
Synopsis
Synopsis
0
CEP/COS
0
JDMF
0
EU WC
0
KDMF
0
NDC API
0
VMF
0
Listed Suppliers
0
Europe
0
Canada
0
Australia
0
South Africa
0
Listed Dossiers
DRUG PRODUCT COMPOSITIONS
0
US Patents
0
US Exclusivities
0
Health Canada Patents
US Medicaid
NA
Annual Reports
NA
Regulatory FDF Prices
NA
0
API
0
FDF
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
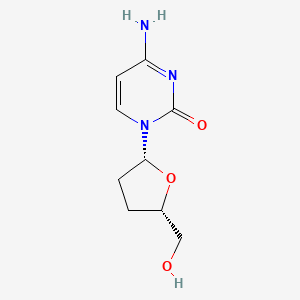

1. 2',3' Dideoxycytidine
2. 2',3'-dideoxycytidine
3. Ddc (antiviral)
4. Dideoxycytidine
5. Hivid
6. Hivid Roche
7. Nsc 606170
8. Nsc-606170
9. Nsc606170
1. Dideoxycytidine
2. 7481-89-2
3. 2',3'-dideoxycytidine
4. Ddcyd
5. Hivid
6. Cytidine, 2',3'-dideoxy-
7. Ddc
8. Zalcitibine
9. 4-amino-1-((2r,5s)-5-(hydroxymethyl)tetrahydrofuran-2-yl)pyrimidin-2(1h)-one
10. Nsc 606170
11. Ro 24-2027/000
12. Ro-24-2027/000
13. Nsc-606170
14. Chembl853
15. 6l3xt8cb3i
16. 4-amino-1-[(2r,5s)-5-(hydroxymethyl)tetrahydrofuran-2-yl]pyrimidin-2(1h)-one
17. Chebi:10101
18. Ncgc00090705-08
19. Ro-242027000
20. Ro-24-2027000
21. Dsstox_cid_3747
22. 4-amino-1-[(2r,5s)-5-(hydroxymethyl)oxolan-2-yl]-1,2-dihydropyrimidin-2-one
23. Dsstox_rid_77182
24. Dsstox_gsid_23747
25. 2,3-dideoxycytidine
26. Nsc606170
27. 4-amino-1-[(2r,5s)-5-(hydroxymethyl)tetrahydrofuran-2-yl]pyrimidin-2-one
28. Smr000058253
29. Ccris 692
30. Hivid(tm)
31. Hivid (tn)
32. Hsdb 7156
33. Interferon Ad + Ddc
34. Ddc & Gm-csf
35. Ddc & Scd4
36. Pc-sod & Ddc
37. Ddc (ddc)
38. Unii-6l3xt8cb3i
39. Brn 0654956
40. Zalcitabine (jan/usp/inn)
41. Ds-4152 & Ddc
42. 1-(2,3-dideoxy-beta-d-ribofuranosyl)cytosine
43. Ddc & Np (from Phca Or Hsa)
44. Sri-7707
45. Zalcitabine [usan:usp:inn:ban]
46. Cas-7481-89-2
47. Zalcitabine- Bio-x
48. Mfcd00012188
49. Ks-1130
50. Ddc & Ifn.alpha.
51. .beta.-d-ddc
52. Dideoxycytidine (ddc)
53. Zalcitabine [mi]
54. 4-amino-1-[(2r,5s)-5-(hydroxymethyl)oxolan-2-yl]pyrimidin-2-one
55. Prestwick0_001037
56. Prestwick1_001037
57. Prestwick2_001037
58. Prestwick3_001037
59. Ddc & Interferon.alpha.
60. Zalcitabine [inn]
61. Zalcitabine [jan]
62. 2', 3'-dideoxycytidine
63. Zalcitabine [hsdb]
64. Zalcitabine [iarc]
65. Zalcitabine [usan]
66. Bmse000712
67. Upcmld-dp115
68. D 5782
69. Zalcitabine [vandf]
70. Schembl3598
71. Timtec1_004969
72. Zalcitabine [mart.]
73. Lopac0_000360
74. Bspbio_001253
75. Zalcitabine [usp-rs]
76. Zalcitabine [who-dd]
77. 3'-azido-3'-deoxythymidine/2',3'-dideoxycytidine
78. 5-25-14-00313 (beilstein Handbook Reference)
79. Mls000069636
80. Mls000759540
81. Mls001055363
82. Mls001424210
83. Mls006011951
84. Spbio_003104
85. Bpbio1_001378
86. Gtpl4828
87. Dtxsid0023747
88. Upcmld-dp115:001
89. Zinc39906
90. 2',3'-dideoxycytidine & Scd4(soluble Recombinant Protein)
91. Zalcitabine [orange Book]
92. Hms1548b19
93. Hms1571o15
94. Hms2051h18
95. Hms2090c12
96. Hms2098o15
97. Hms2236n08
98. Hms3261g21
99. Hms3715o15
100. Pharmakon1600-01502360
101. Zalcitabine [usp Impurity]
102. .beta.-d-2',3'-dideoxycytidine
103. Zalcitabine, 2'3'-dideoxycytidine
104. Bcp13878
105. Tox21_113491
106. Tox21_201655
107. Tox21_303169
108. Tox21_500360
109. Ac-824
110. Bdbm50145605
111. Lecithinized Superoxide Dismutase & .beta.-d-2',3'-dideoxycytidine
112. Nsc759655
113. S1719
114. Sulfated Polysaccharide-peptidoglycan Ds-4152 & 2',3'-dideoxycytidine
115. Akos015854844
116. Akos015894505
117. Tox21_113491_1
118. Ccg-101050
119. Cs-1110
120. Db00943
121. Lp00360
122. Nc00300
123. Nsc-759655
124. Sdccgsbi-0050348.p002
125. 2',3'-dideoxycytidine & Nanoparticles (from Human Serum Albumin Or Polyhexylcyanoacrylate)
126. Sri-7707-13
127. Sri-7707-14
128. Sri-7707_15
129. Sri-7707_17
130. Ncgc00090705-01
131. Ncgc00090705-02
132. Ncgc00090705-03
133. Ncgc00090705-05
134. Ncgc00090705-06
135. Ncgc00090705-07
136. Ncgc00090705-09
137. Ncgc00090705-10
138. Ncgc00090705-11
