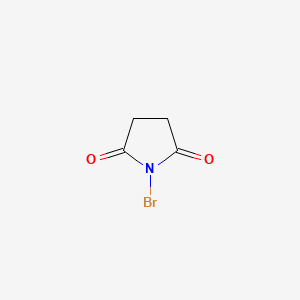

1. Bromosuccinimide
2. N Bromosuccinimide
3. Succinbromimide
1. 128-08-5
2. Succinbromimide
3. Succinbromide
4. Succinibromimide
5. N-bromosuccimide
6. 1-bromo-2,5-pyrrolidinedione
7. Nbs
8. Succinimide, N-bromo-
9. Nsc 16
10. Ccris 2899
11. 1-bromo-2,5-pyrrolidine-dione
12. Unii-k8g1f2ucjf
13. K8g1f2ucjf
14. Einecs 204-877-2
15. Nsc-16
16. Ai3-16619
17. Chebi:53174
18. Hsdb 8381
19. Ec 204-877-2
20. 204-877-2
21. 1-bromopyrrolidine-2,5-dione
22. 2,5-pyrrolidinedione, 1-bromo-
23. Bromosuccinimide
24. N-bromsuccinimide
25. N-bromo-succinimide
26. Mfcd00005510
27. N-bromosuccinimide (nbs)
28. 1-bromodihydro-1h-pyrrole-2,5-dione
29. N-bromo Succinimide
30. N-bromobutanimide
31. N-bromsuccinimid
32. N-bromosuccinimid
33. N-bromosuccmimide
34. 1-bromo-2,5-pyrrolidinedione; Bromosuccinimide; Nbs; Nsc 16; Succinbromimide;
35. N-brom-succinimide
36. N-bromo Succinimid
37. N-bromosuccinirnide
38. N-brornosuccinimide
39. Jv-bromosuccinimide
40. N -bromosuccinimide
41. N-bromosuccin-imide
42. N- Bromosuccinimide
43. N-bromo Succinic Imide
44. Schembl58
45. 2, 1-bromo-
46. Wln: T5vnvtj Be
47. N-bromosuccinic Acid Imide
48. N-bromo Succinic Acid Imide
49. Nsc16
50. 1-brompyrrolidine-2,5-dione
51. N-bromopyrrolidine-2,5-dione
52. Sgcut00108
53. N-bromosuccinimide [mi]
54. 1-bromo-pyrrolidine-2,5-dione
55. Dtxsid2038738
56. Bcp15478
57. Hy-y0775
58. To_000027
59. Stl146592
60. Akos000120001
61. Cs-w008781
62. Fb09835
63. Bp-30011
64. N-bromosuccinimide, Reagentplus(r), 99%
65. Db-025349
66. N-bromosuccinimide, For Synthesis, 98.0%
67. B0656
68. N-bromosuccinimide, Purum, >=95.0% (rt)
69. Ns00004835
70. En300-20081
71. D77695
72. N-bromosuccinimide (< 0.1% Wt Chlorine Content)
73. Q286939
74. F1908-0173
75. Inchi=1/c4h4brno2/c5-6-3(7)1-2-4(6)8/h1-2h
| Molecular Weight | 177.98 g/mol |
|---|---|
| Molecular Formula | C4H4BrNO2 |
| XLogP3 | -0.1 |
| Hydrogen Bond Donor Count | 0 |
| Hydrogen Bond Acceptor Count | 2 |
| Rotatable Bond Count | 0 |
| Exact Mass | Da |
| Monoisotopic Mass | Da |
| Topological Polar Surface Area | 37.4 |
| Heavy Atom Count | 8 |
| Formal Charge | 0 |
| Complexity | 129 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
Bromine is mainly absorbed via inhalation, but may also enter the body through dermal contact. Bromine salts can be ingested. Due to its reactivity, bromine quickly forms bromide and may be deposited in the tissues, displacing other halogens. (L626)
