

1. Verquvo
1. 1350653-20-1
2. Verquvo
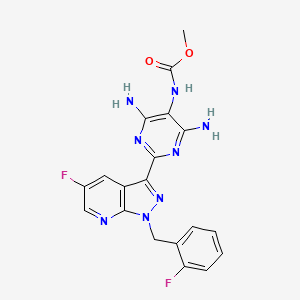
3. Methyl (4,6-diamino-2-(5-fluoro-1-(2-fluorobenzyl)-1h-pyrazolo[3,4-b]pyridin-3-yl)pyrimidin-5-yl)carbamate
4. Bay-1021189
5. Mk-1242
6. Vericiguatum
7. Bay1021189
8. Lv66adm269
9. Methyl N-[4,6-diamino-2-[5-fluoro-1-[(2-fluorophenyl)methyl]pyrazolo[3,4-b]pyridin-3-yl]pyrimidin-5-yl]carbamate
10. Chebi:142432
11. Dtxsid001318361
12. Methyl (4,6-diamino-2-(5-fluoro-1-((2-fluorophenyl)methyl)-1h-pyrazolo(3,4-b)pyridin-3-yl(pyrimidin-5-yl)carbamate
13. Methyl N-(4,6-diamino-2-(5-fluoro-1-(2-fluorobenzyl)-1h-pyrazolo(3,4-b)pyridin-3-yl)pyrimidin-5-yl)carbamate
14. Methyl N-[4,6-diamino-2-[5-fluoro-1-(2-fluorobenzyl)-1h-pyrazolo[3,4-b]pyridin-3-yl]pyrimidin-5-yl]carbamate
15. Methyl (4,6-diamino-2-(5-fluoro-1-(2-fluorobenzyl)-1h-pyrazolo(3,4-b)pyridin-3-yl)pyrimidin-5-yl)carbamate
16. Methyl N-(4,6-diamino-2-{5-fluoro-1-[(2-fluorophenyl)methyl]-1h-pyrazolo[3,4-b]pyridin-3-yl}pyrimidin-5-yl)carbamate
17. Methyl {4,6-diamino-2-[5-fluoro-1-(2-fluorobenzyl)-1h-pyrazolo[3,4-b]pyridin-3-yl]pyrimidin-5-yl}carbamate
18. Methyl [4,6-diamino-2-[5-fluoro-1-(2-fluorobenzyl)-1h-pyrazolo[3,4-b]pyridin-3-yl]pyrimidin-5-yl]carbamate
19. Methyl N-(4,6-diamino-2-(5-fluoro-1-((2-fluorophenyl)methyl)-1h-pyrazolo(3,4-b)pyridin-3-yl)pyrimidin-5-yl)carbamate
20. Methyl N-(4,6-diamino-2-(5-fluoro-1-((2-fluorophenyl)methyl)pyrazolo(3,4-b)pyridin-3-yl)pyrimidin-5-yl)carbamate
21. Refchem:58201
22. Dtxcid001748172
23. 814-370-3
24. Bay 1021189
25. Vericiguat [inn]
26. Mfcd28502029
27. Vericiguat [usan]
28. Unii-lv66adm269
29. C19h16f2n8o2
30. Verquvo (tn)
31. Vericiguat (standard)
32. Vericiguat [jan]
33. Vericiguat [usan:inn]
34. Vericiguatbay1021189
35. Vericiguat [who-dd]
36. Bay1021189; Verquvo
37. Schembl429958
38. Vericiguat (jan/usan/inn)
39. Orb1307618
40. Chembl4066936
41. Schembl29379616
42. Schembl29382575
43. Schembl30395636
44. Vericiguat [orange Book]
45. Gtpl10010
46. Qzfhixarhdbpby-uhfffaoysa-n
47. Glxc-07291
48. Hms5087f03
49. Vericiguat (bay 1021189)?
50. Ex-a4694
51. Who 9805
52. Hy-16774r
53. S9693
54. Akos025289795
55. Cs-6981
56. Db15456
57. Sb16806
58. Ac-36737
59. Da-58986
60. Hy-16774
61. Ms-27517
62. D11051
63. P14957
64. Q27283201
65. Carbamic Acid, N-[4,6-diamino-2-[5-fluoro-1-[(2-fluorophenyl)methyl]-1h-pyrazolo[3,4-b]pyridin-3-yl]-5-pyrimidinyl]-, Methyl Ester
66. Methyl (4,6-diamino-2-{5-fluoro-1-[(2-fluorophenyl)methyl]-1h-pyrazolo[3,4-b]pyridin-3-yl}pyrimidin-5-yl)carbamate
67. Methyl 4,6-diamino-2-(5-fluoro-1-(2-fluorobenzyl)-1h-pyrazolo[3,4-b]pyridin-3-yl)pyrimidin-5-ylcarbamate
68. Methyl{4,6-diamino-2-[5-fluoro-1-(2-fluorobenzyl)-1h-pyrazolo[3,4-b]pyridin-3-yl]pyrimidin-5-yl}carbamate
69. Vericiguat; Carbamic Acid, N-[4,6-diamino-2-[5-fluoro-1-[(2-fluorophenyl)methyl]-1h-pyrazolo[3,4-b]pyridin-3-yl]-5-pyrimidinyl]-, Methyl Ester; Bay 1021189; Mk 1242; Methyl N-[4,6-diamino-2-[5-fluoro-1-(2-fluorobenzyl)-1h-pyrazolo[3,4-b]pyridin-3-yl]pyrimidin-5-yl]ca
| Molecular Weight | 426.4 g/mol |
|---|---|
| Molecular Formula | C19H16F2N8O2 |
| XLogP3 | 1.5 |
| Hydrogen Bond Donor Count | 3 |
| Hydrogen Bond Acceptor Count | 10 |
| Rotatable Bond Count | 5 |
| Exact Mass | Da |
| Monoisotopic Mass | Da |
| Topological Polar Surface Area | 147 |
| Heavy Atom Count | 31 |
| Formal Charge | 0 |
| Complexity | 622 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
Vericiguat is indicated in adults with symptomatic, chronic heart failure and an ejection fraction of <45% to reduce the risk of cardiovascular death and heart failure-related hospitalization following a hospitalization for heart failure or need for outpatient intravenous diuretics.
