
Synopsis
Synopsis
0
EU WC
0
KDMF
0
NDC API
0
VMF
0
Australia
US Medicaid
NA
0
FDF
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
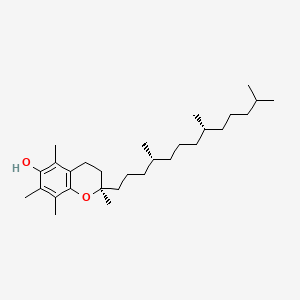

1. Alpha-tocopherol
2. D-alpha-tocopherol
3. 59-02-9
4. (+)-alpha-tocopherol
5. 5,7,8-trimethyltocol
6. Aquasol E
7. Tocopherol
8. 2074-53-5
9. (r,r,r)-alpha-tocopherol
10. Phytogermine
11. Eprolin
12. A-tocopherol
13. (2r,4'r,8'r)-alpha-tocopherol
14. Dl-a-tocopherol
15. Denamone
16. Viteolin
17. Esorb
18. Alpha-tocopherol, D-
19. Tocopherol Alpha
20. Alpha-vitamin E
21. Alpha Tocopherol
22. D-alpha Tocopherol
23. (2r)-2,5,7,8-tetramethyl-2-[(4r,8r)-4,8,12-trimethyltridecyl]chroman-6-ol
24. (r)-2,5,7,8-tetramethyl-2-((4r,8r)-4,8,12-trimethyltridecyl)chroman-6-ol
25. Vitamin Ea
26. Mixed Tocopherols
27. 1406-18-4
28. Syntopherol
29. Tocopherol (r,s)
30. Chebi:18145
31. Vitamin-e
32. Evitaminum
33. Profecundin
34. Waynecomycin
35. Almefrol
36. Emipherol
37. Epsilan
38. Etamican
39. Tokopharm
40. Vascuals
41. Vitayonon
42. Etavit
43. Ilitia
44. Evion
45. (2r)-2,5,7,8-tetramethyl-2-[(4r,8r)-4,8,12-trimethyltridecyl]-3,4-dihydro-2h-chromen-6-ol
46. (2r)-2,5,7,8-tetramethyl-2-[(4r,8r)-4,8,12-trimethyltridecyl]-3,4-dihydrochromen-6-ol
47. Bpbio1_000362
48. 18920-62-2
49. Vitaplex E
50. Eprolin S
51. Spavit E
52. Ido-e
53. Endo E
54. N9pr3490h9
55. Vita E
56. Lan-e
57. Med-e
58. Antisterility Vitamin
59. A-vitamin E
60. 2h-1-benzopyran-6-ol, 3,4-dihydro-2,5,7,8-tetramethyl-2-[(4r,8r)-4,8,12-trimethyltridecyl]-, (2r)-
61. Vi-e
62. E307
63. Dsstox_cid_6339
64. (+/-)-alpha-tocopherol
65. 2,5,7,8-tetramethyl-2-(4',8',12'-trimethyltridecyl)-6-chromanol
66. Dsstox_rid_78103
67. Dsstox_gsid_26339
68. 2h-1-benzopyran-6-ol, 3,4-dihydro-2,5,7,8-tetramethyl-2-[(4r,8r)-4,8,12-trimethyltridecyl]-, (2r)-rel-
69. Viprimol
70. Alpha-tokoferol
71. D-a-tocopherol
72. Mfcd00072045
73. Rrr-alpha-tocopherol
74. Vitamin E Alpha
75. Viterra E
76. E Prolin
77. Cas-59-02-9
78. 2h-1-benzopyran-6-ol, 3,4-dihydro-2,5,7,8-tetramethyl-2-((4r,8r)-4,8,12-trimethyltridecyl)-, (2r)-
79. 2h-1-benzopyran-6-ol, 3,4-dihydro-2,5,7,8-tetramethyl-2-(4,8,12-trimethyltridecyl)-, (2r-(2r*(4r*,8r*)))-
80. 2h-1-benzopyran-6-ol, 3,4-dihydro-2,5,7,8-tetramethyl-2-(4,8,12-trimethyltridecyl)-, [2r-[2r*(4r*,8r*)]]-
81. Smr000471844
82. Viv
83. Alpha-tocopherol Acid
84. Tenox Gt 1
85. Rhenogran Ronotec 50
86. Covitol F 1000
87. 3,4-dihydro-2,5,7,8-tetramethyl-2-(4,8,12-trimethyltridecyl)-2h-benzopyran-6-ol
88. E 307 (tocopherol)
89. (all-r)-alpha-tocopherol
90. Phytogermin
91. Palmvtee
92. Alpha-tocoferol
93. (+-)-med-e
94. E-oil 1000
95. Unii-n9pr3490h9
96. Vitamin Ealpha
97. Ccris 3588
98. Nsc-20812
99. A-d-tocopherol
100. .alpha.-tocopherol, D-
