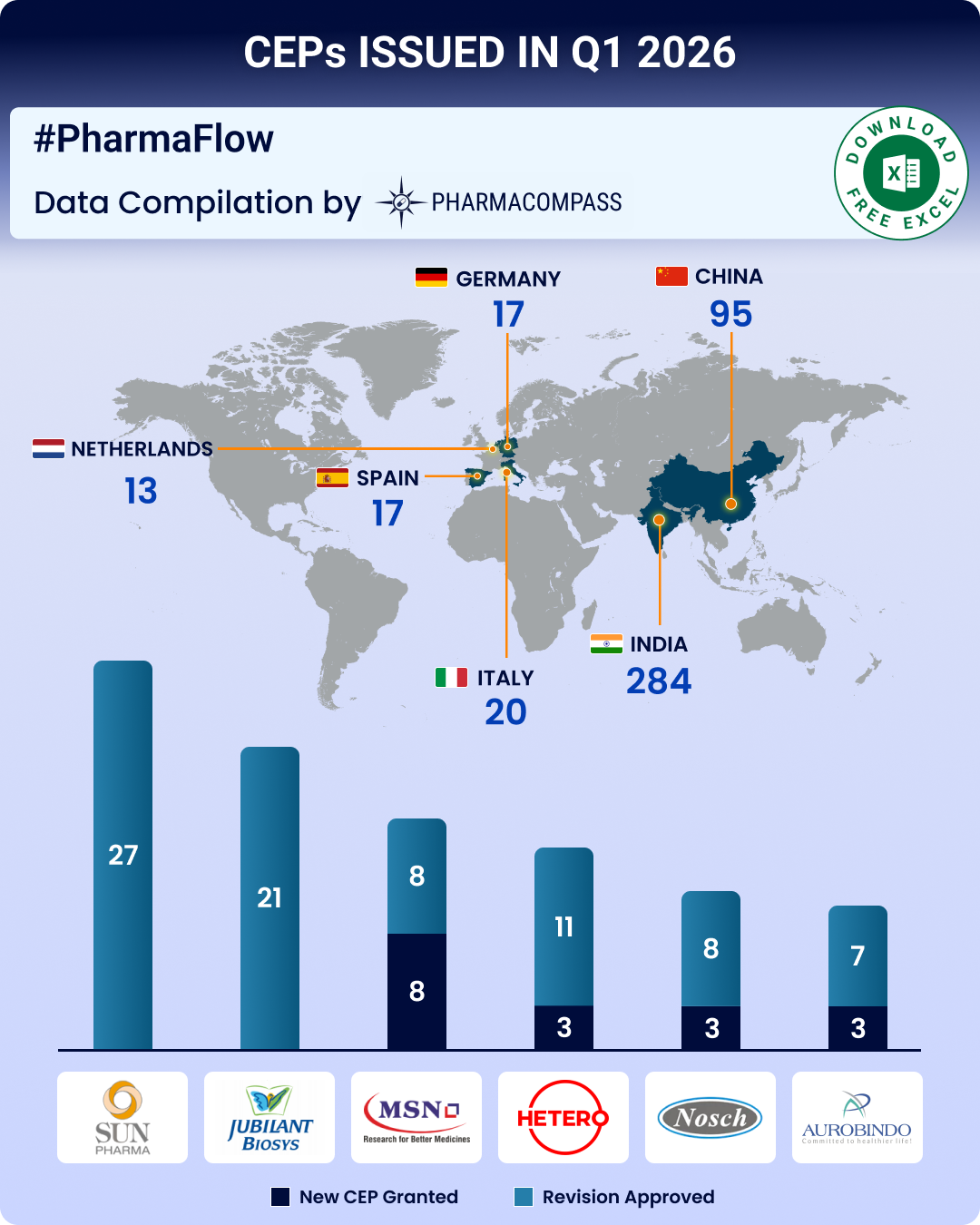
26 Jan 2026
// PRESS RELEASE
15 Oct 2025
// PRESS RELEASE
27 Mar 2017
// PRESS RELEASE
Latest Content by PharmaCompass

Archimica: Excellence in complex chemical technologies, regulatory-approved manufacturing, and API innovation.
About
Industry Trade Show
Exhibiting
06-08 October, 2026
TIDES Europe 2026TIDES Europe 2026
Industry Trade Show
Exhibiting
03-05 November, 2026
American Pharma Outsou...American Pharma Outsourcing Summit
Industry Trade Show
Attending
19-20 November, 2026
CONTACT DETAILS






