NDC Code(s) : 0245-0880-89, 0245-0880-30, 0245-0880-66, 0245-0881-89, 0245-0881-30, 0245-0881-66, 0245-0882-89, 0245-0882-30, 0245-0882-66
Packager : Upsher-Smith Laboratories, Inc.
Category : HUMAN PRESCRIPTION DRUG LABEL
DEA Schedule : none
Marketing Status : New Drug Application
INGREDIENTS AND APPEARANCE
| DivigelEstradiol GEL | |||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
| DivigelEstradiol GEL | |||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
| DivigelEstradiol GEL | |||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
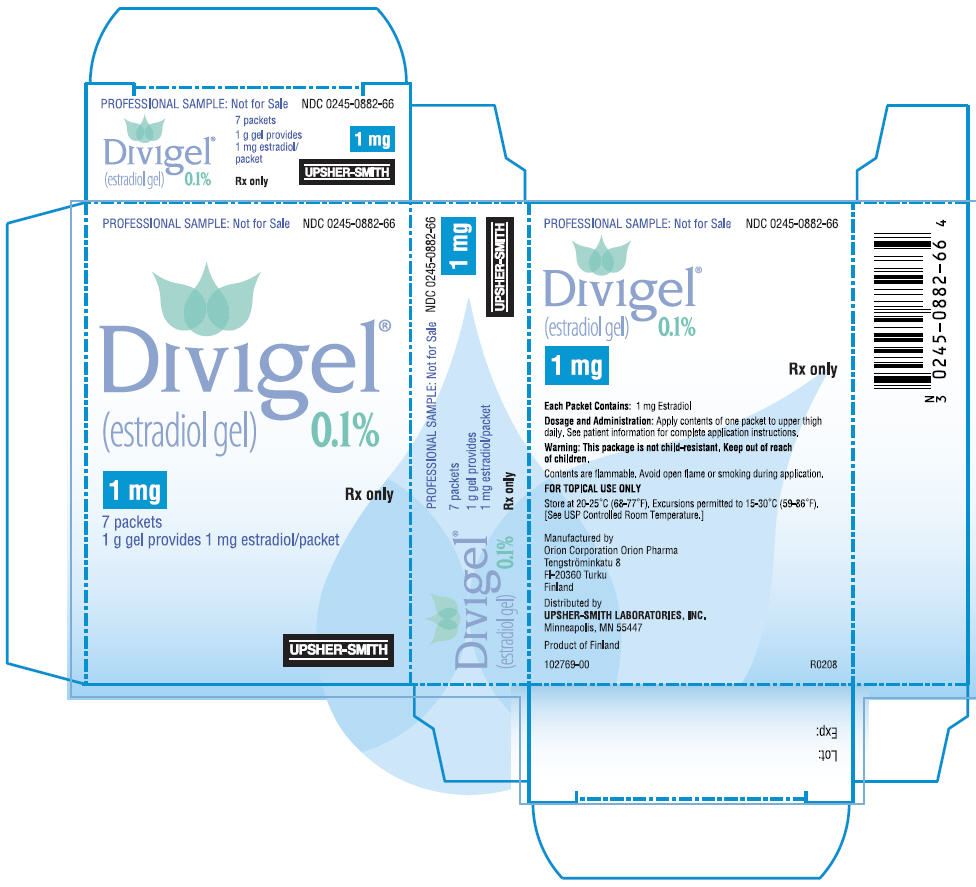
PRINCIPAL DISPLAY PANEL
PRINCIPAL DISPLAY PANEL - 0.25 mg Packet Carton
PROFESSIONAL SAMPLE: Not for Sale
