Absorption
Tmax is 0.75 h. Administration with a high-fat meal reduces Cmax by 35% and increases Tmax to 2.5 h.
Route of Elimination
57% of naldemedine is excreted in the urine with 16-18% as the parent compound and 35% is excreted in the feces.
Volume of Distribution
The apparent volume of disribution during the terminal phase is 155 L
Naldemedine is mainly metabolized to nor-naldemedine by CYP3A. Some metabolism to naldemedine-3-glucuronide occurs via UGT1A3. Both metabolites are acitive but less potent than naldemedine. The relative exposures of these metabolites are 9-13% and <3% for nor-naldemedine and naldemedine-3-glucuronide respectively. Naldemedine is also cleaved in the intestine to form benzamidine and naldemedine carboxylic acid.
The terminal elimination half life is 11 h.
Naldemedine binds to and antagonizes mu-, delta-, and kappa-opioid receptors. The binding of opioid agonists to peripheral mu-opioid receptors slows the transit of feces through the intestine resulting in constipation. By antagonizing mu-opioid receptors, naldemedine inhibits this effect.






