

1. Maleic Acid Hydrazide
2. Milurit
3. Milurite
1. 123-33-1
2. 3,6-dihydroxypyridazine
3. 3,6-pyridazinediol
4. 1,2-dihydropyridazine-3,6-dione
5. 3,6-pyridazinedione, 1,2-dihydro-
6. 1,2-dihydro-3,6-pyridazinedione
7. Maleic Acid Hydrazide
8. Stuntman
9. Vondalhyde
10. Malzid
11. Retard
12. Sucker-stuff
13. Antergon
14. Antyrost
15. Malazide
16. Regulox
17. Vondrax
18. Regulox 36
19. Maleic Hydrazine
20. Sprout/off
21. De-sprout
22. Sprout-stop
23. Super-de-sprout
24. De-cut
25. Maintain 3
26. Super Sucker-stuff
27. Burtolin
28. Fazor
29. Mazide
30. Super De-sprout
31. N,n-maleoylhydrazine
32. Royal Mh-30
33. 3,6-dioxopyridazine
34. Royal Slo-gro
35. Drexel-super P
36. Regulox W
37. Super Sprout Stop
38. 6-hydroxy-3(2h)-pyridazinone
39. Fair Ps
40. Unriprim
41. Malein 30
42. Fair-2
43. Regulox 50 W
44. Fair Plus
45. Super Sucker-stuff Hc
46. Fair 30
47. Hydrazide Maleique
48. 3(2h)-pyridazinone, 6-hydroxy-
49. Maleinsaeurehydrazid
50. Mh 36 Bayer
51. Maleic Acid Cyclic Hydrazide
52. Mh 30
53. Mh-40
54. 6-hydroxy-2h-pyridazin-3-one
55. Ent 18,870
56. Hydrazid Kyseliny Maleinove
57. 1,2-dihydro-3,6-pyradizinedione
58. Malepin
59. Dtxsid9020792
60. 1,2,3,6-tetrahydro-3,6-dioxopyridazine
61. Tq7l3fcv66
62. Nsc-13892
63. 1,2,3,6-tetrahydropyridazine-3,6-dione
64. Dtxcid10792
65. Hydrazide, Maleic
66. Acid Hydrazide, Maleic
67. Hydrazide, Maleic Acid
68. Refchem:800769
69. 204-619-9
70. 600-121-4
71. Gotax
72. Milurit
73. Pyridazine-3,6-diol
74. 116595-47-2
75. Mg-t
76. Rcra Waste Number U148
77. Mfcd00006665
78. Maleic Acid, Hydrazide
79. Maleic Acid, Cyclic Hydrazide
80. Kmh
81. Mah
82. Chembl1489913
83. Chebi:81771
84. 1,6-pyridazinedione
85. 1,6-dione
86. 3, 1,2-dihydro-
87. Wln: T6vmmvj
88. F1918-0075
89. 6-hydroxy-2h-pyridazin-2-one
90. Caswell No. 352
91. Maleichydrazide
92. 1,3,6-tetrahydro-3,6-dioxopyridazine
93. Cas-123-33-1
94. Maleinsaeurehydrazid [german]
95. Maleic Hydrazide [bsi:iso]
96. Ccris 1879
97. Hsdb 1162
98. Hydrazide Maleique [iso-french]
99. Hydrazid Kyseliny Maleinove [czech]
100. Einecs 204-619-9
101. Nsc 13892
102. Rcra Waste No. U148
103. Unii-tq7l3fcv66
104. Epa Pesticide Chemical Code 051501
105. Maleinhydrazid
106. Sorbatran
107. Yadi
108. Ai3-18870
109. Meleic Hydrazide
110. Fair-plus
111. Pyridazine-3,6(1h,2h)-dione
112. Nsc-8823
113. Mfcd00071686
114. Nsc-48832
115. Maleic Cyclic Hydrazide
116. Maleic Hydrazide (standard)
117. Pyridazine, 3,6-dihydroxy-
118. Schembl55071
119. Maleic Hydrazide [mi]
120. Schembl154932
121. Schembl554403
122. Schembl554404
123. Maleic Hydrazide [iso]
124. Orb1705423
125. Orb3139811
126. Schembl3678851
127. Schembl4138427
128. Schembl7277672
129. Schembl8536307
130. Maleic Hydrazide [hsdb]
131. Maleic Hydrazide [iarc]
132. Schembl29740320
133. Nsc8823
134. Smssf-0044116
135. Albb-020885
136. Msk24017
137. Nsc13892
138. Nsc48832
139. Str03411
140. Tox21_201829
141. Tox21_300662
142. Bdbm50021573
143. Ebc-12511
144. Ebc-44277
145. Hy-59354r
146. Sbb004322
147. Stk357922
148. Akos000121025
149. Akos000275072
150. Akos017269103
151. Fm15520
152. Ks-5282
153. Maleic Hydrazide, Reagentplus(r), 99%
154. Ncgc00091173-01
155. Ncgc00091173-02
156. Ncgc00091173-03
157. Ncgc00091173-04
158. Ncgc00254570-01
159. Ncgc00259378-01
160. Ac-11591
161. Hy-59354