NDC 0245-0880-66
Divigel
®
(estradiol gel)
0.1%
0.25 mg
Rx only
7 packets
0.25 g gel provides 0.25 mg estradiol/packet
UPSHER-SMITH
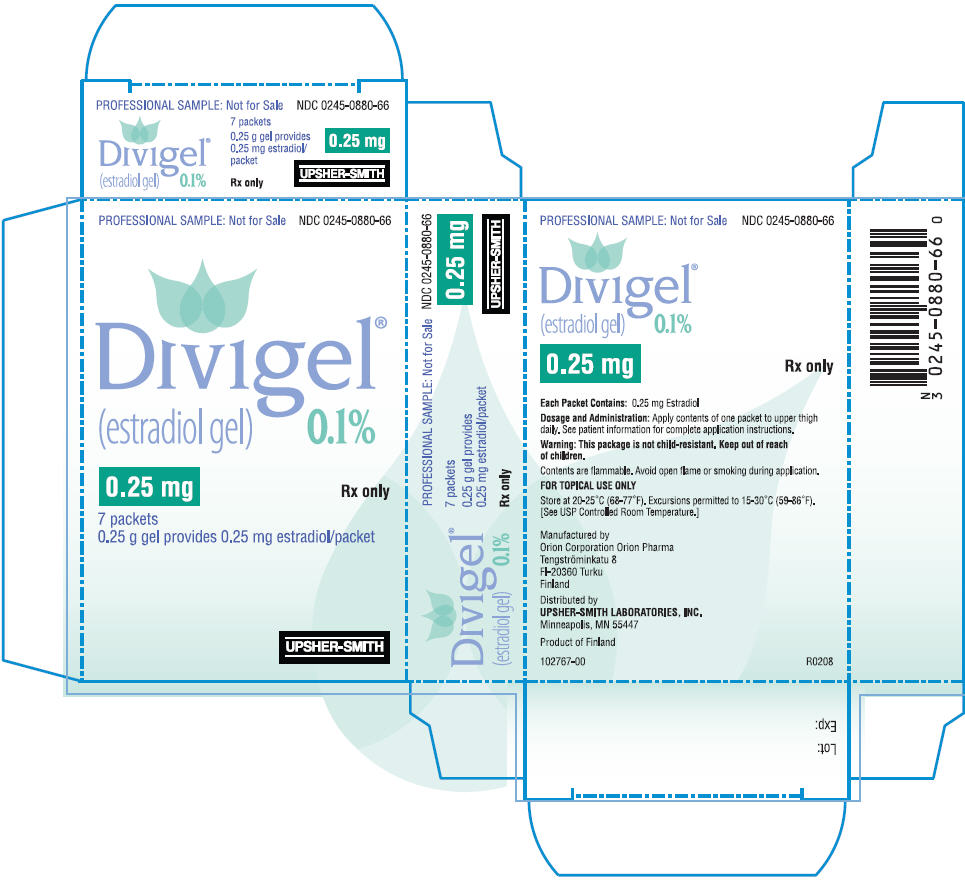
PRINCIPAL DISPLAY PANEL
PRINCIPAL DISPLAY PANEL - 0.5 mg Packet Carton
PROFESSIONAL SAMPLE: Not for Sale
NDC 0245-0881-66
Divigel
®
(estradiol gel)
0.1%
0.5 mg
Rx only
7 packets
0.5 g gel provides 0.5 mg estradiol/packet
UPSHER-SMITH
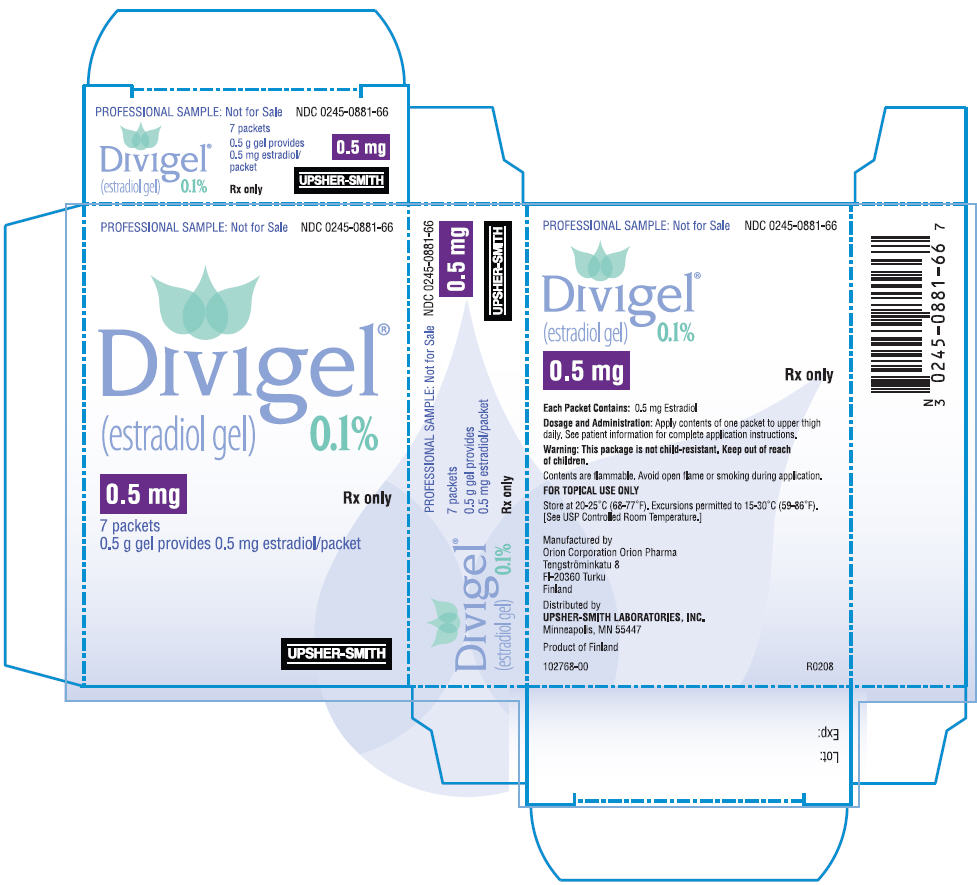
PRINCIPAL DISPLAY PANEL
PRINCIPAL DISPLAY PANEL - 1 mg Packet Carton
PROFESSIONAL SAMPLE: Not for Sale
NDC 0245-0882-66
Divigel
®
(estradiol gel)
0.1%
1 mg
Rx only
7 packets
1 g gel provides 1 mg estradiol/packet
UPSHER-SMITH