Vericiguat is a novel agent, an oral soluble guanylate cyclase stimulator used to treat heart failure with reduced ejection fraction (HFrEF) patients. It is also useful for chronic heart failure with recently decompensated or worsening heart failure, high-risk heart failure with reduced ejection fraction, especially for patients who had been rehospitalized or are on intravenous diuretic therapy, or patients requiring urgent treatment for heart failure.
C - Cardiovascular system
C01 - Cardiac therapy
C01D - Vasodilators used in cardiac diseases
C01DX - Other vasodilators used in cardiac diseases
C01DX22 - Vericiguat
ATCvet Code
QC - Cardiovascular system
QC01 - Cardiac therapy
QC01D - Vasodilators used in cardiac diseases
QC01DX - Other vasodilators used in cardiac diseases
QC01DX22 - Vericiguat
Absorption
Following the administration of 10mg of vericiguat by mouth once daily, the average steady-state Cmax and AUC in patients with heart failure is 350 mcg/L and 6,680 mcgh/L, respectively, with a Tmax of 1 hour. The absolute bioavailability of orally-administered vericiguat is approximately 93% when taken with food - co-administration with meals has been shown to reduce pharmacokinetic variability, increase Tmax to roughly 4 hours, and increase Cmax and AUC by 41% and 44%, respectively.
Route of Elimination
Following the oral administration of radiolabeled vericiguat, approximately 53% of the administered radioactivity was recovered in the urine and 45% in the feces. A human mass balance study found that the portion recovered in the urine comprised approximately 40.8% N-glucuronide metabolite, 7.7% other metabolites, and 9% unchanged parent drug, while virtually the entire portion recovered in the feces comprised unchanged vericiguat.
Volume of Distribution
In healthy subjects the steady-state volume of distribution of vericiguat is approximately 44 liters.
Clearance
Vericiguat is a low-clearance drug, with an observed plasma clearance of 1.6 L/h in healthy volunteers and 1.3 L/h in patients with systolic heart failure.
Vericiguat is primarily metabolized via phase II conjugation reactions, with CYP-mediated oxidative metabolism comprising a small (<5%) portion of its overall biotransformation. The major inactive metabolite, vericiguat N-glucuronide (M1), is formed by UGT1A9 and, to a lesser extent, UGT1A1. Other identified metabolites include a denbenzylated compound and an M15 metabolite thought to be the result of oxidative metabolism, although these metabolites are poorly characterized.
In patients with heart failure, the half-life of vericiguat is 30 hours.
Heart failure (HF) involves, amongst other morphologic and physiologic changes, the impaired synthesis of nitric oxide (NO) and decreased activity of soluble guanylate cyclase (sGC). Functioning normally, NO binds to sGC and stimulates the synthesis of intracellular cyclic guanosine monophosphate (cGMP), a second messenger involved in the maintenance of vascular tone, as well as cardiac contractility and remodeling. Defects in this pathway are thought to contribute to the myocardial and vascular dysfunction associated with heart failure and are therefore a desirable target in its treatment. Vericiguat directly stimulates sGC by binding to a target site on its beta-subunit, bypassing the need for NO-mediated activation, and in doing so causes an increase in the production of intracellular cGMP that results in vascular smooth muscle relaxation and vasodilation.
BUILDING BLOCK


