
Synopsis
Synopsis
0
API Suppliers
0
USDMF
0
CEP/COS
0
JDMF
0
EU WC
0
KDMF
0
NDC API
0
VMF
0
Listed Suppliers
0
EDQM
0
USP
0
JP
0
Others
0
Europe
0
Canada
0
Australia
0
South Africa
0
Listed Dossiers
DRUG PRODUCT COMPOSITIONS
US Medicaid
NA
Annual Reports
NA
Regulatory FDF Prices
NA
0
FDF
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
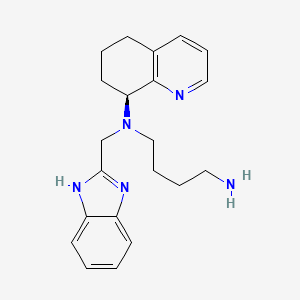

1. Amd 070
2. Amd 11070
3. Amd-070
4. Amd-11070
5. Amd070
6. Amd11070
7. Mavorixafor
8. N'-((1h-benzo(d)imidazol-2-yl)methyl)-n'-(5,6,7,8-tetrahydroquinolin-8-yl)butane-1,4-diamine
9. X 4p-001
10. X4p-001
11. X4p001
1. 558447-26-0
2. Amd-070
3. Mavorixafor
4. Amd070
5. Amd11070
6. Amd 070
7. Amd-11070
8. X4p-001
9. Mavorixafor [usan]
10. X4p001
11. Cxcr4 Inhibitor X4p-001
12. 0g9lgb5o2w
13. X 4p-001
14. Chembl518924
15. Mavorixafor (usan)
16. N'-(1h-benzimidazol-2-ylmethyl)-n'-[(8s)-5,6,7,8-tetrahydroquinolin-8-yl]butane-1,4-diamine
17. Amd-070 (amd11070)
18. Unii-0g9lgb5o2w
19. Amd 11070
20. Compound 2 [pmid: 20297846]
21. Amd-070; Mavorixafor
22. Mavorixafor [inn]
23. Mavorixafor [who-dd]
24. Gtpl8580
25. Schembl2511950
26. Dtxsid60971247
27. Chebi:138865
28. X4p-001-io
29. X4p-001-ld
30. Bcp07108
31. Ex-a2661
32. Bdbm50315305
33. Mfcd11977316
34. Nsc775637
35. Who 10724
36. Zinc33359232
37. Amd-070 Hcl 75%(w/w%)
38. Cs-0352
39. Cs-1257
40. Db05501
41. Nsc-775637
42. Ncgc00378947-01
43. Ncgc00378947-02
44. Ncgc00378947-03
45. Hy-50101
46. N~1~-[(1h-benzimidazol-2-yl)methyl]-n~1~-(5,6,7,8-tetrahydroquinolin-8-yl)butane-1,4-diamine
47. C20266
48. D11510
49. F31237
50. Q27074430
51. (s)-n-((1h-benzo[d]imidazol-2-yl)methyl)-n-(4-aminobutyl)-5,6,7,8-tetrahydroquinolin-8-amine
52. 1,4-butanediamine, N-(1h-benzimidazol-2-ylmethyl)-n-((8s)-5,6,7,8-tetrahydro-8-quinolinyl)-
53. 1,4-butanediamine, N1-(1h-benzimidazol-2-ylmethyl)-n1-((8s)-5,6,7,8-tetrahydro-8-quinolinyl)-
54. 690656-53-2
55. N'-((1h-benzo(d)imidazol-2-yl)methyl)-n'-(5,6,7,8-tetrahydroquinolin-8-yl)butane-1,4-diamine
56. N-(1h-benzimidazol-2-ylmethyl)-n-[(8s)-5,6,7,8-tetrahydroquinolin-8-yl]butane-1,4-diamine
57. N1-(1h-benzimidazol-2-ylmethyl)-n1-((s)-5,6,7,8-tetrahydroquinolin-8-yl)-butane-1,4-diamine
| Molecular Weight | 349.5 g/mol |
|---|---|
| Molecular Formula | C21H27N5 |
| XLogP3 | 2.4 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 4 |
| Rotatable Bond Count | 7 |
| Exact Mass | 349.22664588 g/mol |
| Monoisotopic Mass | 349.22664588 g/mol |
| Topological Polar Surface Area | 70.8 Ų |
| Heavy Atom Count | 26 |
| Formal Charge | 0 |
| Complexity | 431 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 1 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
Investigated for use/treatment in HIV infection.
AMD-070 is a small molecule drug candidate that belongs to a new investigational class of anti-HIV drugs known as entry (fusion) inhibitors. Approximately 76% of HIV-patients with measurable viral load are infected with a strain of virus that is resistant to one or more classes of antiretroviral agents, thus reducing treatment options. Unlike many existing HIV drugs that target the virus after it has infected a healthy cell, AMD-070 blocks the virus from entering a healthy cell, thus preventing