NDC Code(s) : 66993-210-19, 66993-211-19
Packager : Prasco Laboratories
Category : HUMAN PRESCRIPTION DRUG LABEL
DEA Schedule : none
Marketing Status : New Drug Application
INGREDIENTS AND APPEARANCE
| Vancomycin Hydrochloridevancomycin hydrochloride CAPSULE | ||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| Vancomycin Hydrochloridevancomycin hydrochloride CAPSULE | ||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
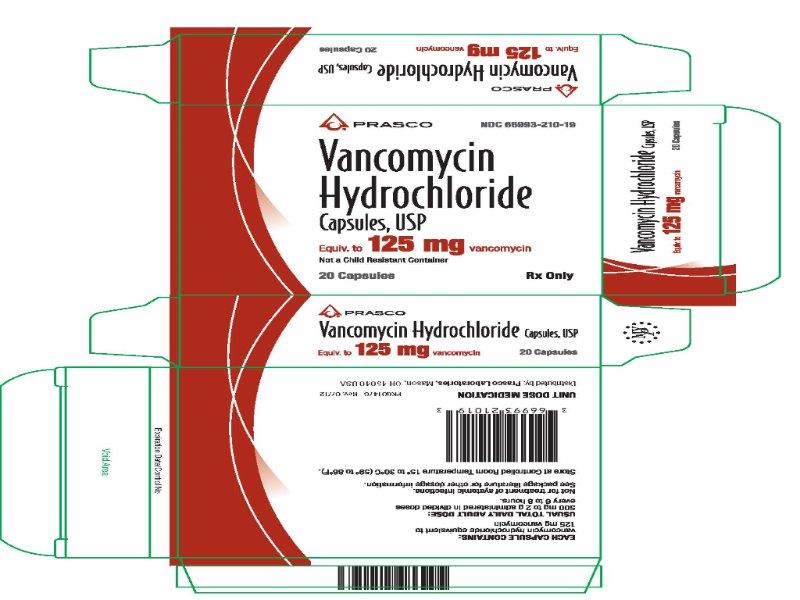
PRINCIPAL DISPLAY PANEL
Vancomycin Hydrochloride Capsules USP, Equiv. to 125 mg vancomycin
NDC 66993-210-19
Rx Only
20 Capsules
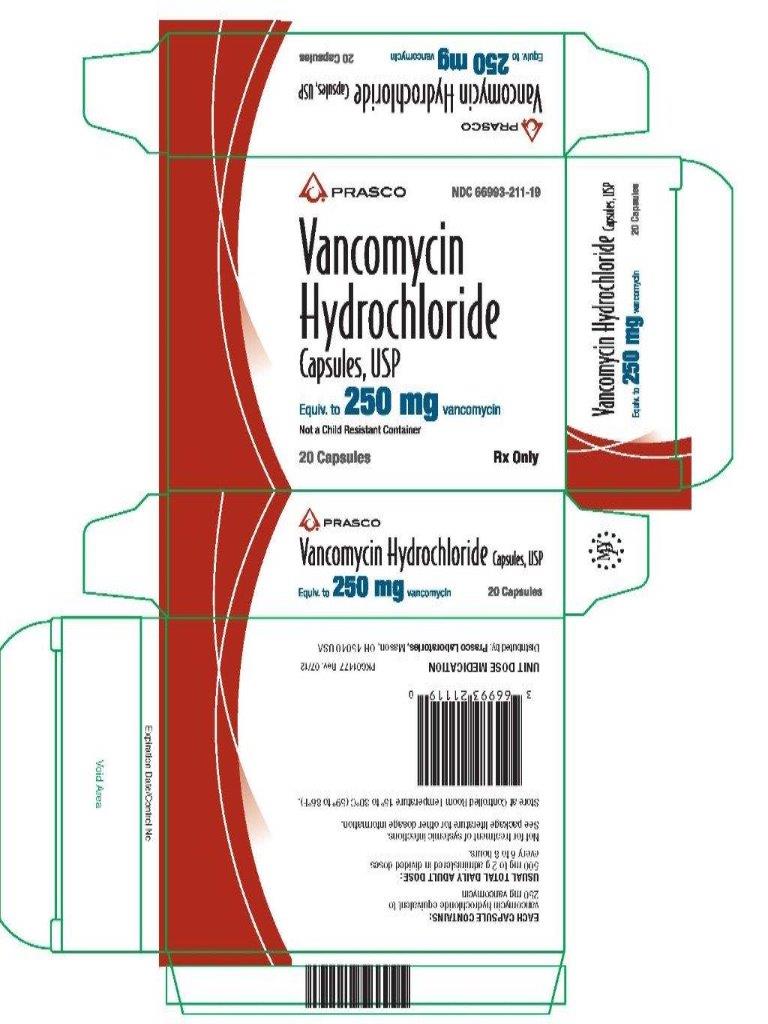
PRINCIPAL DISPLAY PANEL
Vancomycin Hydrochloride Capsules USP Equiv. to 250 mg vancomycin
NDC 66993-211-19
Rx Only
20 Capsules