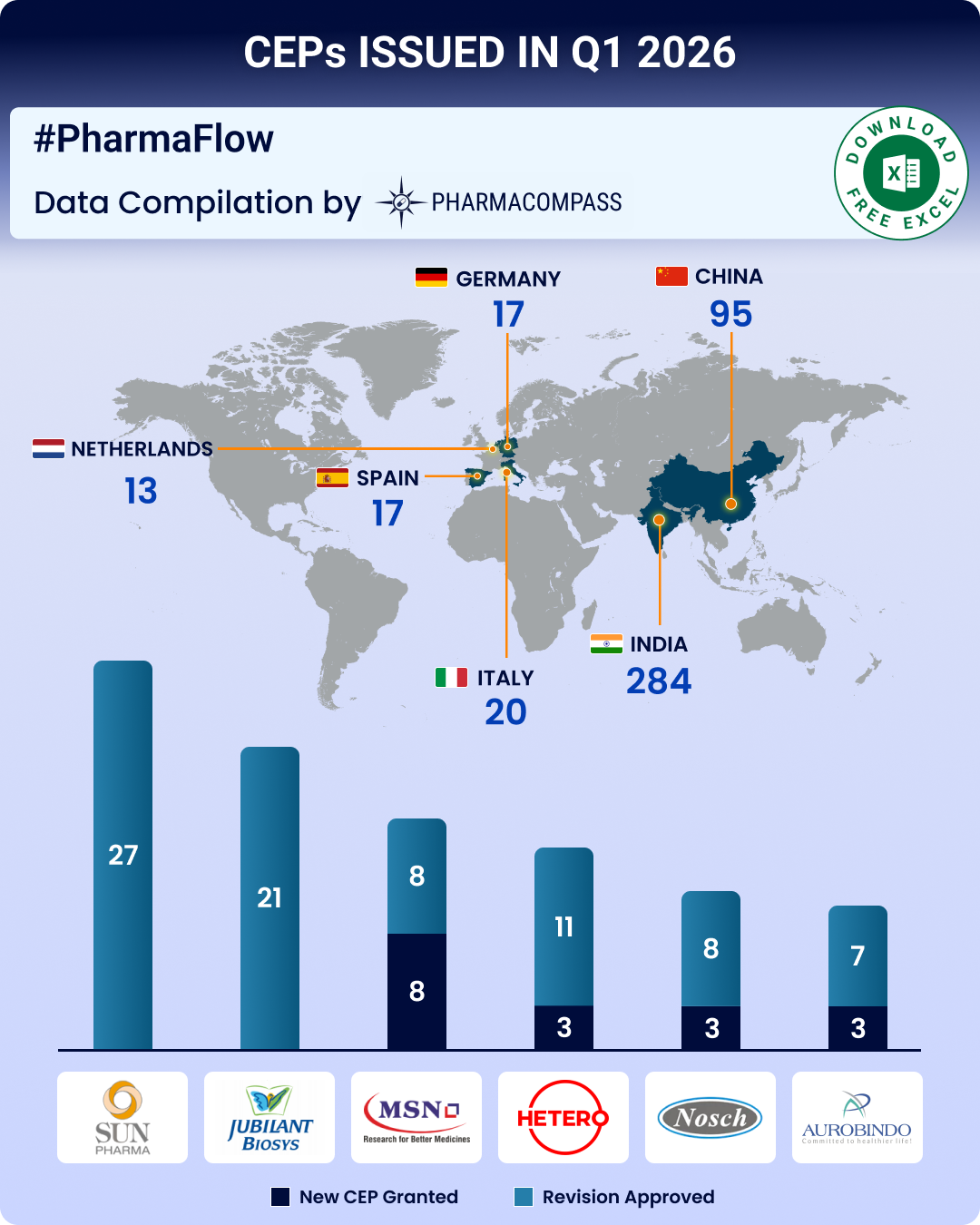
CEP Q1 2026 Update: CEP 2.0, EDQM’s new guidelines strengthen ecosystem; Indian firms top list of CEPs issued
PharmaCompass is introducing a new regulatory update
that tracks developments in Certificates of Su
