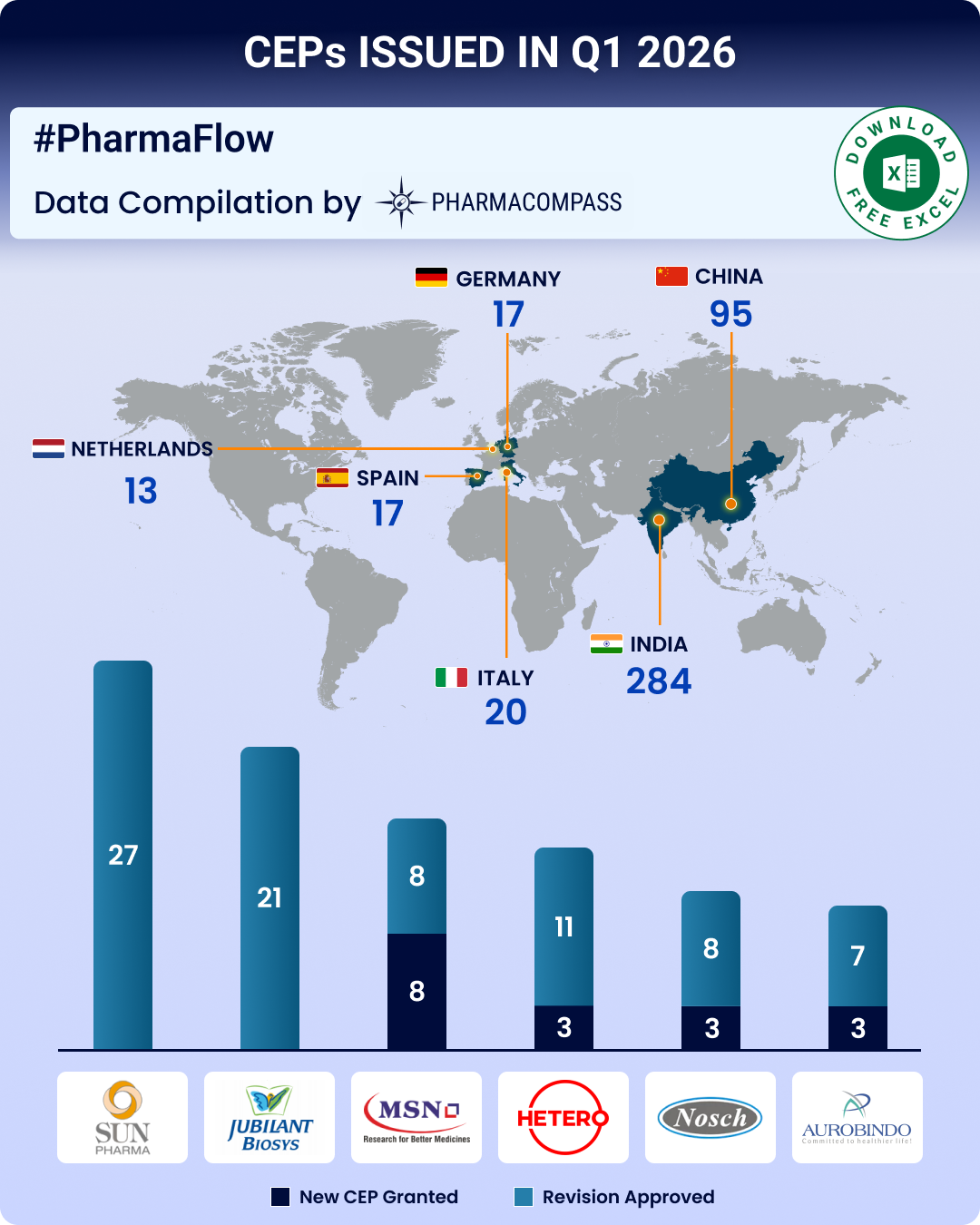
CEP Q1 2026 Update: CEP 2.0, EDQM’s new guidelines strengthen ecosystem; Indian firms top list of CEPs issued
PharmaCompass is introducing a new regulatory update
that tracks developments in Certificates of Su
US drug shortages hit record high in Q1 2024, impacts cancer, ADHD drugs; Lilly, Novo ramp up production
Drug shortages are threatening healthcare systems the world over.
Be it the US, Canada, Europe or A