101. Hsdb 2556
102. Pheryl-e
103. Vita Plus E
104. D-..-tocopherol
105. Vitamin E D-alpha
106. Ncgc00016688-02
107. (+)--tocopherol
108. Prestwick_653
109. Einecs 200-412-2
110. Einecs 215-798-8
111. Einecs 218-197-9
112. .alpha.-vitamin E
113. (+)-a-tocopherol
114. Nsc 20812
115. Nsc 82623
116. Rrr-alpha-tocopheryl
117. Vitamin E [usp]
118. ()-alpha-tocopherol
119. Delta-alpha-tocopherol
120. Alpha-delta-tocopherol
121. Tocopherol, D-alpha-
122. Vitamin E (d-form)
123. Chembl47
124. (r,r,r)-a-tocopherol
125. Prestwick3_000404
126. E 307
127. (+)- Alpha -tocopherol
128. (+)-.alpha.-tocopherol
129. Bmse000600
130. R,r,r-.alpha.-tocopherol
131. Ec 200-412-2
132. Schembl3097
133. Unii-h4n855pnz1
134. Bidd:pxr0174
135. Bspbio_000328
136. Mls001066396
137. Mls001335981
138. Mls001335982
139. Bidd:er0562
140. Ins No.307a
141. T1539_sigma
142. H4n855pnz1
143. Ins-307a
144. (+)-alpha-tocopherol-
145. Alpha-tocopherol [hsdb]
146. Rrr-alpha-tocopherol Concentrate
147. Dtxsid0026339
148. (2r,4'r,8'r)-a-tocopherol
149. .alpha.-tocopherol [mi]
150. Hms2096a10
151. Hms2231g08
152. C29h50o2 (d-alpha-tocopherol)
153. D-alpha Tocopherol [mart.]
154. Hy-n0683
155. Zinc4095858
156. Tox21_110563
157. Tox21_113208
158. Tox21_202081
159. Bdbm50458513
160. E-307a
161. Lmpr02020001
162. Akos004910417
163. Cs-8161
164. Db00163
165. (2r)-2-((4r,8r)-4,8,12-trimethyltridecyl)-2,5,7,8-tetramethylchroman-6-ol
166. Ncgc00142625-01
167. Ncgc00142625-04
168. Ncgc00142625-05
169. Ncgc00142625-06
170. Ncgc00142625-07
171. Ncgc00142625-10
172. Ncgc00259630-01
173. (2r*(4r*,8r*))-(1)-3,4-dihydro-2,5,7,8-tetramethyl-2-(4,8,12-trimethyltridecyl)-2h-benzopyran-6-ol
174. As-13990
175. J24.260h
176. T2309
177. (+/-)-alpha-tocopherol (vitamin E) Solution
178. Rrr-alpha-tocopherol Concentrate [fcc]
179. C02477
180. D-alpha, D-beta, D-gamma & D-delta Tocopherols
181. Q158348
182. Q-201932
183. W-107596
184. W-109164
185. 07aa93f0-3339-4eec-b50b-adb70f657087
186. (+)-alpha-tocopherol, From Vegetable Oil, Type V, ~1000 Iu/g
187. (2r,4'r,8'r)-2,5,7,8-tetramethyl-2-(4',8',12'-trimethyltridecyl)-6-chromanol
188. 3,4-dihydro-2,5,7,8-tetramethyl-2-(4,8,12-trimethyltridecyl)-2h-1- -benzopyran-6-ol
189. (+)-alpha-tocopherol, Type Vi, From Vegetable Oil, Neat (liquid, >=0.88m Based On Potency, Density And Molecular Wt.), Bioreagent, Suitable For Insect Cell Culture, >=1000 Iu/g
190. (2r)-3,4-dihydro-2,5,7,8-tetramethyl-2-[(4r,8r)-4,8,12-trimethyltridecyl]-2h-1-benzopyran-6-ol
191. (r)-2,5,7,8-tetramethyl-2-((4r,8r)-4,8,12-trimethyltridecyl)-3,4-dihydro-2h-chromen-6-ol
192. 2h-1-benzopyran-6-ol, 3,4-dihydro-2,5,7,8-tetramethyl-2-(4,8,12-trimethyltridecyl)-, 2r- 2r*(4r*,8r*) -
193. 2h-1-benzopyran-6-ol, 3,4-dihydro-2,5,7,8-tetramethyl-2-(4,8,12-trimethyltridecyl)-, (2r*(4r*,8r*))-(+-)-
194. Alpha-tocopherol (constituent Of Lycopene And Tomato Extract Containing Lycopene) [dsc]
| Molecular Weight | 430.7 g/mol |
|---|---|
| Molecular Formula | C29H50O2 |
| XLogP3 | 10.7 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 2 |
| Rotatable Bond Count | 12 |
| Exact Mass | 430.381080833 g/mol |
| Monoisotopic Mass | 430.381080833 g/mol |
| Topological Polar Surface Area | 29.5 Ų |
| Heavy Atom Count | 31 |
| Formal Charge | 0 |
| Complexity | 503 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 3 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
As a dietary supplement when vitamin E intake may be inadequate.
Drug Facts and Comparisons 2015. Clinical Drug Information, LLC St. Louis, MO 2015, p. 15
Vitamin E deficiency in premature infants may result in hemolytic anemia, thrombocytosis, and increased platelet aggregation.
Drug Facts and Comparisons 2015. Clinical Drug Information, LLC St. Louis, MO 2015, p. 16
/Vitamin E/ has also been used in cancer, skin conditions, sexual dysfunction, to reduce the incidence of non-fatal myocardial infarction, to lower the incidence of coronary artery disease, aging, fibrocystic breast disease (cystic mastitis), to treat dapsone-associated hemolysis, and arthritis. /not included in the US product label/
Drug Facts and Comparisons 2015. Clinical Drug Information, LLC St. Louis, MO 2015, p. 15
Vitamin E has been used in certain premature infants to reduce the toxic effects of oxygen therapy on the lung parenchyma... and the retina... /not included in the US product label/
Drug Facts and Comparisons 2015. Clinical Drug Information, LLC St. Louis, MO 2015, p. 15
For more Therapeutic Uses (Complete) data for VITAMIN E (19 total), please visit the HSDB record page.
Vitamin E in high doses (greater than 4000 IU) may increase the hypoprothrombinemic effects of oral anticoagulants.
Drug Facts and Comparisons 2015. Clinical Drug Information, LLC St. Louis, MO 2015, p. 16
Vitamin E may impair the hematologic response to iron therapy in children with iron-defiiency anemia.
Drug Facts and Comparisons 2015. Clinical Drug Information, LLC St. Louis, MO 2015, p. 16
FDA Pregnancy Risk Category: A CONTROLLED STUDIES SHOW NO RISK. Adequate, well controlled studies in pregnant women have failed to demonstrate a risk to the fetus in any trimester of pregnancy.
Dart, R.C. (ed). Medical Toxicology. Third Edition, Lippincott Williams & Wilkins. Philadelphia, PA. 2004., p. 1020
Vitamin E has not been shown to be teratogenic. There is no evidence that vitamin E requirements in pregnant women differ from women who are not pregnant.
