



1. Alecensa
2. Ch5424802
3. Ro5424802
1. 1256580-46-7
2. Ch5424802
3. Alecensa
4. Af-802
5. Ch 5424802
6. Af802
7. Ro5424802
8. Alectinib (ch5424802)
9. Unii-lij4ct1z3y
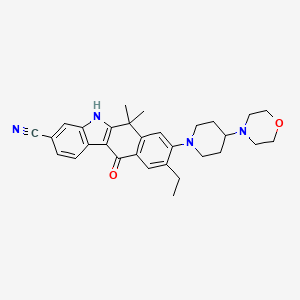
10. 9-ethyl-6,6-dimethyl-8-(4-morpholinopiperidin-1-yl)-11-oxo-6,11-dihydro-5h-benzo[b]carbazole-3-carbonitrile
11. Ch-5424802
12. 9-ethyl-6,6-dimethyl-8-(4-morpholin-4-ylpiperidin-1-yl)-11-oxo-5h-benzo[b]carbazole-3-carbonitrile
13. Af 802
14. Lij4ct1z3y
15. 9-ethyl-6,11-dihydro-6,6-dimethyl-8-[4-(4-morpholinyl)-1-piperidinyl]-11-oxo-5h-benzo[b]carbazole-3-carbonitrile
16. Chembl1738797
17. Ro-5424802
18. Arq-761
19. 9-ethyl-6,6-dimethyl-8-[4-(morpholin-4-yl)piperidin-1-yl]-11-oxo-6,11-dihydro-5h-benzo[b]carbazole-3-carbonitrile
20. 9-ethyl-6,6-dimethyl-8-[4-(morpholin-4-yl)piperidin-1-yl]-11-oxo-5h-benzo[b]carbazole-3-carbonitrile
21. Alectinib [inn]
22. 9-ethyl-6,11-dihydro-6,6-dimethyl-8-(4-(4-morpholinyl)-1-piperidinyl)-11-oxo-5h-benzo(b)carbazole-3-carbonitrile
23. Alectinib [usan:inn]
24. 5h-benzo(b)carbazole-3-carbonitrile, 9-ethyl-6,11-dihydro-6,6-dimethyl-8-(4-(4-morpholinyl)-1-piperidinyl)-11-oxo-
25. 5h-benzo[b]carbazole-3-carbonitrile, 9-ethyl-6,11-dihydro-6,6-dimethyl-8-[4-(4-morpholinyl)-1-piperidinyl]-11-oxo-
26. 9-ethyl-6,6-dimethyl-8-(4-(morpholin-4-yl)piperidin-1-yl)-11-oxo-6,11-dihydro-5h-benzo(b)carbazole-3-carbonitrile
27. Alectinib Free Base
28. Emh
29. Alectinib [mi]
30. Alectinib (usan/inn)
31. Alectinib [usan]
32. Alectinib [who-dd]
33. Ch5424802(alectinib)
34. Ch 5424802, Alectinib
35. Mls006010971
36. Schembl896753
37. Gtpl7739
38. Chebi:90936
39. Amy3288
40. Dtxsid50154840
41. Ex-a406
42. Ch-5424802 Hcl
43. Hms3672c13
44. Hms3744i17
45. Bcp02589
46. Bdbm50362781
47. Mfcd19440988
48. Nsc764040
49. Nsc764821
50. Nsc794611
51. Nsc799328
52. S2762
53. Zinc66166864
54. 1256580-46-7 (free Base)
55. Akos025290737
56. Bcp9000514
57. Cs-0518
58. Db11363
59. Nsc-764040
60. Nsc-764821
61. Nsc-794611
62. Nsc-799328
63. Rg-7853
64. Sb16515
65. Ncgc00346688-01
66. Ncgc00346688-03
67. Ncgc00346688-04
68. 9-ethyl-6,11-dihydro-6,6-dimethyl-8-[4-(4-morpholinyl)-1-piperidinyl]-11-oxo-5h-benzo[b]carbazole-3-
69. Ac-29020
70. As-17008
71. Hy-13011
72. Smr004702775
73. A8393
74. Ft-0748626
75. Ch-5424802/rg-7853
76. D10542
77. J-005256
78. J-519506
79. Q21099132
80. Ch5424802,cas:1256580-46-7
81. 9-ethyl-6,11-dihydro-6,6-dimethyl-8-[4-(4-morpholinyl)-1-piperidinyl]-11-oxo-5h-benzo[b]carbazole-3-carbonitrilech5424802
82. 9-ethyl-6,6-dimethyl-8-(4-morpholin-4-yl-piperidin-1-yl)-11-oxo-6,11-dihydro-5h-benzo[b]carbazole-3-carbonitrile
83. 9-ethyl-6,6-dimethyl-8-(4-morpholin-4-ylpiperidin-1-yl)-11-oxo-5h-benzo[b]carbazol-3-carbonitril
| Molecular Weight | 482.6 g/mol |
|---|---|
| Molecular Formula | C30H34N4O2 |
| XLogP3 | 5.2 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 5 |
| Rotatable Bond Count | 3 |
| Exact Mass | 482.26817634 g/mol |
| Monoisotopic Mass | 482.26817634 g/mol |
| Topological Polar Surface Area | 72.4 Ų |
| Heavy Atom Count | 36 |
| Formal Charge | 0 |
| Complexity | 867 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
| 1 of 1 | |
|---|---|
| Drug Name | ALECENSA |
| Active Ingredient | ALECTINIB HYDROCHLORIDE |
| Company | HOFFMANN-LA ROCHE (Application Number: N208434. Patents: 9126931, 9365514, 9440922) |
Alectinib is a kinase inhibitor indicated for the treatment of patients with anaplastic lymphoma kinase (ALK)-positive, metastatic non-small cell lung cancer (NSCLC) who have progressed on or are intolerant to crizotinib. This indication is approved under accelerated approval based on tumor response rate and duration of response. Continued approval for this indication may be contingent upon verification and description of clinical benefit in confirmatory trials.
FDA Label
Alecensa as monotherapy is indicated for the first-line treatment of adult patients with anaplastic lymphoma kinase (ALK)-positive advanced non-small cell lung cancer (NSCLC).
Alecensa as monotherapy is indicated for the treatment of adult patients with ALKpositive advanced NSCLC previously treated with crizotinib.
L01ED03
L01XE36
S76 | LUXPHARMA | Pharmaceuticals Marketed in Luxembourg | Pharmaceuticals marketed in Luxembourg, as published by d'Gesondheetskeess (CNS, la caisse nationale de sante, www.cns.lu), mapped by name to structures using CompTox by R. Singh et al. (in prep.). List downloaded from https://cns.public.lu/en/legislations/textes-coordonnes/liste-med-comm.html. Dataset DOI:10.5281/zenodo.4587355
L - Antineoplastic and immunomodulating agents
L01 - Antineoplastic agents
L01E - Protein kinase inhibitors
L01ED - Anaplastic lymphoma kinase (alk) inhibitors
L01ED03 - Alectinib
Absorption
Alectinib reached maximal concentrations at 4 hours following administration of 600 mg twice daily under fed conditions in patients with ALK-positive NSCLC. The absolute bioavailability was 37% in the fed state. A high-fat, high-calorie meal increased the combined exposure of alectinib and its major metabolite M4 by 3.1-fold following oral administration of a single 600 mg dose.
Route of Elimination
When radioactively labeled, 98% of radioactivity was found in feces with 84% of that amount excreted as unchanged alectinib and 6% as M4. Less than 0.5% was found to be recovered in urine.
Volume of Distribution
4016 L
Clearance
The apparent clearance is 81.9L/hr for alectinib and 217 L/hr for M4.
Alectinib is metabolized by CYP3A4 to its major active metabolite M4. M4 is then further metabolized by CYP3A4. Both alectinib and M4 demonstrate similar in vivo and in vitro activity. In vitro studies suggest that alectinib is not a substrate for P-gp while M4 is.
The mean elimination half life is 33 hr for alectinib and 31 hr for M4.
Alectinib is a second generation oral drug that selectively inhibits the activity of anaplastic lymphoma kinase (ALK) tyrosine kinase. It is specifically used in the treatment of non-small cell lung cancer (NSCLC) expressing the ALK-EML4 (echinoderm microtubule-associated protein-like 4) fusion protein that causes proliferation of NSCLC cells. Inhibition of ALK prevents phosphorylation and subsequent downstream activation of STAT3 and AKT resulting in reduced tumour cell viability. Both alectinib and its major active metabolite M4 demonstrate similar in vivo and in vitro activity against multiple mutant forms of ALK.




