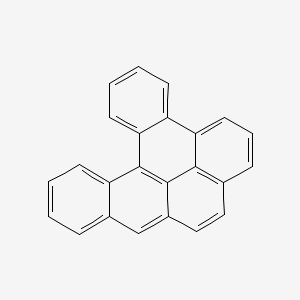

1. 1,2,3,4-dibenzopyrene
2. 1,2,9,10-dibenzopyrene
3. 4,5,6,7-dibenzpyrene
4. Ba 51-090462
5. Dibenzo(a,l)pyrene
6. Dibenzo(def,p)chrysene
1. Dibenzo[def,p]chrysene
2. 191-30-0
3. Dibenzo(a,l)pyrene
4. 1,2:3,4-dibenzopyrene
5. 1,2,9,10-dibenzopyrene
6. 4,5,6,7-dibenzpyrene
7. Db(a,l)p
8. Dibenzopyrene
9. 1,2,3,4-dibenzpyrene
10. 2,3:4,5-dibenzopyrene
11. Dibenzo(def,p)chrysene
12. Dibenzo(a,d)pyrene
13. Dibenzo(d,e,f,p)chrysene
14. Ba 51-090462
15. Naphtho[1,2,3,4-pqr]tetraphene
16. Nsc 90324
17. 1,2:9,10-dibenzopyrene
18. G3x629ve4a
19. Dibenzo[a,d]pyrene
20. Nsc-90324
21. Hexacyclo[10.10.2.02,7.08,24.015,23.017,22]tetracosa-1(23),2,4,6,8,10,12(24),13,15,17,19,21-dodecaene
22. Ccris 210
23. Hsdb 4029
24. Einecs 205-886-4
25. 2,3:4,5-dibenzpyrene
26. Brn 2054068
27. Unii-g3x629ve4a
28. Dibenz[a,l]pyrene
29. 1,4-dibenzopyrene
30. 1,3,4-dibenzpyrene
31. 4,6,7-dibenzpyrene
32. Dibenzo[d,f,p]chrysene
33. 1,9,10-dibenzopyrene
34. 4,5:6,7-dibenzpyrene
35. 2-decyldodecanoicacid
36. Dibenzo(a,l)pyrene [polycyclic Aromatic Compounds]
37. Dibenzo(a,l)pyrene [polycyclic Aromatic Hydrocarbons]
38. Dtxsid9059753
39. Chebi:35861
40. Dibenzo(a,l)pyrene [hsdb]
41. Dibenzo(a,l)pyrene [iarc]
42. Nsc90324
43. Zinc1580749
44. Dibenzo[a,l]pyrene, Vial Of 25 Mg
45. Akos024319580
46. Ft-0666414
47. C19174
48. Wln: L D66 K666 B6 2ab A & J
49. Dibenzo[a,l]pyrene 10 Microg/ml In Acetonitrile
50. Dibenzo[a,l]pyrene 10 Microg/ml In Cyclohexane
51. J-012363
52. Q26840756
53. Dibenzo[a,l]pyrene, Bcr(r) Certified Reference Material
| Molecular Weight | 302.4 g/mol |
|---|---|
| Molecular Formula | C24H14 |
| XLogP3 | 7.2 |
| Hydrogen Bond Donor Count | 0 |
| Hydrogen Bond Acceptor Count | 0 |
| Rotatable Bond Count | 0 |
| Exact Mass | 302.109550447 g/mol |
| Monoisotopic Mass | 302.109550447 g/mol |
| Topological Polar Surface Area | 0 Ų |
| Heavy Atom Count | 24 |
| Formal Charge | 0 |
| Complexity | 480 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
Carcinogens
Substances that increase the risk of NEOPLASMS in humans or animals. Both genotoxic chemicals, which affect DNA directly, and nongenotoxic chemicals, which induce neoplasms by other mechanism, are included. (See all compounds classified as Carcinogens.)
Pharmacokinetics of dibenzo(def,p)chrysene (DBC) was determined in 3 female and 6 male human volunteers following oral microdosing (29 ng, 5 nCi) of [(14)C]-DBC. This study was made possible with highly sensitive accelerator mass spectrometry (AMS), capable of detecting [(14)C]-DBC equivalents in plasma and urine following a dose considered of de minimus risk to human health. Plasma and urine were collected over 72 hr. The plasma Cmax was 68.8 +/- 44.3 fg/mL with a Tmax of 2.25 +/- 1.04 hr. Elimination occurred in two distinct phases: a rapid (a)-phase, with a /half life/ of 5.8 +/- 3.4 hr and an apparent elimination rate constant (Kel) of 0.17 +/- 0.12 fg/hr, followed by a slower (beta)-phase, with a /half life/ of 41.3 +/- 29.8 hr and an apparent Kel of 0.03 +/- 0.02 fg/hr. In spite of the high degree of hydrophobicity (log Kow of 7.4), DBC was eliminated rapidly in humans, as are most PAHs in animals, compared to other hydrophobic persistent organic pollutants such as, DDT, PCBs and TCDD. Preliminary examination utilizing a new UHPLC-AMS interface, suggests the presence of polar metabolites in plasma as early as 45 min following dosing. This is the first in vivo data set describing pharmacokinetics in humans of a high molecular weight PAH and should be a valuable addition to risk assessment paradigms.
