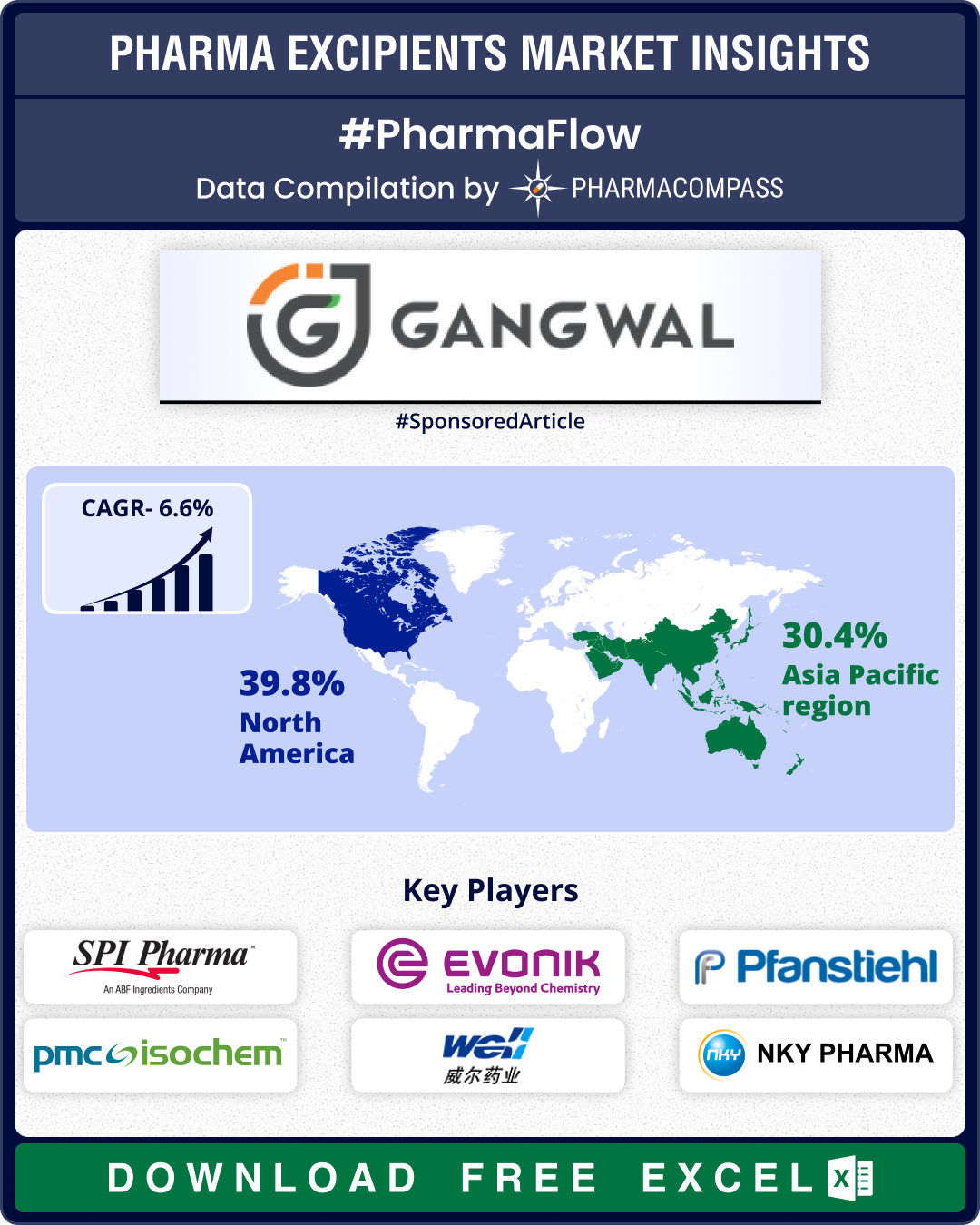
Excipient Market Overview: Clariant sets up its first PEG excipients facility in US; India harmonizes 22 excipient monographs
The market for pharmaceutical excipients — or inactive substances used in the production of dr
Excipient Market Overview: Evonik launches high-purity excipients; India mandates disclosures from March 2026
The global pharmaceutical excipients market continued to evolve in the third quarter (Q3) of 2025, s
DMF filings rise 4.5% in Q3 2025; China holds lead, India records 20% growth in submissions
The
third quarter (Q3) of 2025 witnessed a steady rise in Drug Master File (DMF) submissions to the