the replication process. AMD-070 targets the CXCR4 receptor on HIV and prevents the virus from entering and infecting healthy cells. * AMD-070 is specific for the CXCR4 receptor and does not interact with any other chemokine receptors in vitro * AMD-070 strongly inhibits viral infection by all CXCR4 using virus (including virus using CXCR4 alone and/or virus using CXCR4 and CCR5) in vitro * AMD-070 is orally bioavailable in animals * Suitable PK and toxicity profile for oral dosing * AMD-070 shows additive or synergistic effects in vitro in combination with other known anti-HIV agents * AMD-070 is active against CXCR4 using HIV strains that are resistant to existing antiretroviral therapies in vitro * Potent anti-HIV activity against CXCR4-using laboratory strains and clinical isolates
Chemokine receptors expressed on the surface of immune cells are known to play a critical role in virus infection and transmission. CXCR4, and another chemokine receptor CCR5, are involved in HIV infection. The process of HIV entry begins with binding of the viral envelope glycoprotein to both the CD4 receptor and one of only two chemokine receptors, and ends with fusion of viral and cell membranes. Viral entry provides novel therapeutic targets against HIV. To date, at least 3 sub classes of HIV viral entry/fusion inhibitors have emerged: 1. CD4 binding or attachment - targets initial recognition and binding of the viral glycoprotein gp120 to the cell-surface CD4 antigen. 2. Chemokine co-receptor binding - targets binding of virus to the CCR5 or CXCR4 co-receptor. 3. Fusion Inhibition - targets the viral glycoprotein gp41 inhibiting the fusion of virus with the cell. Different strains of HIV prefer one receptor or the other, or may use either receptor to infect cells. * 35% of strains use both CXCR4 and CCR5 * 5% of strains are pure CXCR4 using * 60% of strains are pure CCR5 using * An infected individual may harbor different levels of both CXCR4 and CCR5 using virus * CXCR4 using virus independently predicts CD4 decline and HIV clinical progression and is associated with earlier mortality
 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Details:
The net proceeds from the placement for continued development towards a potential additional approval of Xolremdi (mavorixafor) in chronic neutropenia.
Lead Product(s): Mavorixafor,Inapplicable
Therapeutic Area: Hematology Brand Name: Xolremdi
Study Phase: Phase IIIProduct Type: Miscellaneous
Sponsor: Empery Asset Management
Deal Size: $85.0 million Upfront Cash: Undisclosed
Deal Type: Private Placement August 13, 2025

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Lead Product(s) : Mavorixafor,Inapplicable
Therapeutic Area : Hematology
Highest Development Status : Phase III
Partner/Sponsor/Collaborator : Empery Asset Management
Deal Size : $85.0 million
Deal Type : Private Placement
X4 Pharmaceuticals Announces Closing of Upsized $85 Million Private Placement
Details : The net proceeds from the placement for continued development towards a potential additional approval of Xolremdi (mavorixafor) in chronic neutropenia.
Product Name : Xolremdi
Product Type : Miscellaneous
Upfront Cash : Undisclosed
August 13, 2025