PMID:25418912 Full text: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4303324 Madeen E et al; Chem Res Toxicol 28 (1): 126-34 (2015)
... Like other polycyclic aromatic hydrocarbons, dibenzol(a,l)pyrene requires metabolic activation to exert its mutagenic and/or carcinogenic activity. In the human mammary carcinoma cell line MCF-7, dibenzol(a,l)pyrene is stereoselectively metabolized to the (-)-anti- and (+)-3-syn-dibenzol(a,l)pyrene-11,12-diol 13,14-epoxides (DB(a,l)PDE) which both bind extensively to deoxyadenosine residues in DNA. To further characterize the underlying mechanism of its strong carcinogenicity, the relationship between DNA binding and mutagenicity of dibenzol(a,l)pyrene was determined. Racemic dibenzol(a,l)pyrene-11,12-dihydrodiol and the two individual (+)- and (-)-enantiomers, the metabolic precursors of the stereoisomeric fjord region dihydrodiol epoxides, were also investigated. Induction of mutations at the HPRT locus was measured in a MCF-7 cell-mediated Chinese hamster V79 cell mutation assay. The parent hydrocarbon, (+ or -)-dibenzol(a,l)pyrene-11,12-dihydrodiol, and (-)-dibenzol(a,l)pyrene-11,12-dihydrodiol were highly mutagenic under the assay conditions. In contrast, (+)-dibenzol(a,l)pyrene-(llS,12S)-dihydrodiol was not mutagenic using MCF-7 cells as the metabolic activating system. Analysis of DNA adducts in the same experiments revealed that MCF-7 cells treated with (-)-dibenzol(a,l)pyrene-11,12-dihydrodiol formed exclusively (-)-anti-DB(a,l)PDE adducts whereas cells treated with (+)-dibenzol(a,l)pyrene-11,12-dihydrodiol did not contain detectable levels of DNA adducts. These results suggest that specific cytochrome p450 enzymes may have high stereoselectivity for activation of the two dibenzol(a,l)pyrene-11,12-dihydrodiol enantiomers, and this may play an important role in the metabolic activation of the strong carcinogen dibenzol(a,l)pyrene in human cells.
PMID:9208176 Ralston SL et al; Chemical Research in Toxicology 10 (6): 687-93 (1997)
Metabolic activation of the racemic benzo[c]chrysene-trans-9,10-, benzo[g]chrysene-trans-11,12- and dibenzo[a,l]pyrene-trans-11,12-dihydrodiols to fjord region syn- and anti-dihydrodiol epoxides by microsomes of Aroclor 1254-treated Sprague-Dawley rats has been examined. Since the fjord region dihydrodiol epoxides were hydrolytically unstable under the experimental conditions, their enzymatic formation was determined by analyzing the tetraols as their products of acidic hydrolysis upon addition of perchloric acid. The various stereoisomeric tetraols formed were separated by HPLC and identified by co-chromatography with authentic tetraols, which had been prepared by acidic hydrolysis of synthetically available syn- and anti-dihydrodiol epoxides and characterized by NMR and UV spectroscopy. Under standardized conditions the acidic hydrolysis of syn-dihydrodiol epoxides of benzo[c]chrysene, benzo[g]chrysene and dibenzo[a,l]pyrene resulted in the formation of two tetraols with cis/trans ratios of 81:19, 77:23 and 80:20, respectively, whereas the anti-dihydrodiol epoxides underwent almost exclusively trans hydrolysis. The proportion of the stereoisomeric tetraols obtained from microsomal incubations indicates that all three dihydrodiols are predominantly oxidized at the adjacent olefinic double bond to the anti-diastereomers of the corresponding fjord region dihydrodiol epoxides accounting for 4-35% of the ethyl acetate-extractable metabolites. To allow quantitative assessment of the metabolites (3)H-labeled trans-dihydrodiols were synthesized by reduction of the corresponding o-quinones with sodium borotritide. Metabolic conversion of benzo[c]chrysene-trans-9,10- and dibenzo[a,l]pyrene-trans-11,12-dihydrodiol by rat liver microsomes were in a similar low range during the first 10 min of incubation (6.2 +/- 1.2 and 3.4 +/- 1.0 nmol substrate/nmol cytochrome P450/10 min, respectively), whereas the conversion of benzo[g]chrysene-trans-11,12-dihydrodiol was much higher (20.6 +/- 2.2 nmol substrate/nmol cytochrome P450/10 min). Given the strong intrinsic mutagenic and carcinogenic activity of the fjord region dihydrodiol epoxides, our data indicate that their formation, even at a relatively low level, may contribute significantly to the biological activity of the parent hydrocarbons.