Email ID:
comm.dpt@euticals.com
LinkedIn Official link:
https://www.linkedin.com/company/euticals-spa
Events
Webinars & Exhibitions
Industry Trade Show
Exhibiting
06-08 October, 2026
TIDES Europe 2026TIDES Europe 2026
Industry Trade Show
Exhibiting
03-05 November, 2026
American Pharma Outsou...American Pharma Outsourcing Summit
Industry Trade Show
Attending
19-20 November, 2026
CORPORATE CONTENT #SupplierSpotlight
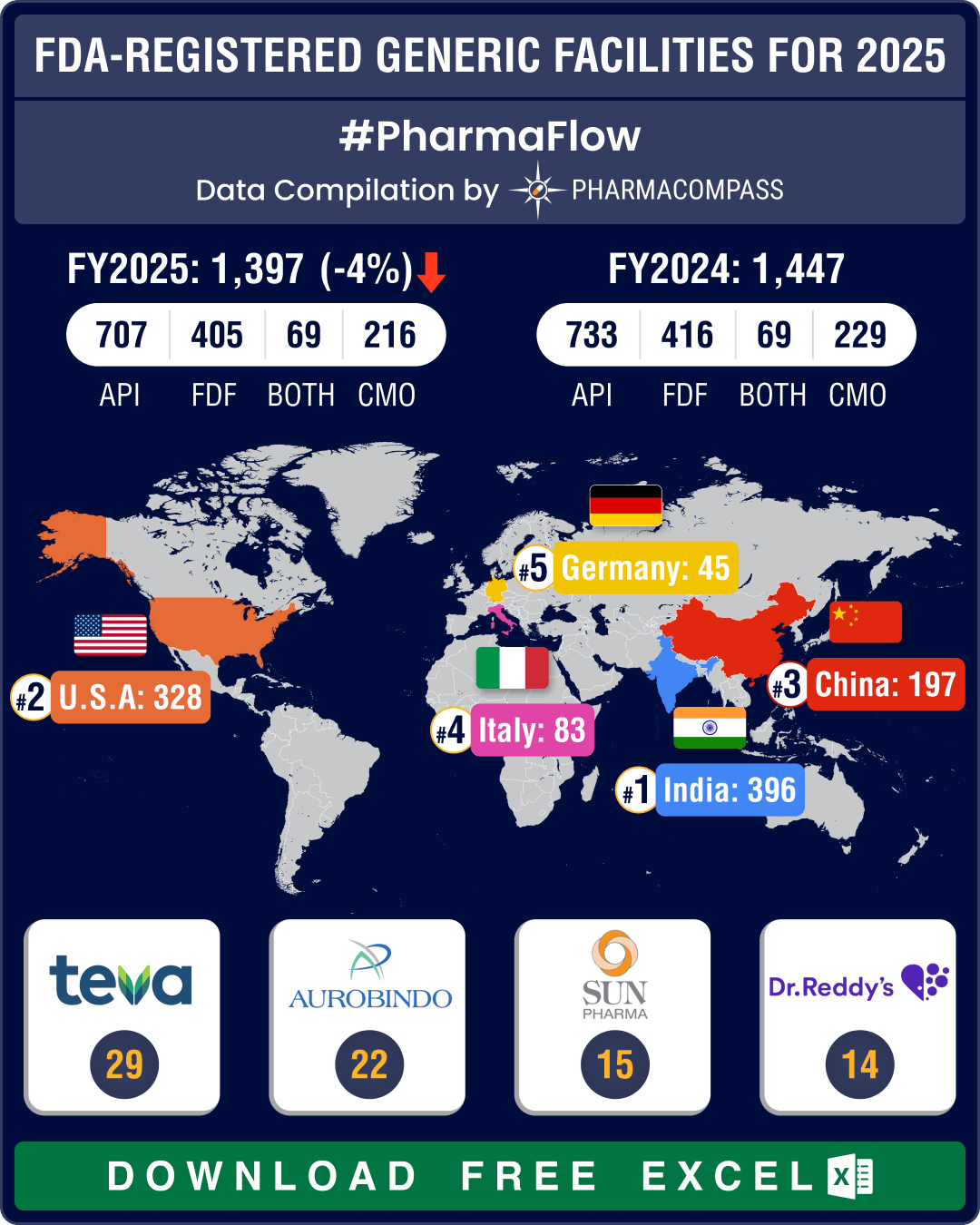
https://www.pharmacompass.com/radio-compass-blog/chinese-fda-registered-generic-facilities-gain-steam-india-maintains-lead-with-396-facilities

26 Jan 2026
// PRESS RELEASE

15 Oct 2025
// PRESS RELEASE
https://pihealthsciences.com/pi-health-sciences-announces-the-acquisition-of-archimica-s-p-a-from-livia-group/

27 Mar 2017
// PRESS RELEASE
https://www.pharmacompass.com/pdf/news/axyntis-independent-multi-site-leader-in-the-strategic-sector-of-fine-chemicals-in-france-1624093922.pdf
Inspections and registrations
ABOUT THIS PAGE
Archimica is a supplier offers 14 products (APIs, Excipients or Intermediates).
Find Cytarabine bulk with DMF, CEP offered by Archimica
Find Hydroxyurea bulk with DMF, CEP offered by Archimica
Find Bromfenac bulk with DMF offered by Archimica
Find Capecitabine bulk with DMF offered by Archimica
Find Cytarabine bulk with CEP offered by Archimica
Find Disopyramide bulk with DMF offered by Archimica
Find Hydralazine Hydrochloride bulk with DMF offered by Archimica
Find Lithium Carbonate bulk with DMF offered by Archimica
Find Mianserin Hydrochloride bulk with CEP offered by Archimica
Find Oxcarbazepine bulk with DMF offered by Archimica
Find Pirenzepine bulk with DMF offered by Archimica
Find Sucralfate bulk with DMF offered by Archimica
Find Sulfapyridine bulk with DMF offered by Archimica
Find Choline Alfoscerate bulk offered by Archimica


 Archimica
Archimica