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Details:
X4 expects to use the net proceeds for continued development towards a potential additional approval of Xolremdi (mavorixafor) in chronic neutropenia, in addition to the commercialization of WHIM.
Lead Product(s): Mavorixafor,Inapplicable
Therapeutic Area: Hematology Brand Name: Xolremdi
Study Phase: Phase IIIProduct Type: Miscellaneous
Sponsor: Stifel
Deal Size: $60.0 million Upfront Cash: Undisclosed
Deal Type: Financing August 12, 2025

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Lead Product(s) : Mavorixafor,Inapplicable
Therapeutic Area : Hematology
Highest Development Status : Phase III
Partner/Sponsor/Collaborator : Stifel
Deal Size : $60.0 million
Deal Type : Financing
X4 Pharmaceuticals Announces $60 Million Equity Financing
Details : X4 expects to use the net proceeds for continued development towards a potential additional approval of Xolremdi (mavorixafor) in chronic neutropenia, in addition to the commercialization of WHIM.
Product Name : Xolremdi
Product Type : Miscellaneous
Upfront Cash : Undisclosed
August 12, 2025

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Details:
Mavorixafor is a Other Small Molecule drug candidate, which is currently being evaluated in phase I clinical studies for the treatment of undefined medical condition.
Lead Product(s): Mavorixafor,Carbamazepine,Efavirenz
Therapeutic Area: Undisclosed Brand Name: Undisclosed
Study Phase: Phase IProduct Type: Miscellaneous
Sponsor: Undisclosed
Deal Size: Inapplicable Upfront Cash: Inapplicable
Deal Type: Inapplicable April 06, 2025

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Lead Product(s) : Mavorixafor,Carbamazepine,Efavirenz
Therapeutic Area : Undisclosed
Highest Development Status : Phase I
Partner/Sponsor/Collaborator : Undisclosed
Deal Size : Inapplicable
Deal Type : Inapplicable
Drug-Drug Interaction Potential of Mavorixafor
Details : Mavorixafor is a Other Small Molecule drug candidate, which is currently being evaluated in phase I clinical studies for the treatment of undefined medical condition.
Product Name : Undisclosed
Product Type : Miscellaneous
Upfront Cash : Inapplicable
April 06, 2025

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Details:
Mavorixafor is a Other Small Molecule drug candidate, which is currently being evaluated in phase I clinical studies for the treatment of Hepatic Insufficiency.
Lead Product(s): Mavorixafor,Inapplicable
Therapeutic Area: Hepatology (Liver, Pancreatic, Gall Bladder) Brand Name: Undisclosed
Study Phase: Phase IProduct Type: Miscellaneous
Sponsor: Undisclosed
Deal Size: Inapplicable Upfront Cash: Inapplicable
Deal Type: Inapplicable March 05, 2025

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Lead Product(s) : Mavorixafor,Inapplicable
Therapeutic Area : Hepatology (Liver, Pancreatic, Gall Bladder)
Highest Development Status : Phase I
Partner/Sponsor/Collaborator : Undisclosed
Deal Size : Inapplicable
Deal Type : Inapplicable
Details : Mavorixafor is a Other Small Molecule drug candidate, which is currently being evaluated in phase I clinical studies for the treatment of Hepatic Insufficiency.
Product Name : Undisclosed
Product Type : Miscellaneous
Upfront Cash : Inapplicable
March 05, 2025

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Details:
Under the agreement, Taiba will be responsible for the distribution and commercialization of Xolremdi (mavorixafor), an oral, once-daily treatment for WHIM syndrome in MENA region.
Lead Product(s): Mavorixafor,Inapplicable
Therapeutic Area: Rare Diseases and Disorders Brand Name: Xolremdi
Study Phase: Approved FDFProduct Type: Miscellaneous
Sponsor: Taiba Healthcare
Deal Size: Undisclosed Upfront Cash: Undisclosed
Deal Type: Agreement February 19, 2025

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Lead Product(s) : Mavorixafor,Inapplicable
Therapeutic Area : Rare Diseases and Disorders
Highest Development Status : Approved FDF
Partner/Sponsor/Collaborator : Taiba Healthcare
Deal Size : Undisclosed
Deal Type : Agreement
X4 Pharma, Taiba Rare Ink Xolremdi Deal for WHIM in Middle East
Details : Under the agreement, Taiba will be responsible for the distribution and commercialization of Xolremdi (mavorixafor), an oral, once-daily treatment for WHIM syndrome in MENA region.
Product Name : Xolremdi
Product Type : Miscellaneous
Upfront Cash : Undisclosed
February 19, 2025