PMID:9600349 Luch A et al; Carcinogenesis 19 (4): 639-48 (1998)
Dibenzo(a,l)pyrene (DB(a,l)P), an environmental hydrocarbon and very potent carcinogen in rodent bioassays, could be activated to DNA-binding intermediates in cells through formation of three different regioisomeric bay- or fjord-region diol-epoxides or other more highly oxidized metabolites. The mechanism of metabolic activation of dibenzo(a,l)pyrene in the human mammary carcinoma cell line MCF-7 was elucidated by analyzing the dibenzo(a,l)pyrene-DNA adducts formed by (35)S-phosphorothioate post labeling, immobilized boronate chromatography, and high-performance liquid chromatography. Six dibenzo(a,l)pyrene-DNA adducts were detected. Comparison with those formed in cells by dibenzo(a,l)pyrene-11,12-diol and by reaction of DNA with syn- and anti-(benzylic hydroxyl and epoxide oxygen cis and trans, respectively) dibenzo(a,l)pyrene-11,12-diol-13,14-epoxide (DB(a,l)PDE) demonstrated that all dibenzo(a,l)pyrene-DNA adducts in MCF-7 cells were formed by these diolepoxide isomers. Cellular DNA contained large amounts of two syn- and one anti-DB(a,l)PDE-DNA adducts and small amounts of one syn- and two anti-dibenzo(a,l)pyrene-11,12-diol-13,14-epoxide-DNA adducts. The ability of human cells to activate dibenzo(a,l)pyrene to its fjord-region 11,12-diol 13,14-epoxides suggests that environmental exposure to dibenzo(a,l)pyrene could pose a risk for humans.
PMID:8313376 Ralston SL et al; Cancer Research 54 (4): 887-90 (1994)
The metabolic activation of dibenzo[a,l]pyrene (DB[a,l]P) ... to the 11,12-dihydrodiol, a precursor of the ultimate carcinogens, the 11,12-diol-13,14-epoxides, was investigated using eleven human recombinant cytochrome p450s, as well as human lung and liver microsomes. Of all human p450s, lAl was the most active in the metabolism of dibenzo[a,l]pyrene (310 pmol/min, nmol p450) and had 5-23-fold higher catalytic activity than other p450s examined. The order of activity in the formation of the 11,12-dihydrodiol was as follows: lAl (116 pmol/min, nmol p450) > 2C9 (29) > lA2 (22) > 2B6 (18) > 3A4 (16) > others (< or = 5). The Km of lAl for dibenzo[a,l]pyrene and Vmax for the formation of 11,12-dihydrodiol were 3.9 uM and 0.13/min, respectively. Liver microsomes from 14 individuals were shown to metabolize dibenzo[a,l]pyrene and the rates for production of 11,12-dihydrodiol ranged from 4 to 71 pmol/min, nmol p450. Lung microsomes from six organ donors formed the 11,12-dihydrodiol at a rate from 0.1 to 1.3 pmol/min, mg of microsomal protein. These findings describe the potential of individual p450s present in liver and lung to contribute to the metabolic activation and the carcinogenicity of dibenzo[a,l]pyrene.
PMID:8968059 Shou M et al; Carcinogenesis 17 (11): 2429-33 (1996)
For more Metabolism/Metabolites (Complete) data for Dibenzo(a,l)pyrene (9 total), please visit the HSDB record page.
Dibenzo[a,l]pyrene has known human metabolites that include 6-oxaheptacyclo[11.10.2.02,8.05,7.010,24.017,25.018,23]pentacosa-1(24),2(8),3,5(7),9,11,13(25),14,16,18,20,22-dodecaene.
S73 | METXBIODB | Metabolite Reaction Database from BioTransformer | DOI:10.5281/zenodo.4056560
Pharmacokinetics of dibenzo(def,p)chrysene (DBC) was determined in 3 female and 6 male human volunteers following oral microdosing (29 ng, 5 nCi) of [(14)C]-DBC. This study was made possible with highly sensitive accelerator mass spectrometry (AMS), capable of detecting [(14)C]-DBC equivalents in plasma and urine following a dose considered of de minimus risk to human health. Plasma and urine were collected over 72 hr. The plasma Cmax was 68.8 +/- 44.3 fg/mL with a Tmax of 2.25 +/- 1.04 hr. Elimination occurred in two distinct phases: a rapid (a)-phase, with a /half life/ of 5.8 +/- 3.4 hr and an apparent elimination rate constant (Kel) of 0.17 +/- 0.12 fg/hr, followed by a slower (beta)-phase, with a /half life/ of 41.3 +/- 29.8 hr and an apparent Kel of 0.03 +/- 0.02 fg/hr. ...
PMID:25418912 Full text: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4303324 Madeen E et al; Chem Res Toxicol 28 (1): 126-34 (2015)