139. Ncgc00090705-13
140. Ncgc00090705-15
141. Ncgc00090705-24
142. Ncgc00090705-25
143. Ncgc00179242-01
144. Ncgc00257202-01
145. Ncgc00259204-01
146. Ncgc00261045-01
147. Bd164564
148. Hy-17392
149. Ddc;dideoxycytidine;2',3'-dideoxycytidine
150. 2',3'-dideoxycytidine & Interferon.alpha.
151. 2',3'-dideoxycytidine, >=98% (hplc)
152. Db-019728
153. D3581
154. Eu-0100360
155. Sw197364-4
156. 2',3'-dideoxycytidine, >=99.0% (hplc)
157. C07207
158. C76390
159. D00412
160. Cytidine, 2',3'-dideoxy- & Interferon.alpha.
161. 481d892
162. A838234
163. Sr-01000075822
164. Sr-01000736919
165. Zalcitabine (dideoxycytidine,ddc) [vandf]
166. J-700276
167. Q-201941
168. Q2344582
169. Sr-01000075822-1
170. Sr-01000736919-5
171. Cytidine, 2',3'-dideoxy- & Colony-stimulating Factor
172. Ro-242027000/ro-24-2027-000
173. Z1550648753
174. Zalcitabine, United States Pharmacopeia (usp) Reference Standard
175. 4-amino-1-(5-hydroxymethyl-tetrahydro-furan-2-yl)-1h-pyrimidin-2-one
176. .beta.-d-2',3'-dideoxycytidine & Granulocyte-macrophage Colony-stimulating Factor
| Molecular Weight | 211.22 g/mol |
|---|---|
| Molecular Formula | C9H13N3O3 |
| XLogP3 | -1.3 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 3 |
| Rotatable Bond Count | 2 |
| Exact Mass | 211.09569129 g/mol |
| Monoisotopic Mass | 211.09569129 g/mol |
| Topological Polar Surface Area | 88.2 Ų |
| Heavy Atom Count | 15 |
| Formal Charge | 0 |
| Complexity | 327 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 2 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
Zalcitabine is indicated in combination with zidovudine, for the treatment of HIV infection in patients with limited prior exposure (< 3 months) to zidovudine. Zalcitabine is also indicated, in combination with antiretroviral protease inhibitors, for the treatment of HIV infection. /Included in US product labeling/
MICROMEDEX Thomson Health Care. USPDI - Drug Information for the Health Care Professional. 23rd ed. Volume 1. MICROMEDEX Thomson Health Care, Greenwood Village, CO. 2003. Content Reviewed and Approved by the U.S. Pharmacopeial Convention, Inc., p. 2780
Zalcitabine is indicated as a monotherapy for the treatment of advanced HIV infection in patients who are intolerant of, or who have disease progression while receiving, alternative antiretroviral therapy. /Included in US product labeling/
MICROMEDEX Thomson Health Care. USPDI - Drug Information for the Health Care Professional. 23rd ed. Volume 1. MICROMEDEX Thomson Health Care, Greenwood Village, CO. 2003. Content Reviewed and Approved by the U.S. Pharmacopeial Convention, Inc., p. 2780
Peripheral neuropathy occurring during zalcitabine therapy generally is a sensorimotor neuropathy characterized initially as numbness and burning dysesthesia involving the distal extremities. When manifestations of peripheral neuropathy occur, they often become evident during the first 7-24 weeks of therapy and involve pain and discomfort in the feet, followed several weeks later by a burning dysesthesia in the same area. Clinically, patients may have diminished light touch, pin prick, temperature, and vibration sensations in the area of the feet and up to the midcalf; ankle deep-tendon reflexes may be decreased or absent. If the drug is not discontinued when these initial manifestations appear, sharp shooting pains or severe continuous burning pain may occur and can progress to severe pain requiring opiate analgesics and is potentially irreversible. Discomfort may be severe enough to cause gait disturbances. In some patients, manifestations of peripheral neuropathy progress to include the hands and are sometimes described as occurring in a stocking-and-glove pattern. Nerve biopsy and electrophysical studies indicate that axonal degeneration may occur.
McEvoy, G.K. (ed.). American Hospital Formulary Service - Drug Information 2003. Bethesda, MD: American Society of Health-System Pharmacists, Inc. 2003 (Plus Supplements)., p. 716
Lactic acidosis and severe hepatomegaly with steatosis (sometimes fatal) have been reported rarely in patients receiving zalcitabine and also have been reported in patients receiving other nucleoside reverse transcriptase agents. Most reported cases have involved women; obesity and long-term therapy with a nucleoside antiretroviral also may be risk factors.
McEvoy, G.K. (ed.). American Hospital Formulary Service - Drug Information 2003. Bethesda, MD: American Society of Health-System Pharmacists, Inc. 2003 (Plus Supplements)., p. 716
Moderate to severe peripheral neuropathy has been reported in 3-35% of patients receiving oral zalcitabine at usual dosage (0.75 mg every 8 hours) either alone or in conjunction with zidovudine. Peripheral neuropathy occurs more frequently in patients with advanced HIV infection than in those with less advanced disease.
McEvoy, G.K. (ed.). American Hospital Formulary Service - Drug Information 2003. Bethesda, MD: American Society of Health-System Pharmacists, Inc. 2003 (Plus Supplements)., p. 716
The duration of clinical benefit from antiretroviral therapy may be limited. Alterations in antiretroviral therapy should be considered if disease progression occurs during treatment with zalcitabine.
MICROMEDEX Thomson Health Care. USPDI - Drug Information for the Health Care Professional. 23rd ed. Volume 1. MICROMEDEX Thomson Health Care, Greenwood Village, CO. 2003. Content Reviewed and Approved by the U.S. Pharmacopeial Convention, Inc., p. 2780
For more Drug Warnings (Complete) data for ZALCITABINE (33 total), please visit the HSDB record page.
For the treatment of Human immunovirus (HIV) infections in conjunction with other antivirals.
FDA Label
Zalcitabine is an analog of 2'-deoxycytidine that is pharmacologically related to but structurally different from other nucleotide reverse transcriptase inhibitors (NRTIs). Zalcitabine inhibits the activity of HIV-1 reverse transcriptase (RT) both by competing with the natural substrate dGTP and by its incorporation into viral DNA.
Anti-HIV Agents
Agents used to treat AIDS and/or stop the spread of the HIV infection. These do not include drugs used to treat symptoms or opportunistic infections associated with AIDS. (See all compounds classified as Anti-HIV Agents.)
Antimetabolites
Drugs that are chemically similar to naturally occurring metabolites, but differ enough to interfere with normal metabolic pathways. (From AMA Drug Evaluations Annual, 1994, p2033) (See all compounds classified as Antimetabolites.)
Reverse Transcriptase Inhibitors
Inhibitors of reverse transcriptase (RNA-DIRECTED DNA POLYMERASE), an enzyme that synthesizes DNA on an RNA template. (See all compounds classified as Reverse Transcriptase Inhibitors.)
J - Antiinfectives for systemic use
J05 - Antivirals for systemic use
J05A - Direct acting antivirals
J05AF - Nucleoside and nucleotide reverse transcriptase inhibitors
J05AF03 - Zalcitabine
Absorption
Bioavailability is over 80% following oral administration.
