
Synopsis
Synopsis
0
USDMF
0
CEP/COS
0
JDMF
0
EU WC
0
KDMF
0
NDC API
0
VMF
0
EDQM
0
USP
0
JP
0
Others
0
FDF Dossiers
0
FDA Orange Book
0
Europe
0
Canada
0
Australia
0
South Africa
0
Listed Dossiers
DRUG PRODUCT COMPOSITIONS
0
US Patents
0
US Exclusivities
0
Health Canada Patents
US Medicaid
NA
Annual Reports
NA
Regulatory FDF Prices
NA
0
API
0
FDF
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
0
News #PharmaBuzz
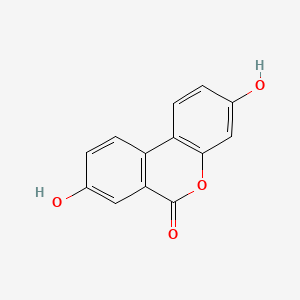

1. 3,8-dihydroxy-6h-dibenzo(b,d)pyran-6-one
1. 1143-70-0
2. 3,8-dihydroxy-6h-benzo[c]chromen-6-one
3. 3,8-dihydroxyurolithin
4. 3,8-dihydroxybenzo[c]chromen-6-one
5. 3,8-dihydroxy-6h-dibenzo(b,d)pyran-6-one
6. 6h-dibenzo[b,d]pyran-6-one, 3,8-dihydroxy-
7. 3,8-hydroxydibenzo-alpha-pyrone
8. Ilj8nef6dt
9. 3,8-dihydroxy-6h-dibenzo[b,d]pyran-6-one
10. Mfcd20275235
11. 6h-dibenzo(b,d)pyran-6-one, 3,8-dihydroxy-
12. Uro-a
13. 3,8-dihydroxybenzo(c)chromen-6-one
14. Urolithin-a
15. Unii-ilj8nef6dt
16. 3,8-dihydroxy-urolithin
17. Schembl803408
18. Chembl1836264
19. Dtxsid40150694
20. Chebi:168442
21. Urolithin A, >=97% (hplc)
22. Ex-a4345
23. S5312
24. Zinc13484727
25. Akos028108778
26. Ccg-266776
27. Cs-6305
28. Db15464
29. Ac-31959
30. Ds-19328
31. Sy070232
32. Db-122496
33. Hy-100599
34. Ft-0778244
35. Y10574
36. A901528
37. J-003086
38. Q15634120
39. 2-biphenylcarboxylic Acid, 2',4,4'-trihydroxy-, Delta-lactone
| Molecular Weight | 228.20 g/mol |
|---|---|
| Molecular Formula | C13H8O4 |
| XLogP3 | 2.3 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 4 |
| Rotatable Bond Count | 0 |
| Exact Mass | 228.04225873 g/mol |
| Monoisotopic Mass | 228.04225873 g/mol |
| Topological Polar Surface Area | 66.8 Ų |
| Heavy Atom Count | 17 |
| Formal Charge | 0 |
| Complexity | 317 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
 Hiray Pharma Solutions partners with global innovators to develop, manufacture and commercialize high-quality APIs and intermediates.
Hiray Pharma Solutions partners with global innovators to develop, manufacture and commercialize high-quality APIs and intermediates.
 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
 Hiray Pharma Solutions partners with global innovators to develop, manufacture and commercialize high-quality APIs and intermediates.
Hiray Pharma Solutions partners with global innovators to develop, manufacture and commercialize high-quality APIs and intermediates.
About the Company : Hiray Pharma Solutions is a global end-to-end CDMO specializing in APIs and intermediates. The company offers integrated services across Process R&D, Analytical R&D, manufacturing,...
About the Company : Enaltec, founded in 2006, specializes in developing and manufacturing complex, small-volume, technology-driven APIs at competitive prices, leveraging the India advantage for global...
 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results] Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Details:
Urolithin A is a dietary supplement drug, which is currently being evaluated in clinical studies for the treatment of undisclosed medical condition.
Lead Product(s): Urolithin A,Inapplicable
Therapeutic Area: Undisclosed Brand Name: Undisclosed
Study Phase: UndisclosedProduct Type: Dietary Supplement
Sponsor: Mprex Healthcare
Deal Size: Inapplicable Upfront Cash: Inapplicable
Deal Type: Inapplicable April 08, 2026