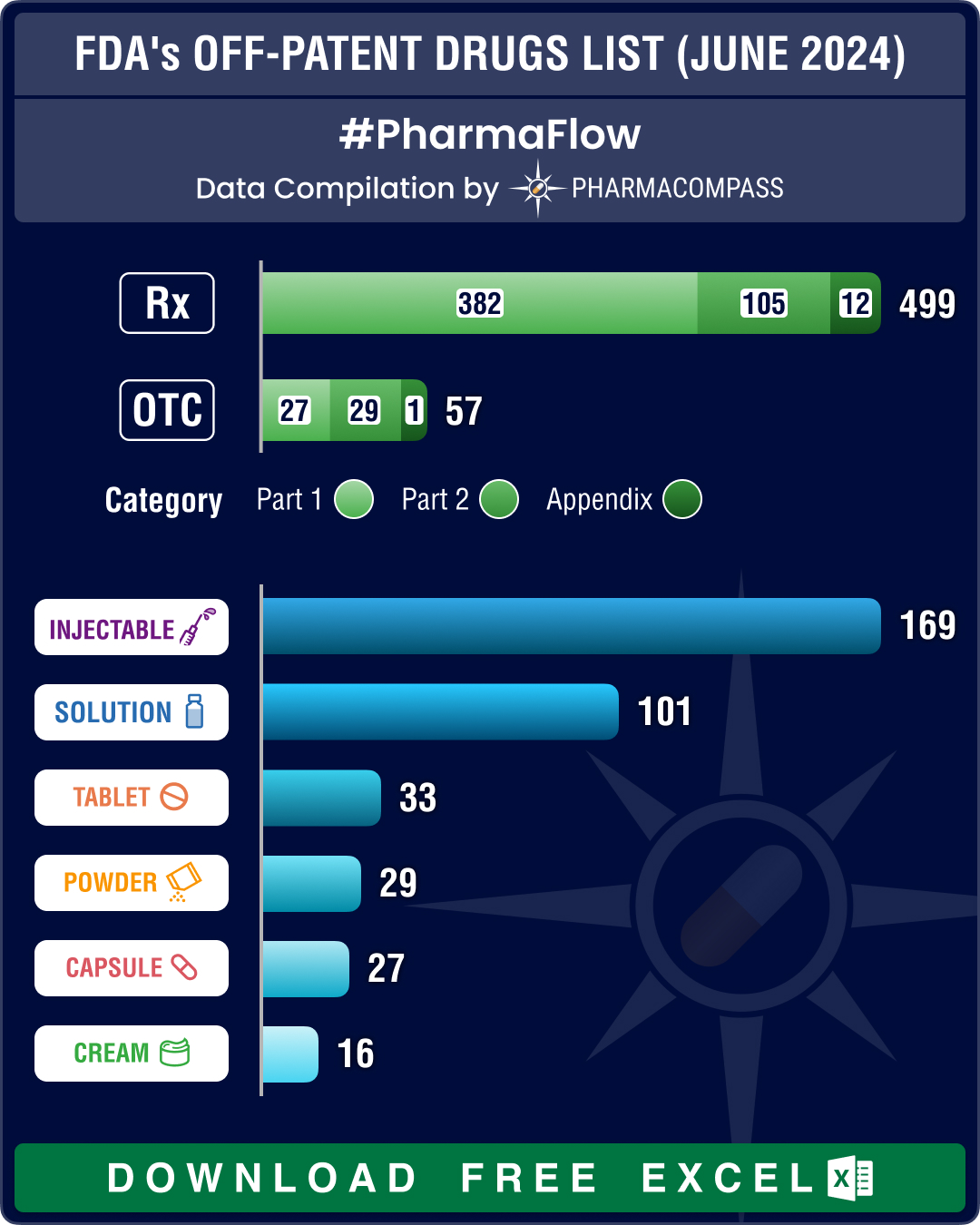
FDA’s December 2025 OPOE list features 784 prescription drugs, 73 OTC drugs
This
week, PharmaCompass brings you key highlights of the US Food and Drug Administration’s D