162. Db-028316
163. Cs-0007730
164. M0015
165. Ns00005908
166. St50807893
167. En300-17761
168. C18474
169. Maleic Hydrazide, Vetec(tm) Reagent Grade, 98%
170. Ac-907/30002020
171. Q677933
172. Maleic Hydrazide, Pestanal(r), Analytical Standard
173. Z57032544
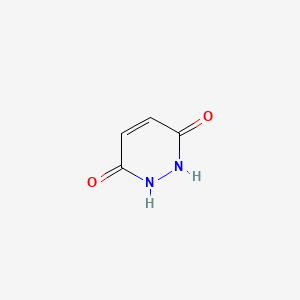
174. 1,2-dihydropyridazine-3,6-dione; 6-hydroxy-3-pyridazinone
175. 3,6-pyridazinediol; Maleic Acid Hydrazide; Maleic Hydrazide
176. Inchi=1/c4h4n2o2/c7-3-1-2-4(8)6-5-3/h1-2h,(h,5,7)(h,6,8
| Molecular Weight | 112.09 g/mol |
|---|---|
| Molecular Formula | C4H4N2O2 |
| XLogP3 | -0.8 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 2 |
| Rotatable Bond Count | 0 |
| Exact Mass | Da |
| Monoisotopic Mass | Da |
| Topological Polar Surface Area | 58.2 |
| Heavy Atom Count | 8 |
| Formal Charge | 0 |
| Complexity | 143 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
Antimetabolites
Drugs that are chemically similar to naturally occurring metabolites, but differ enough to interfere with normal metabolic pathways. (From AMA Drug Evaluations Annual, 1994, p2033)
Enzyme Inhibitors
Compounds or agents that combine with an enzyme in such a manner as to prevent the normal substrate-enzyme combination and the catalytic reaction.
Gout Suppressants
Agents that increase uric acid excretion by the kidney (URICOSURIC AGENTS), decrease uric acid production (antihyperuricemics), or alleviate the pain and inflammation of acute attacks of gout.
Herbicides
Pesticides used to destroy unwanted vegetation, especially various types of weeds, grasses (POACEAE), and woody plants. Some plants develop HERBICIDE RESISTANCE.
Plant Growth Regulators
Any of the hormones produced naturally in plants and active in controlling growth and other functions. There are three primary classes: auxins, cytokinins, and gibberellins.
Free Radical Scavengers
Substances that eliminate free radicals. Among other effects, they protect PANCREATIC ISLETS against damage by CYTOKINES and prevent myocardial and pulmonary REPERFUSION INJURY.
...After oral doses of 100 mg/kg bw, rabbits excreted 43 to 62% maleic hydrazide unchanged in urine within 48 hr.
IARC. Monographs on the Evaluation of the Carcinogenic Risk of Chemicals to Humans. Geneva: World Health Organization, International Agency for Research on Cancer, 1972-PRESENT. (Multivolume work).
(14)C Maleic hydrazide was administered orally to rats. After 3 days, very little (14)C activity was detected in tissues or blood and carbon dioxide accounted for only 0.2% of administered dose. Maleic hydrazide was rapidly excreted via urine unchanged (>90%) and as conjugate (6-8%).
Menzie, C.M. Metabolism of Pesticides. U.S. Department of the Interior, Bureau of Sport Fisheries and Wildlife, Publication 127. Washington, DC: U.S. Government Printing Office, 1969., p. 236
Slowly absorbed over period of 24 hr by quackgrass. Rain within this period will reduce effectiveness. Rate of absorption is function of cell turgidity. Most effective absorption occurs when soil moisture is at field capacity and relative high humidity. Translocates more effectively downward. Once absorbed, maleic hydrazide is freely translocated to active growing points in plant. maleic hydrazide becomes fixed within the plant and is not metabolized.
