
1. 9041-08-1
2. Heparin, Sodium Salt
3. Nadroparin Sodium
4. Heparin Sodium Salt (mw 15kda)
5. D78319
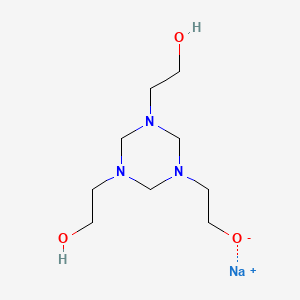
| Molecular Weight | 241.26 g/mol |
|---|---|
| Molecular Formula | C9H20N3NaO3 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 6 |
| Rotatable Bond Count | 6 |
| Exact Mass | g/mol |
| Monoisotopic Mass | g/mol |
| Topological Polar Surface Area | 73.2 |
| Heavy Atom Count | 16 |
| Formal Charge | 0 |
| Complexity | 162 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 2 |
BUILDING BLOCK