American Society of Health-System Pharmacists 2015; Drug Information 2015. Bethesda, MD. 2015
Reports of toxicity to enterally administered vitamin E are rare in infants. However, increased risks of sepsis and necrotizing enterocolitis have been reported after both enteral and parenteral vitamin E, primarily when plasma (or serum) vitamin E levels exceed 3.5 mg/dL. Levels this high are seldom seen with enteral vitamin E when intake is 25 mg d-tocopherol equivalent/(kg/day) or less. Intakes below this threshold will be provided by infant formulas with vitamin E to energy ratios of up to 20 mg/100 kcal (30 IU/100 kcal) so long as energy intake does not exceed 125 kcal/(kg/day).
PMID:2693643 Bell EF; J Nutr 119 (12 Suppl): 1829-31 (1989)
Vitamin E supplementation is indicated for treatment of vitamin E deficiency which can occur in cystic fibrosis, cholestasis and severe liver disease, abetalipoproteinemia or simply poor diet.
Tocopherol can be used as a dietary supplement for patients with a deficit of vitamin E; this is mainly prescribed in the alpha form. Vitamin E deficiency is rare, and it is primarily found in premature babies of very low birth weight, patients with fat malabsorption or patients with abetalipoproteinemia. Tocopherol, due to its antioxidant properties, is studied for its use in prevention or treatment in different complex diseases such as cancer, atherosclerosis, cardiovascular diseases, and age-related macular degeneration.
Vitamin E is a collective term used to describe 8 separate fat soluble antioxidants, most commonly alpha-tocopherol. Vitamin E acts to protect cells against the effects of free radicals, which are potentially damaging by-products of the body's metabolism. Vitamin E deficiency is seen in persons with abetalipoproteinemia, premature, very low birth weight infants (birth weights less than 1500 grams, or 3½ pounds), cystic fibrosis, and cholestasis and severe liver disease. Preliminary research suggests vitamin E may help prevent or delay coronary heart disease and protect against the damaging effects of free radicals, which may contribute to the development of chronic diseases such as cancer. It also protects other fat-soluble vitamins (A and B group vitamins) from destruction by oxygen. Low levels of vitamin E have been linked to increased incidence of breast and colon cancer.
The antioxidant effects of tocopherol can be translated into different changes at the pharmacodynamic level. In vitro studies have shown that this antioxidant activity can produce modification in protein kinase C (PKC) which will later be translated into an inhibition of cell death. Some other derivate effects are the anti-inflammatory properties of tocopherol which can be related to the modulation of cytokines or prostaglandins, prostanoids and thromboxanes.
Vitamins
Organic substances that are required in small amounts for maintenance and growth, but which cannot be manufactured by the human body. (See all compounds classified as Vitamins.)
Antioxidants
Naturally occurring or synthetic substances that inhibit or retard oxidation reactions. They counteract the damaging effects of oxidation in animal tissues. (See all compounds classified as Antioxidants.)
Absorption
10-33% of deuterium labelled vitamin E is absorbed in the small intestine. Absorption of Vitamin E is dependant upon absorption of the fat in which it is dissolved. For patients with poor fat absorption, a water soluble form of vitamin E may need to be substituted such as tocopheryl polyethylene glycol-1000 succinate. In other studies the oral bioavailability of alpha-tocopherol was 36%, gamma-tocotrienol was 9%. The time to maximum concentration was 9.7 hours for alpha-tocopherol and 2.4 hours for gamma-tocotrienol.
Route of Elimination
Alpha tocopherol is excreted in urine as well as bile in the feces mainly as a carboxyethyl-hydrochroman (CEHC) metabolite, but it can be excreted in it's natural form.
Volume of Distribution
0.41L/kg in premature neonates given a 20mg/kg intramuscular injection.
Clearance
6.5mL/hr/kg in premature neonates given a 20mg/kg intramuscular injection.
Absorption
The absorption of tocopherol in the digestive tract requires the presence of fat. The approximate tmax of the four different isomers of tocopherol is attained in the range of 3-6 hours. When compared with tocotrienols, tocopherols showed a reduced bioavailability. The bioavailability of tocopherols is highly dependent on the type of isomer that is administered where the alpha-tocopherol can present a bioavailability of 36%. This isomer specificity also determines the intestinal permeability in which the gamma-tocopherol presents a very low permeability. After oral administration, the Cmax, AUC and mean residence time of tocopherols showed to be dependent on the isomer and ranged from 590-2915 ng/ml, 3740-10169 ng/ml and 4.85-5.74 h, respectively.
