



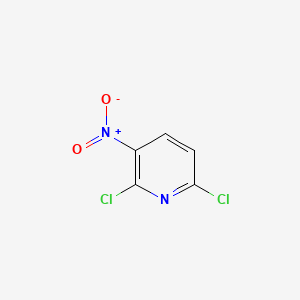

1. 16013-85-7
2. Pyridine, 2,6-dichloro-3-nitro-
3. 2,6-dichloro-3-nitro-pyridine
4. 3-nitro-2,6-dichloropyridine
5. Mfcd00006234
6. Kh7f529l8w
7. 2,6-dichloro-3-nitro Pyridine
8. Einecs 240-151-1
9. Cholest-4-ene, 3-chloro-, (3
10. A)-
11. 2,6-dichloronitropyridine
12. Dichloronitropyridine
13. Unii-kh7f529l8w
14. Schembl56616
15. 2,6-dichioro 3-nitropyridine
16. 2,6-dichloro 3-nitropyridine
17. 2,6-dichloro-5-nitropyridine
18. Dtxsid10166803
19. Zinc967353
20. Act01414
21. 2,6-dichloro-3-nitropyridine Tech.
22. Stk801264
23. 2,6-bis(chloranyl)-3-nitro-pyridine
24. Akos005255252
25. Ac-2768
26. Cs-w001992
27. Pb28956
28. Ps-3147
29. Cholest-4-ene,3-chloro-,(3a)-(9ci)
30. Bp-10097
31. Sy002863
32. Db-005147
33. Am20070294
34. D2761
35. Ft-0610569
36. Ft-0651285
37. 2,6-dichloro-3-nitropyridine 13901-10-5
38. En300-38973
39. 13d857
40. A807471
41. Ac-907/30002032
42. W-107981
43. 2,6-dichloro-3-nitropyridine, Technical Grade, 92%
44. F0001-0573
| Molecular Weight | 192.98 g/mol |
|---|---|
| Molecular Formula | C5H2Cl2N2O2 |
| XLogP3 | 2.6 |
| Hydrogen Bond Donor Count | 0 |
| Hydrogen Bond Acceptor Count | 3 |
| Rotatable Bond Count | 0 |
| Exact Mass | 191.9493327 g/mol |
| Monoisotopic Mass | 191.9493327 g/mol |
| Topological Polar Surface Area | 58.7 Ų |
| Heavy Atom Count | 11 |
| Formal Charge | 0 |
| Complexity | 161 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |



