


1. J5oul9shw5
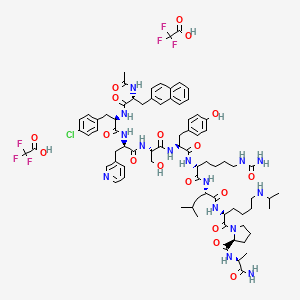
2. D-alaninamide, N-acetyl-3-(2-naphthalenyl)-d-alanyl-4-chloro-d-phenylalanyl-3-(3-pyridinyl)-d-alanyl-l-seryl-l-tyrosyl-n6-(aminocarbonyl)-d-lysyl-l-leucyl-n6-(1-methylethyl)-l-lysyl-l-prolyl-, Bis(trifluoroacetate) (salt)
3. Unii-j5oul9shw5
4. 244792-29-8
5. 500717-24-8
| Molecular Weight | 1687.2 g/mol |
|---|---|
| Molecular Formula | C78H102ClF6N15O18 |
| Hydrogen Bond Donor Count | 17 |
| Hydrogen Bond Acceptor Count | 26 |
| Rotatable Bond Count | 41 |
| Exact Mass | g/mol |
| Monoisotopic Mass | g/mol |
| Topological Polar Surface Area | 520 |
| Heavy Atom Count | 118 |
| Formal Charge | 0 |
| Complexity | 2890 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 10 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 3 |