Route of Elimination
The pharmacokinetic profile of tocopherol indicates a longer time of excretion for tocopherols when compared to tocotrienols. The different conjugated metabolites are excreted in the urine or feces depending on the length of their side-chain. Due to their polarity, intermediate-chain metabolites and short-chain metabolites are excreted via urine as glucoside conjugates. A mixture of all the metabolites and precursors can be found in feces. The long-chain metabolites correspond to >60% of the total metabolites in feces. It is estimated that the fecal excretion accounts for even 80% of the administered dose.
Volume of Distribution
The apparent volume of distribution of tocopherol is approximately 0.5 ml.
Clearance
The clearance rate of tocopherol is approximately 0.15 l/h.
alpha-Tocopherol is absorbed via the lymphatic pathway and transported in association with chylomicrons. In plasma, alpha-tocopherol is found in all lipoprotein fractions but mostly is associated with apo B containing lipoproteins. alpha-Tocopherol is associated with very low density lipoprotein when it is secreted from the liver. In the rat, about 90% of total body mass of alpha-tocopherol is recovered in the liver, skeletal muscle and adipose tissue. Most alpha-tocopherol is located in the mitochondrial fractions and in the endoplasmic reticulum, whereas little is found in cytosol and peroxisomes.
PMID:2181082 Bjorneboe A et al; J Nutr 120 (3): 233-42 (1990)
The level of retinal alpha-tocopherol of newborn rats could be altered by dietary manipulation of the mothers. The mothers were fed diets containing either 1 g alpha-tocopherol acetate/kg food or none, starting 21-25 days before the birth of their litters and lasting throughout the exposure period. This treatment resulted in three to fourfold differences in the retinal alpha-tocopherol levels of the pups. The combination of dietary and oxygen treatments also resulted in significant differences in retinal glutathione peroxidase activity, with the vitamin E deprived, oxygen exposed group having highest levels. Newborn rats both supplemented with and deprived of alpha-tocopherol had less vasoobliteration than did those nursed by mothers fed rat chow.
PMID:1582786 Penn JS et al; Invest Ophthalmol Vis Sci 33 (6): 1836-45 (1992)
Vitamin E is stored unmodified in tissues (principally the liver and adipose tissue) and excreted via the feces. Excess vitamin E is converted to a lactone, esterified to glucuronic acid, and subsequently excreted in the urine.
Drug Facts and Comparisons 2015. Clinical Drug Information, LLC St. Louis, MO 2015, p. 16
Vitamin E is 20% to 50% absorbed by intestinal epithelial cells in the small intestine. Bile and pancreatic juice are needed for tocopherol absorption. Absorption is increased when administered with medium-chain triglycerides. Distribution to tissues via the lymphatic system occurs as a lipoprotein complex. High concentrations of vitamin E are found in the adrenals, pituitary, testes, and trombocytes.
Drug Facts and Comparisons 2015. Clinical Drug Information, LLC St. Louis, MO 2015, p. 16
For more Absorption, Distribution and Excretion (Complete) data for VITAMIN E (11 total), please visit the HSDB record page.
Alpha and gamma tocopherol are undergo beta oxidation and a process mediated by cytochrome P450s such as CYP4F2, CYP3A4, and CYP3A5. These processes convert alpha and gamma tocopherol to alpha-CEHC (2,5,7,8-tetramethyl-2-(2-carboxyethyl)-6-hydroxychroman) and gamma-CEHC (2,7,8-trimethyl-2-(2-carboxyethyl)-6-hydroxychroman) respectively, however the full process is not known.