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Details:
Xolremdi (mavorixafor) is a selective CXC chemokine receptor 4 (CXCR4) antagonist, which is FDA approved for the treatment of WHIM syndrome in patients 12 years of age and older.
Lead Product(s): Mavorixafor,Inapplicable
Therapeutic Area: Rare Diseases and Disorders Brand Name: Xolremdi
Study Phase: Approved FDFProduct Type: Miscellaneous
Sponsor: Undisclosed
Deal Size: Inapplicable Upfront Cash: Inapplicable
Deal Type: Inapplicable January 24, 2025

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Lead Product(s) : Mavorixafor,Inapplicable
Therapeutic Area : Rare Diseases and Disorders
Highest Development Status : Approved FDF
Partner/Sponsor/Collaborator : Undisclosed
Deal Size : Inapplicable
Deal Type : Inapplicable
X4 Pharmaceuticals Announces EMA Validation of Mavorixafor for WHIM Syndrome
Details : Xolremdi (mavorixafor) is a selective CXC chemokine receptor 4 (CXCR4) antagonist, which is FDA approved for the treatment of WHIM syndrome in patients 12 years of age and older.
Product Name : Xolremdi
Product Type : Miscellaneous
Upfront Cash : Inapplicable
January 24, 2025

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Details:
Under the licensing agreement, Norgine will hold exclusive rights to commercialize Xolremdi (mavorixafor) in Europe, Australia, etc. It is being indicated for the treatment of WHIM syndrome.
Lead Product(s): Mavorixafor,Inapplicable
Therapeutic Area: Rare Diseases and Disorders Brand Name: Xolremdi
Study Phase: Approved FDFProduct Type: Miscellaneous
Sponsor: Norgine
Deal Size: $260.5 million Upfront Cash: $29.1 million
Deal Type: Licensing Agreement January 13, 2025

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Lead Product(s) : Mavorixafor,Inapplicable
Therapeutic Area : Rare Diseases and Disorders
Highest Development Status : Approved FDF
Partner/Sponsor/Collaborator : Norgine
Deal Size : $260.5 million
Deal Type : Licensing Agreement
X4 Pharmaceuticals, Norgine Sign License for Mavorixafor in Europe, Australia
Details : Under the licensing agreement, Norgine will hold exclusive rights to commercialize Xolremdi (mavorixafor) in Europe, Australia, etc. It is being indicated for the treatment of WHIM syndrome.
Product Name : Xolremdi
Product Type : Miscellaneous
Upfront Cash : $29.1 million
January 13, 2025

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Details:
Xolremdi (mavorixafor) is a selective CXCR4 antagonist, which is being evaluated for the treatment of idiopathic chronic neutropenic disorders who are experiencing recurrent &/or serious infections.
Lead Product(s): Mavorixafor,Inapplicable
Therapeutic Area: Hematology Brand Name: Xolremdi
Study Phase: Phase IIIProduct Type: Miscellaneous
Sponsor: Undisclosed
Deal Size: Inapplicable Upfront Cash: Inapplicable
Deal Type: Inapplicable November 13, 2024

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Lead Product(s) : Mavorixafor,Inapplicable
Therapeutic Area : Hematology
Highest Development Status : Phase III
Partner/Sponsor/Collaborator : Undisclosed
Deal Size : Inapplicable
Deal Type : Inapplicable
X4 Pharmaceuticals Announces Positive Phase 2 Mavorixafor Results
Details : Xolremdi (mavorixafor) is a selective CXCR4 antagonist, which is being evaluated for the treatment of idiopathic chronic neutropenic disorders who are experiencing recurrent &/or serious infections.
Product Name : Xolremdi
Product Type : Miscellaneous
Upfront Cash : Inapplicable
November 13, 2024