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Lead Product(s) : Urolithin A,Inapplicable
Therapeutic Area : Undisclosed
Highest Development Status : Undisclosed
Partner/Sponsor/Collaborator : Mprex Healthcare
Deal Size : Inapplicable
Deal Type : Inapplicable
Cerebrospinal Fluid Kinetics of Urolithin A
Details : Urolithin A is a dietary supplement drug, which is currently being evaluated in clinical studies for the treatment of undisclosed medical condition.
Product Name : Undisclosed
Product Type : Dietary Supplement
Upfront Cash : Inapplicable
April 08, 2026

Details:
Whey Protein is a protein drug, which is currently being evaluated in clinical studies for the treatment of aging.
Lead Product(s): Whey Protein,Creatine Monohydrate,Fucoidan,Urolithin A,Nicotinamide Mononucleotide,Multivitamin,L-Ergothioneine
Therapeutic Area: Cardiology/Vascular Diseases Brand Name: Undisclosed
Study Phase: UndisclosedProduct Type: Protein
Sponsor: Xprize Foundation | Abinopharm Inc | DoNotAge.org
Deal Size: Inapplicable Upfront Cash: Inapplicable
Deal Type: Inapplicable March 05, 2026

Lead Product(s) : Whey Protein,Creatine Monohydrate,Fucoidan,Urolithin A,Nicotinamide Mononucleotide,Multivitamin,L-Ergothioneine
Therapeutic Area : Cardiology/Vascular Diseases
Highest Development Status : Undisclosed
Partner/Sponsor/Collaborator : Xprize Foundation | Abinopharm Inc | DoNotAge.org
Deal Size : Inapplicable
Deal Type : Inapplicable
PRecision gerOMedicinE: Tailored Healthy agEing With Lifestyle, sUpplements and drugS (PROMETHEUS)
Details : Whey Protein is a protein drug, which is currently being evaluated in clinical studies for the treatment of aging.
Product Name : Undisclosed
Product Type : Protein
Upfront Cash : Inapplicable
March 05, 2026

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Details:
Urolithin A is a Dietary Supplement drug candidate, which is currently being evaluated in Undisclosed clinical studies for the treatment of Aging.
Lead Product(s): Urolithin A,Inapplicable
Therapeutic Area: Cardiology/Vascular Diseases Brand Name: Undisclosed
Study Phase: UndisclosedProduct Type: Dietary Supplement
Sponsor: Atlantia Food Clinical Trials
Deal Size: Inapplicable Upfront Cash: Inapplicable
Deal Type: Inapplicable November 17, 2025

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Lead Product(s) : Urolithin A,Inapplicable
Therapeutic Area : Cardiology/Vascular Diseases
Highest Development Status : Undisclosed
Partner/Sponsor/Collaborator : Atlantia Food Clinical Trials
Deal Size : Inapplicable
Deal Type : Inapplicable
The Effect of Urolithin A (Mitopure®) Supplementation on Muscle Strength in Healthy Middle-Aged A...
Details : Urolithin A is a Dietary Supplement drug candidate, which is currently being evaluated in Undisclosed clinical studies for the treatment of Aging.
Product Name : Undisclosed
Product Type : Dietary Supplement
Upfront Cash : Inapplicable
November 17, 2025

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Details:
Urolithin A is a Dietary Supplement drug candidate, which is currently being evaluated in Undisclosed clinical studies for the treatment of undefined medical condition.
Lead Product(s): Urolithin A,Inapplicable
Therapeutic Area: Undisclosed Brand Name: Undisclosed
Study Phase: UndisclosedProduct Type: Dietary Supplement
Sponsor: Mprex Healthcare Pvt. Ltd.
Deal Size: Inapplicable Upfront Cash: Inapplicable
Deal Type: Inapplicable November 17, 2025

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Lead Product(s) : Urolithin A,Inapplicable
Therapeutic Area : Undisclosed
Highest Development Status : Undisclosed
Partner/Sponsor/Collaborator : Mprex Healthcare Pvt. Ltd.
Deal Size : Inapplicable
Deal Type : Inapplicable
Bioavailability of Four Mitopure Formulations
Details : Urolithin A is a Dietary Supplement drug candidate, which is currently being evaluated in Undisclosed clinical studies for the treatment of undefined medical condition.
Product Name : Undisclosed
Product Type : Dietary Supplement
Upfront Cash : Inapplicable
November 17, 2025