Excess tocopherol is converted into their corresponding carboxyethylhydroxychroman (CEHC), based on the isomer of tocopherol. More deeply, the metabolism of tocopherol begins with the hepatic metabolism which is led by a CYP4F2/CYP3A4-dependent -hydroxylation of the side chains which leads to the formation of 13'-carboxychromanol. The metabolic pathway is followed by five cycles of -oxidation. The -oxidation cycles function by shortening the side chains, the first cycle results in the formation of carboxydimethyldecylhydroxychromanol followed by carboxymethyloctylhydroxychromanol. These two metabolites are categorized as long-chain metabolites and they are not excreted in the urine. Some intermediate-chain metabolites that are products of two rounds of -oxidation are carboxymethylhexylhydroxychromanol and carboxymethylbutylhydroxychromanol. These intermediate-chain metabolites can be found in human feces and urine. The catabolic end-product of tocopherols, as stated before, is CEHC which can be largely found in urine and feces. Two new metabolites have been detected in human and mice feces. These new metabolites are 12'-hydroxychromanol and 11'-hydroxychromanol. Because of their chemistry, it is thought that these metabolites can be the evidence for a -1 and -2 hydroxylation which leads to an impaired oxidation of 12'-OH followed side-chain truncation.
Vitamin E is stored unmodified in tissues (principally the liver and adipose tissue) and excreted via the feces. Excess vitamin E is converted to a lactone, esterified to glucuronic acid, and subsequently excreted in the urine.
Drug Facts and Comparisons 2015. Clinical Drug Information, LLC St. Louis, MO 2015, p. 16
Alpha-tocopherol (Vitamin E), formed from homogentisic acid yields, in rabbits 4-hydroxy-4-methyl-6-(2,4,5-trimethyl-3,6-quinonyl)caproic acid ...
Goodwin, B.L. Handbook of Intermediary Metabolism of Aromatic Compounds. New York: Wiley, 1976., p. T-15
Vitamin E is likely the most important antioxidant in the human diet and alpha-tocopherol is the most active isomer. Alpha-tocopherol exhibits anti-oxidative capacity in vitro, and inhibits oxidation of ldl. Beside this, alpha-tocopherol shows anti-inflammatory activity and modulates expression of proteins involved in uptake, transport and degradation of tocopherols, as well as the uptake, storage and export of lipids such as cholesterol. Despite promising anti-atherogenic features in vitro, vitamin E failed to be atheroprotective in clinical trials in humans. Recent studies highlight the importance of long-chain metabolites of alpha-tocopherol, which are formed as catabolic intermediate products in the liver and occur in human plasma. These metabolites modulate inflammatory processes and macrophage foam cell formation via mechanisms different than that of their metabolic precursor alpha-tocopherol and at lower concentrations. Here we summarize the controversial role of vitamin E as a preventive agent against atherosclerosis and point the attention to recent findings that highlight a role of these long-chain metabolites of vitamin E as a proposed new class of regulatory metabolites. We speculate that the metabolites contribute to physiological as well as pathophysiological processes.
PMID:24624339 Full text: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3949092 Wallert M et al; Redox Biol. 2: 495-503 (2014)
44 hours in premature neonates given a 20mg/kg intramuscular injection. 12 minutes in intravenous injection of intestinal lymph.
The elimination half-life of tocopherol is approximately 2.6 hours.
