

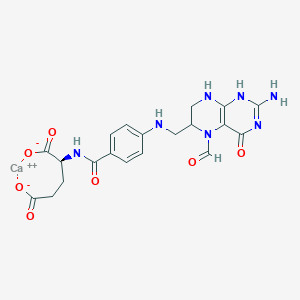

| Molecular Weight | 511.5 g/mol |
|---|---|
| Molecular Formula | C20H21CaN7O7 |
| Hydrogen Bond Donor Count | 5 |
| Hydrogen Bond Acceptor Count | 9 |
| Rotatable Bond Count | 5 |
| Exact Mass | 511.1128369 g/mol |
| Monoisotopic Mass | 511.1128369 g/mol |
| Topological Polar Surface Area | 221 A^2 |
| Heavy Atom Count | 35 |
| Formal Charge | 0 |
| Complexity | 918 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 1 |
| Undefined Atom Stereocenter Count | 1 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 2 |
| 1 of 2 | |
|---|---|
| Drug Name | LEUCOVORIN CALCIUM PRESERVATIVE FREE |
| Active Ingredient | LEUCOVORIN CALCIUM |
| Company | FRESENIUS KABI USA (Application Number: A040258); FRESENIUS KABI USA (Application Number: A040286); MYLAN LABS LTD (Application Number: A203800); SAGENT PHARMS (Application Number: A200753); SAGENT PHARMS (Application Number: A200855); WEST-WARD PHARMS INT (Application Number: A040056); WEST-WARD PHARMS INT (Application Number: A040335); WEST-WARD PHARMS INT (Application Number: A040347) |
| 2 of 2 | |
|---|---|
| Drug Name | LEUCOVORIN CALCIUM |
| Active Ingredient | LEUCOVORIN CALCIUM |
| Company | BARR (Application Number: A071198); BARR (Application Number: A071199); TEVA PHARMS USA (Application Number: A040174); TEVA PHARMS USA (Application Number: A081277); WEST-WARD PHARMS INT (Application Number: A072733); WEST-WARD PHARMS INT (Application Number: A072734); WEST-WARD PHARMS INT (Application Number: A072735); WEST-WARD PHARMS INT (Application Number: A072736); WEST-WARD PHARMS INT (Application Number: A089384); WEST-WARD PHARMS INT (Application Number: A089717) |
