


1. Gs-hiv Sodium
2. Bdt58wj9we
3. 2283356-12-5
4. Unii-bdt58wj9we
5. Gs-6207-02
6. Sunlenca
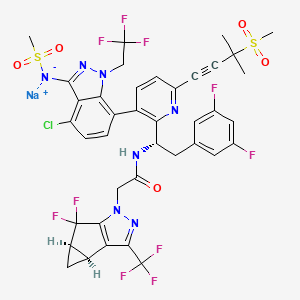
7. 1h-cyclopropa(3,4)cyclopenta(1,2-c)pyrazole-1-acetamide, N-((1s)-1-(3-(4-chloro-3-((methylsulfonyl)amino)-1-(2,2,2-trifluoroethyl)-1h-indazol-7-yl)-6-(3-methyl-3-(methylsulfonyl)-1-butyn-1-yl)-2-pyridinyl)-2-(3,5-difluorophenyl)ethyl)-5,5-difluoro-3b,4,4a,5-tetrahydro-3-(trifluoromethyl)-, Sodium Salt (1:1), (3bs,4ar)-
8. N-((1s)-1-(3-(4-chloro-3-(methanesulfonamido)-1-(2,2,2-trifluoroethyl)-1h-indazol-7-yl)-6-(3-(methanesulfonyl)-3-methylbut-1-yn-1-yl)pyridin-2-yl)-2-(3,5-difluorophenyl)ethyl)-2-((3bs,4ar)-5,5-difluoro-3-(trifluoromethyl)-3b,4,4a,5-tetrahydro1h-cyclopropa(3,4)cyclopenta(1,2-c)pyrazol-1-yl)acetamide, Sodium Salt (1:1)
9. Refchem:58517
10. Gs-hiv Na
11. Lenacapavir Sodium [usan]
12. Yeztugo
13. Sunlenca (tn)
14. Lenacapavir (sodium)
15. Lenacapavir Sodium [who-dd]
16. Orb2814823
17. Chembl4802249
18. Lenacapavir Sodium (jan/usan)
19. Schembl30612482
20. Schembl30685531
21. Ex-a5518a
22. Hy-111964a
23. D12162
| Molecular Weight | 990.3 g/mol |
|---|---|
| Molecular Formula | C39H31ClF10N7NaO5S2 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 19 |
| Rotatable Bond Count | 13 |
| Exact Mass | Da |
| Monoisotopic Mass | Da |
| Topological Polar Surface Area | 164 |
| Heavy Atom Count | 65 |
| Formal Charge | 0 |
| Complexity | 2050 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 3 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 2 |
Sunlenca injection, in combination with other antiretroviral(s), is indicated for the treatment of adults with multidrug resistant HIV 1 infection for whom it is otherwise not possible to construct a suppressive anti viral regimen (see sections 4. 2 and 5. 1). Sunlenca tablet, in combination with other antiretroviral(s), is indicated for the treatment of adults with multidrug resistant HIV 1 infection for whom it is otherwise not possible to construct a suppressive anti viral regimen, for oral loading prior to administration of long-acting lenacapavir injection (see sections 4. 2 and 5. 1).
J05AX