Excipient Market Overview: Roquette announces restructuring post IFF Pharma buyout; WHO, FDA advance regulatory frameworks
The pharmaceutical excipients market saw significant strategic
consolidations, technological develo
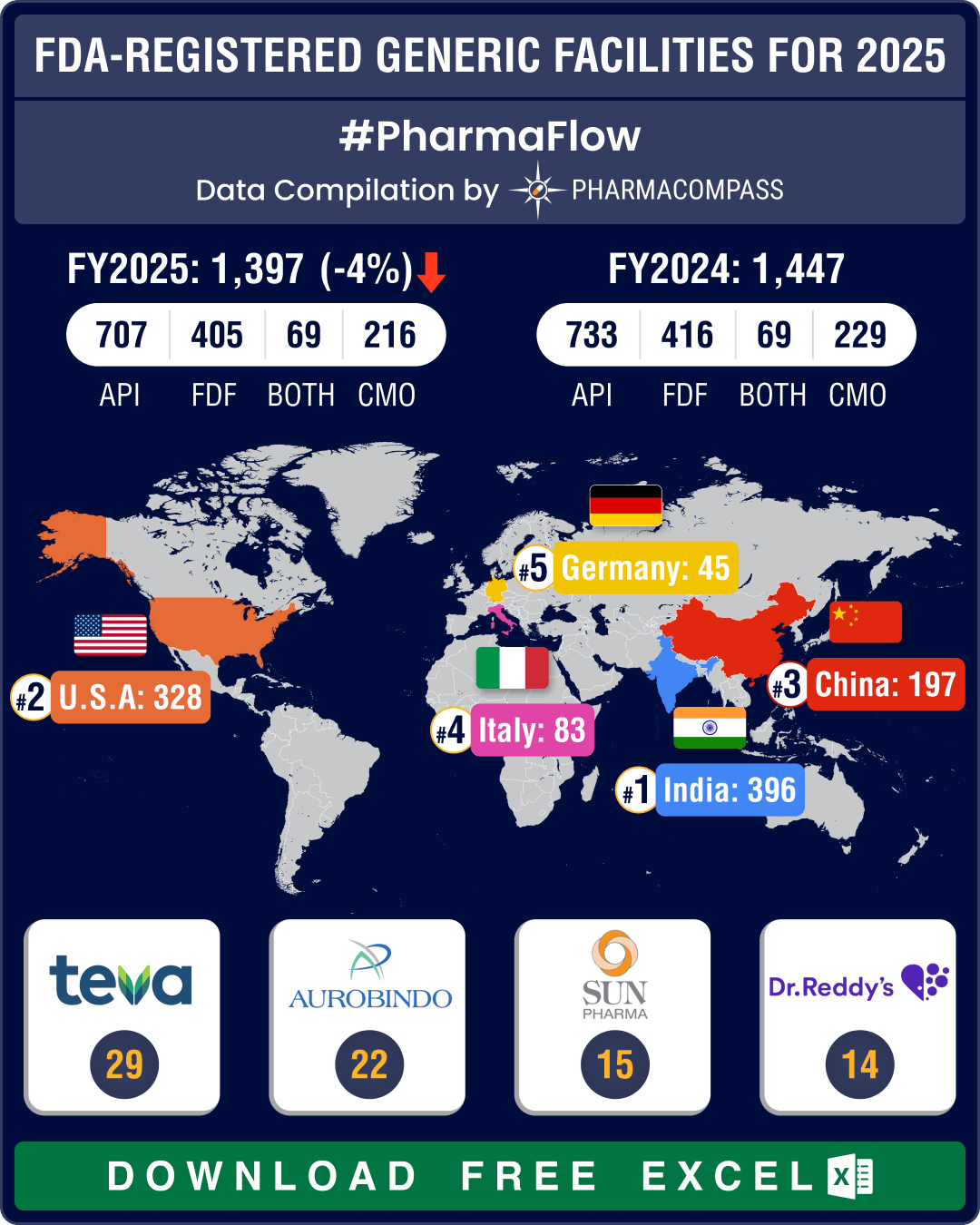
Chinese FDA-registered generic facilities gain steam, India maintains lead with 396 facilities
Every year, the US Food and Drug Administration (FDA) publishes the user fee amounts it will collect
DMF filings hit all-time high in Q3 2024; China tops list with 58% increase in Type II submissions
Drug Master Files, or DMFs, are confidential documents that play a crucial role in the pharmaceutica