Details:
Urolithin A is a Dietary Supplement drug candidate, which is currently being evaluated in Undisclosed clinical studies for the treatment of Neoplasms.
Lead Product(s): Urolithin A,Inapplicable
Therapeutic Area: Oncology Brand Name: Undisclosed
Study Phase: UndisclosedProduct Type: Dietary Supplement
Sponsor: Amazentis | Georg-Speyer-Haus
Deal Size: Inapplicable Upfront Cash: Inapplicable
Deal Type: Inapplicable September 08, 2025

Lead Product(s) : Urolithin A,Inapplicable
Therapeutic Area : Oncology
Highest Development Status : Undisclosed
Partner/Sponsor/Collaborator : Amazentis | Georg-Speyer-Haus
Deal Size : Inapplicable
Deal Type : Inapplicable
Details : Urolithin A is a Dietary Supplement drug candidate, which is currently being evaluated in Undisclosed clinical studies for the treatment of Neoplasms.
Product Name : Undisclosed
Product Type : Dietary Supplement
Upfront Cash : Inapplicable
September 08, 2025

Details:
Urolithin A is a Dietary Supplement drug candidate, which is currently being evaluated in clinical studies for the treatment of Sleep Wake Disorders.
Lead Product(s): Urolithin A,Fisetin
Therapeutic Area: Sleep Brand Name: Undisclosed
Study Phase: UndisclosedProduct Type: Dietary Supplement
Sponsor: Wuhan Wuchang Hospital | Abinopharm Inc
Deal Size: Inapplicable Upfront Cash: Inapplicable
Deal Type: Inapplicable May 25, 2025

Lead Product(s) : Urolithin A,Fisetin
Therapeutic Area : Sleep
Highest Development Status : Undisclosed
Partner/Sponsor/Collaborator : Wuhan Wuchang Hospital | Abinopharm Inc
Deal Size : Inapplicable
Deal Type : Inapplicable
Evaluation of Urolithin A and Fisetin on Improving Sleep and Aging Biomarkers
Details : Urolithin A is a Dietary Supplement drug candidate, which is currently being evaluated in clinical studies for the treatment of Sleep Wake Disorders.
Product Name : Undisclosed
Product Type : Dietary Supplement
Upfront Cash : Inapplicable
May 25, 2025

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Details:
Urolithin A is a Dietary Supplement drug candidate, which is currently being evaluated in clinical studies for the treatment of undefined medical condition.
Lead Product(s): Urolithin A,Inapplicable
Therapeutic Area: Undisclosed Brand Name: Undisclosed
Study Phase: UndisclosedProduct Type: Dietary Supplement
Sponsor: Mprex Healthcare Pvt. Ltd.
Deal Size: Inapplicable Upfront Cash: Inapplicable
Deal Type: Inapplicable February 28, 2025

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Lead Product(s) : Urolithin A,Inapplicable
Therapeutic Area : Undisclosed
Highest Development Status : Undisclosed
Partner/Sponsor/Collaborator : Mprex Healthcare Pvt. Ltd.
Deal Size : Inapplicable
Deal Type : Inapplicable
Bioavailability of Seven Mitopure Formulations
Details : Urolithin A is a Dietary Supplement drug candidate, which is currently being evaluated in clinical studies for the treatment of undefined medical condition.
Product Name : Undisclosed
Product Type : Dietary Supplement
Upfront Cash : Inapplicable
February 28, 2025

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Details:
Urolithin A is a Dietary Supplement drug candidate, which is currently being evaluated in clinical studies for the treatment of Frailty.
Lead Product(s): Urolithin A,Inapplicable
Therapeutic Area: Musculoskeletal Brand Name: Undisclosed
Study Phase: UndisclosedProduct Type: Dietary Supplement
Sponsor: Université du Québec a Montréal | McGill University Health Centre Research Institute
Deal Size: Inapplicable Upfront Cash: Inapplicable
Deal Type: Inapplicable August 16, 2024