Route of Elimination
Renal excretion of unchanged drug appears to be the primary route of elimination, accounting for approximately 80% of an intravenous dose and 60% of an orally administered dose within 24 hours after dosing (n=19). Renal clearance exceeds glomerular filtration rate suggesting renal tubular secretion contributes to the elimination of zalcitabine by the kidneys.
Volume of Distribution
0.304 to 0.734 L/kg
Clearance
285 mL/min [HIV-infected patients receiving 1.5 mg IV infusion for 1 hour]
Zalcitabine is eliminated principally in urine. Approximately 60-70% of an oral dose or 75-80% of an IV dose of the drug is eliminated unchanged in urine within 24 hours; approximately 10% of an orally administered dose is eliminated in feces as unchanged drug and ddU.
McEvoy, G.K. (ed.). American Hospital Formulary Service - Drug Information 2003. Bethesda, MD: American Society of Health-System Pharmacists, Inc. 2003 (Plus Supplements)., p. 722
Only limited information is available on the pharmacokinetics of zalcitabine in children; however, oral bioavailability of the drug appears to be lower in children than in adults. In a study in children 6 months to 13 years of age with symptomatic HIV infection, oral bioavailability of zalcitabine averaged 54% (range: 29-100%). Following oral administration of 0.03- or 0.04-mg/kg doses of zalcitabine every 6 hours in these children, peak plasma concentrations were 23.1-52.5 or 31.5-63 ng/mL, respectively. Following IV administration over 1 hour of 0.03- or 0.04-mg/kg doses of zalcitabine every 6 hours in these children, peak plasma concentrations were 39.9-44.1 or 79.8-165.9 ng/mL, respectively.
McEvoy, G.K. (ed.). American Hospital Formulary Service - Drug Information 2003. Bethesda, MD: American Society of Health-System Pharmacists, Inc. 2003 (Plus Supplements)., p. 721
Pregnant rhesus monkeys (Macaca mulatta) that were near term (146 days) received radiolabelled zalcitabine as a bolus dose of 0.6 mg/kg body weight via the radial vein. During a 3 hour sampling of both the mother and the fetus, the fetal:maternal ratio of the integrated area under the curve of plasma concentration-time was 0.32 +/- 0.02, and the fetal tissues were found to contain zalcitabine (0.05-0.8 umol/l equivalents) and zalcitabine monophosphate (0.008-0.09 nmol/g).
IARC. Monographs on the Evaluation of the Carcinogenic Risk of Chemicals to Humans. Geneva: World Health Organization, International Agency for Research on Cancer, 1972-PRESENT. (Multivolume work). Available at: https://monographs.iarc.fr/ENG/Classification/index.php, p. V76 135 (2000)
About 10% of the drug appears in the feces and approximately 75% is excreted unchanged in the urine, suggesting that renal integrity is important for clearance.
IARC. Monographs on the Evaluation of the Carcinogenic Risk of Chemicals to Humans. Geneva: World Health Organization, International Agency for Research on Cancer, 1972-PRESENT. (Multivolume work). Available at: https://monographs.iarc.fr/ENG/Classification/index.php, p. V76 134 (2000)
For more Absorption, Distribution and Excretion (Complete) data for ZALCITABINE (9 total), please visit the HSDB record page.
Zalcitabine is not reported to undergo significant hepatic metabolism; it is mainly phosphorylated intracellularly to zalcitabine triphosphate, the active substrate for HIV-reverse transcriptase. The concentration of zalcitabine triphosphate remains low for quantitative detection and measurement in the plasma following administration of therapeutic doses.
The metabolic fate of zalcitabine has not been fully evaluated in humans. Dideoxyuridine (ddU) is the principal metabolite that has been identified for zalcitabine. In both urine and feces, ddU accounts for less than 15% of an oral dose of zalcitabine. Zalcitabine is not metabolized substantially in the liver and is relatively resistant to human cytidine deaminase, a catabolic enzyme that degrades many cytidine derivatives. Because zalcitabine is relatively resistant to degradation by cytidine deaminase, the drug is stable in plasma and resistant to first-pass metabolism in the liver.
McEvoy, G.K. (ed.). American Hospital Formulary Service - Drug Information 2003. Bethesda, MD: American Society of Health-System Pharmacists, Inc. 2003 (Plus Supplements)., p. 722
Hepatic metabolites have not been observed. The antiviral action of zalcitabine, like that of zidovudine and didanosine, is dependent on phosphorylation and incorporation into DNA. The first step is the formation of zalcitabine monophosphate by the enzyme 2'-deoxycytidine kinase, which is followed by formation of the diphosphate and triphosphate metabolites through the action of the cytosine monophosphate kinase and nucleotide diphosphate kinase enzymes, respectively.