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Details:
Xolremdi (mavorixafor) is a selective CXCR4 antagonist, which is being evaluated for the treatment of idiopathic chronic neutropenic disorders who are experiencing recurrent &/or serious infections.
Lead Product(s): Mavorixafor,Inapplicable
Therapeutic Area: Hematology Brand Name: Xolremdi
Study Phase: Phase IIIProduct Type: Miscellaneous
Sponsor: Undisclosed
Deal Size: Inapplicable Upfront Cash: Inapplicable
Deal Type: Inapplicable June 27, 2024

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Lead Product(s) : Mavorixafor,Inapplicable
Therapeutic Area : Hematology
Highest Development Status : Phase III
Partner/Sponsor/Collaborator : Undisclosed
Deal Size : Inapplicable
Deal Type : Inapplicable
X4 Pharmaceuticals Reports Positive Interim Data in Chronic Neutropenia Trial
Details : Xolremdi (mavorixafor) is a selective CXCR4 antagonist, which is being evaluated for the treatment of idiopathic chronic neutropenic disorders who are experiencing recurrent &/or serious infections.
Product Name : Xolremdi
Product Type : Miscellaneous
Upfront Cash : Inapplicable
June 27, 2024

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Details:
Through the divestment, X4 sell its rare pediatric disease priority review voucher for its first approved product, Xolremdi (mavorixafor), a selective CXCR4 receptor antagonist, for WHIM syndrome.
Lead Product(s): Mavorixafor,Inapplicable
Therapeutic Area: Rare Diseases and Disorders Brand Name: Xolremdi
Study Phase: Approved FDFProduct Type: Miscellaneous
Sponsor: Undisclosed
Deal Size: $105.0 million Upfront Cash: Undisclosed
Deal Type: Divestment May 09, 2024

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Lead Product(s) : Mavorixafor,Inapplicable
Therapeutic Area : Rare Diseases and Disorders
Highest Development Status : Approved FDF
Partner/Sponsor/Collaborator : Undisclosed
Deal Size : $105.0 million
Deal Type : Divestment
X4 Pharmaceuticals Secures $125M from Voucher Sale and Loan Facility
Details : Through the divestment, X4 sell its rare pediatric disease priority review voucher for its first approved product, Xolremdi (mavorixafor), a selective CXCR4 receptor antagonist, for WHIM syndrome.
Product Name : Xolremdi
Product Type : Miscellaneous
Upfront Cash : Undisclosed
May 09, 2024

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]Market Place

Reply
18 May 2020

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
Patents & EXCLUSIVITIES
ABOUT THIS PAGE
62
PharmaCompass offers a list of Mavorixafor API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, Price,and more, enabling you to easily find the right Mavorixafor manufacturer or Mavorixafor supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Mavorixafor manufacturer or Mavorixafor supplier.
PharmaCompass also assists you with knowing the Mavorixafor API Price utilized in the formulation of products. Mavorixafor API Price is not always fixed or binding as the Mavorixafor Price is obtained through a variety of data sources. The Mavorixafor Price can also vary due to multiple factors, including market conditions, regulatory modifications, or negotiated pricing deals.
A Mavorixafor manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Mavorixafor, including repackagers and relabelers. The FDA regulates Mavorixafor manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Mavorixafor API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
A Mavorixafor supplier is an individual or a company that provides Mavorixafor active pharmaceutical ingredient (API) or Mavorixafor finished formulations upon request. The Mavorixafor suppliers may include Mavorixafor API manufacturers, exporters, distributors and traders.
Mavorixafor Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Mavorixafor GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, API price, and more, enabling you to easily find the right Mavorixafor GMP manufacturer or Mavorixafor GMP API supplier for your needs.
A Mavorixafor CoA (Certificate of Analysis) is a formal document that attests to Mavorixafor's compliance with Mavorixafor specifications and serves as a tool for batch-level quality control.
Mavorixafor CoA mostly includes findings from lab analyses of a specific batch. For each Mavorixafor CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Mavorixafor may be tested according to a variety of international standards, such as European Pharmacopoeia (Mavorixafor EP), Mavorixafor JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Mavorixafor USP).