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Lead Product(s) : Urolithin A,Inapplicable
Therapeutic Area : Musculoskeletal
Highest Development Status : Undisclosed
Partner/Sponsor/Collaborator : Université du Québec a Montréal | McGill University Health Centre Research Institute
Deal Size : Inapplicable
Deal Type : Inapplicable
Impact of Urolithin A (Mitopure) on Mitochondrial Quality in Muscle of Frail Older Adults
Details : Urolithin A is a Dietary Supplement drug candidate, which is currently being evaluated in clinical studies for the treatment of Frailty.
Product Name : Undisclosed
Product Type : Dietary Supplement
Upfront Cash : Inapplicable
August 16, 2024

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Details:
Urolithin A is a Dietary Supplement drug candidate, which is currently being evaluated in clinical studies for the treatment of undefined medical condition.
Lead Product(s): Urolithin A,Inapplicable
Therapeutic Area: Undisclosed Brand Name: Undisclosed
Study Phase: UndisclosedProduct Type: Dietary Supplement
Sponsor: Mprex Healthcare Pvt. Ltd.
Deal Size: Inapplicable Upfront Cash: Inapplicable
Deal Type: Inapplicable April 12, 2024

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Lead Product(s) : Urolithin A,Inapplicable
Therapeutic Area : Undisclosed
Highest Development Status : Undisclosed
Partner/Sponsor/Collaborator : Mprex Healthcare Pvt. Ltd.
Deal Size : Inapplicable
Deal Type : Inapplicable
Bioavailability of Five Mitopure Formulations
Details : Urolithin A is a Dietary Supplement drug candidate, which is currently being evaluated in clinical studies for the treatment of undefined medical condition.
Product Name : Undisclosed
Product Type : Dietary Supplement
Upfront Cash : Inapplicable
April 12, 2024

Details:
Mitopure (Urolithin A) is a Dietary Supplement drug candidate, which is currently being evaluated in phase II clinical studies for the treatment of Pulmonary Disease, Chronic Obstructive.
Lead Product(s): Urolithin A,Inapplicable
Therapeutic Area: Pulmonary/Respiratory Diseases Brand Name: Mitopure
Study Phase: Phase IIProduct Type: Dietary Supplement
Sponsor: Amazentis
Deal Size: Inapplicable Upfront Cash: Inapplicable
Deal Type: Inapplicable March 21, 2024

Lead Product(s) : Urolithin A,Inapplicable
Therapeutic Area : Pulmonary/Respiratory Diseases
Highest Development Status : Phase II
Partner/Sponsor/Collaborator : Amazentis
Deal Size : Inapplicable
Deal Type : Inapplicable
Oral Supplementation During PR Participation in COPD
Details : Mitopure (Urolithin A) is a Dietary Supplement drug candidate, which is currently being evaluated in phase II clinical studies for the treatment of Pulmonary Disease, Chronic Obstructive.
Product Name : Mitopure
Product Type : Dietary Supplement
Upfront Cash : Inapplicable
March 21, 2024

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]ABOUT THIS PAGE
13
PharmaCompass offers a list of Urolithin A API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Urolithin A manufacturer or Urolithin A supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Urolithin A manufacturer or Urolithin A supplier.
A Urolithin A manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Urolithin A, including repackagers and relabelers. The FDA regulates Urolithin A manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Urolithin A API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
click here to find a list of Urolithin A manufacturers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PhamaCompass.
A Urolithin A supplier is an individual or a company that provides Urolithin A active pharmaceutical ingredient (API) or Urolithin A finished formulations upon request. The Urolithin A suppliers may include Urolithin A API manufacturers, exporters, distributors and traders.
click here to find a list of Urolithin A suppliers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PharmaCompass.
Urolithin A Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Urolithin A GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Urolithin A GMP manufacturer or Urolithin A GMP API supplier for your needs.
A Urolithin A CoA (Certificate of Analysis) is a formal document that attests to Urolithin A's compliance with Urolithin A specifications and serves as a tool for batch-level quality control.
Urolithin A CoA mostly includes findings from lab analyses of a specific batch. For each Urolithin A CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Urolithin A may be tested according to a variety of international standards, such as European Pharmacopoeia (Urolithin A EP), Urolithin A JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Urolithin A USP).