IARC. Monographs on the Evaluation of the Carcinogenic Risk of Chemicals to Humans. Geneva: World Health Organization, International Agency for Research on Cancer, 1972-PRESENT. (Multivolume work). Available at: https://monographs.iarc.fr/ENG/Classification/index.php, p. V76 134 (2000)
2 hours
Mean elimination half-life was 1.4 hr (range, 1.0-3.5) /in 23 mildly symptomatic human immunodeficiency virus-infected children (mean age, 4.2 years)/.
PMID:7594705 Chadwick EG et al; J Infect Dis 172 (6): 1475-9 (1995)
... The plasma half-time is reported to be 1-2.7 hours.
IARC. Monographs on the Evaluation of the Carcinogenic Risk of Chemicals to Humans. Geneva: World Health Organization, International Agency for Research on Cancer, 1972-PRESENT. (Multivolume work). Available at: https://monographs.iarc.fr/ENG/Classification/index.php, p. V76 134 (2000)
Zalcitabine is a nucleoside reverse transcriptase inhibitor (NRTI) with activity against Human Immunodeficiency Virus Type 1 (HIV-1). Within cells, zalcitabine is converted to its active metabolite, dideoxycytidine 5'-triphosphate (ddCTP), by the sequential action of cellular enzymes. ddCTP interferes with viral RNA-directed DNA polymerase (reverse transcriptase) by competing for utilization of the natural substrate deoxycytidine 5'-triphosphate (dCTP), as well as incorpating into viral DNA. Due to it's lack of a 3'-OH group, the formation of a 5' to 3' phosphodiester linkage that is necessary for DNA chain elongation is inhibited, thus leading to the termination of viral DNA growth.
Zalcitabine enters the cell through carrier-mediated and non-carrier-mediated mechanisms. It is first phosphorylated by deoxycytidine kinase and further by cellular kinases to its active metabolite, dideoxycytidine 5'-triphosphate. Unlike other nucleoside analogs, zalcitabine is most efficiently triphosphorylated in resting peripheral blood mononuclear cells. The triphosphate terminates viral DNA elongation. Zalcitabine decrease the intracellular pool of deoxycytidine triphosphate and binds somewhat to host beta and gamma DNA polymerases.
Goldfrank, L.R., Flomenbaum, N.E., Lewin, N.A., Weisman, R.S., Howland, M.A., Hoffman, R.S., Goldfrank's Toxicologic Emergencies 6th Ed. (1998)., McGraw-Hill, New York, N.Y., p. 1358
REF. STANDARDS & IMPURITIES

ABOUT THIS PAGE
36
PharmaCompass offers a list of Zalcitabine API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Zalcitabine manufacturer or Zalcitabine supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Zalcitabine manufacturer or Zalcitabine supplier.
A Zalcitabine manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Zalcitabine, including repackagers and relabelers. The FDA regulates Zalcitabine manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Zalcitabine API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
A Zalcitabine supplier is an individual or a company that provides Zalcitabine active pharmaceutical ingredient (API) or Zalcitabine finished formulations upon request. The Zalcitabine suppliers may include Zalcitabine API manufacturers, exporters, distributors and traders.
click here to find a list of Zalcitabine suppliers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PharmaCompass.
A Zalcitabine DMF (Drug Master File) is a document detailing the whole manufacturing process of Zalcitabine active pharmaceutical ingredient (API) in detail. Different forms of Zalcitabine DMFs exist exist since differing nations have different regulations, such as Zalcitabine USDMF, ASMF (EDMF), JDMF, CDMF, etc.
A Zalcitabine DMF submitted to regulatory agencies in the US is known as a USDMF. Zalcitabine USDMF includes data on Zalcitabine's chemical properties, information on the facilities and procedures used, and details about packaging and storage. The Zalcitabine USDMF is kept confidential to protect the manufacturer’s intellectual property.
click here to find a list of Zalcitabine suppliers with USDMF on PharmaCompass.
Zalcitabine Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Zalcitabine GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Zalcitabine GMP manufacturer or Zalcitabine GMP API supplier for your needs.
A Zalcitabine CoA (Certificate of Analysis) is a formal document that attests to Zalcitabine's compliance with Zalcitabine specifications and serves as a tool for batch-level quality control.
Zalcitabine CoA mostly includes findings from lab analyses of a specific batch. For each Zalcitabine CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Zalcitabine may be tested according to a variety of international standards, such as European Pharmacopoeia (Zalcitabine EP), Zalcitabine JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Zalcitabine USP).