Weed Science Society of America. Herbicide Handbook. 5th ed. Champaign, Illinois: Weed Science Society of America, 1983., p. 323
In white ash and black locust seedlings, most of maleic hydrazide was translocated to leaves and stems of black locust seedlings within 1 day after treatment, but for white ash seedlings it remained in the stem tissue. After 30 days, the (14)C was concentrated in the leaves of the black locust seedlings, but only in the stem and at the injection point of white ash seedlings. Chromatography of the extracts showed no detectable metabolite in black locust seedlings, but 2 metabolites were detected in the white ash seedlings.
Mischke CF, Domir SC; Pestic Sci 13 (3): 304-8 (1982)
For more Absorption, Distribution and Excretion (Complete) data for MALEIC HYDRAZIDE (10 total), please visit the HSDB record page.
When applied to tea, Camelia sinensis, MH /(maleic hydrazine)/ was degraded to lactic acid, succinic acid, maleimide and hydrazine. Extracts of treated wheat seedlings... identified... beta-glycoside of MH. ...In presence of oxygen, MH undergoes photolysis to form succinic, maleic and nitric acid; and in absence of oxygen... succinic acid.
Menzie, C.M. Metabolism of Pesticides. U.S. Department of the Interior, Bureau of Sport Fisheries and Wildlife, Publication 127. Washington, DC: U.S. Government Printing Office, 1969., p. 236
When applied to silver maple... and American sycamore... seedlings, maleic hydrazide translocated to all parts of the plant. In the plant tissue, a metabolite was formed. Hydrolysis products of the metabolite indicated a conjugate of maleic hydrazide and glucose. When applied to tobacco plants, (14)C maleic hydrazide was rapidly translocated to growing tissues. Translocation to roots also occurred. A small amount of (14)CO2 evolved. The major metabolite in foliar tissues was identified as the beta-D-glucoside of maleic hydrazide.
Menzie, C.M. Metabolism of Pesticides-Update III. Special Scientific Report- Wildlife No. 232. Washington, DC: U.S.Department of the Interior, Fish and Wildlife Service, 1980., p. 391
In one radio label experiment in rats, 77% of the administered radioactivity was recovered in urine within 6 days. 90% of the activity was found to be unaltered maleic hydrazide. The remainder was present as a conjugate of MH.
Gosselin, R.E., R.P. Smith, H.C. Hodge. Clinical Toxicology of Commercial Products. 5th ed. Baltimore: Williams and Wilkins, 1984., p. II-343
In rats, the urine and feces samples /collected following oral administration of 3,6-dione-labelled 14C-maleic hydrazide/ contained two peaks. Poor chromatographic separation and low levels of radiolabel in the fecal samples precluded reliable identification, but the peaks appeared to represent maleic hydrazide and possibly fumaric acid. The major peak in urine, representing 60% of the urinary radiolabel in males and 80% in females, co-chromatographed with maleic hydrazide. The minor urinary peak was initially found to co-chromatograph with maleimide, fumaric acid, or maleic diamide, depending on the solvent system, but subsequent investigation with deconjugation with a beta-glucuronidase containing sulfatase activity and HPLC showed this peak to be a maleic acid conjugate, probably a sulfate.
FAO/WHO Joint Meeting on Pesticide Residues; Maleic hydrazide (Pesticide Residues in Food: 1996 Evaluations). Available from, as of April 15, 2004: https://www.inchem.org/documents/jmpr/jmpmono/v96pr08.htm
For more Metabolism/Metabolites (Complete) data for MALEIC HYDRAZIDE (7 total), please visit the HSDB record page.
Maleic hydrazide inhibits mitosis in actively growing tissues of treated plants and also has profound effects on rate of respiration. ... Maleic acid ... reacts with -SH compounds ... /and/ competes with receptor sites of ... succinic dehydrogenase. ... Inhibition ... may represent site of action ... in plants.
White-Stevens, R. (ed.). Pesticides in the Environment: Volume 1, Part 1, Part 2. New York: Marcel Dekker, Inc., 1971., p. 65
BUILDING BLOCK