Little is known about alpha-tocopherol's bioavailability as a constituent of food or its dependence on a subject's age. To evaluate the alpha-tocopherol bioavailability from food, we used collard greens grown in deuterated water ((2)H collard greens) as a source of deuterium-labeled ((2)H) alpha-tocopherol consumed by younger and older adults in a post hoc analysis of a vitamin K study. Younger (mean +/- SD age: 32 +/- 7 y; n = 12 women and 9 men) and older (aged 67 +/- 8 y; n = 8 women and 12 men) adults consumed a test breakfast that included 120 g (2)H collard greens (1.2 +/- 0.1 mg (2)H-alpha-tocopherol). Plasma unlabeled alpha-tocopherol and (2)H-alpha-tocopherol were measured by using liquid chromatography-mass spectrometry from fasting (>12 hr) blood samples drawn before breakfast (0 hr) and at 24, 48, and 72 hr and from postprandial samples collected at 4, 5, 6, 7, 9, 12, and 16 hr. Times (12.6 +/- 2.5 h) of maximum plasma (2)H-alpha-tocopherol concentrations (0.82% +/- 0.59% total alpha-tocopherol), fractional disappearance rates (0.63 +/- 0.26 pools/d), half-lives (30 +/- 11 hr), and the minimum estimated (2)H-alpha-tocopherolabsorbed (24% +/- 16%) did not vary between age groups or sexes (n = 41). Unlabeled alpha-tocopherol concentrations were higher in older adults (26.4 +/- 8.6 umol/L) than in younger adults (19.3 +/- 4.2 umol/L; P = 0.0019) and correlated with serum lipids (r = 0.4938, P = 0.0012). In addition, (2)H-alpha-tocopherol half-lives were correlated with lipids (r = 0.4361, P = 0.0044). Paradoxically, alpha-tocopherol remained in circulation longer in participants with higher serum lipids, but the (2)H-alpha-tocopherol absorbed was not dependent on the plasma lipid status. Neither variable was dependent on age. These data suggest that plasma alpha-tocopherol concentrations are more dependent on mechanisms that control circulating lipids rather than those related to its absorption and initial incorporation into plasma.
PMID:25739929 Full text: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4381779 Traber MG et al; Am J Clin Nutr. 101 (4): 752-9 (2015)
The mechanism of action for most of vitamin E's effects are still unknown. Vitamin E is an antioxidant, preventing free radical reactions with cell membranes. Though in some cases vitamin E has been shown to have pro-oxidant activity. One mechanism of vitamin E's antioxidant effect is in the termination of lipid peroxidation. Vitamin E reacts with unstable lipid radicals, producing stable lipids and a relatively stable vitamin E radical. The vitamin E radical is then reduced back to stable vitamin E by reaction with ascorbate or glutathione.
Tocopherol acts as a radical scavenger. It mainly acts as an antioxidant for lipid bilayers. Tocopherol's functions depend on the H-atom donating ability, location, and movement within the membrane, as well as the efficiency in the radical recycling by some cytosolic reductants such as ascorbate. Tocopherol actions are related to the trap of radicals, and it has been shown that even in the absence of substituents in the ortho-positions, tocopherol can trap more than two radicals. The type of radicals available for tocopherol are alkyl and peroxy.
Cancer development and progression are closely associated with inflammation. NF-kappaB (nuclear factor kappaB) provides a mechanistic link between inflammation and cancer, and is a major factor controlling the ability of malignant cells to resist tumor surveillance mechanisms. NF-kappaB might also regulate tumor angiogenesis and invasiveness and the signalling pathways that mediate its activation provide attractive targets for new chemopreventive and chemotherapeutic approaches. ROS (reactive oxygen species) initiate inflammation by up-regulation of pro-inflammatory cytokines and therefore antioxidants provide a major defence against inflammation. alpha-Tocopherol is a lipid-soluble antioxidant. In addition to decreasing lipid peroxidation, alpha-tocopherol may exert intracellular effects. Hence, the aim of this study was to test the effect of alpha-tocopherol supplementation in cancer prevention via suppression of NF-kappaB-mediated pro-inflammatory cytokines. alpha-Tocopherol treatment significantly down-regulates expression, synthesis as well as secretion of pro-inflammatory cytokine IL-6 (interleukin-6) in cancerous mice. It also suppresses NF-kappaB binding to IL-6 promoter in liver leading to decreased secretion of IL-6 in serum. The regulation of the signalling pathway by alpha-tocopherol is found apart from its antioxidant capacity to reduce lipid peroxidation. Thus, the present study provides evidence for the hypothesis that besides the powerful free radical scavenging effects, alpha-tocopherol has genomic effects in down-regulation of pro-inflammatory cytokine and cancer prevention via the NF-kappaB-dependent pathway.
PMID:21320073 Sharma R, Vinayak M; Biosci Rep. 31 (5): 421-8 (2011)
Mitocans are drugs selectively killing cancer cells by destabilizing mitochondria and many induce apoptosis via generation of reactive oxygen species (ROS). However, the molecular events by which ROS production leads to apoptosis has not been clearly defined. In this study with the mitocan alpha-tocopheryl succinate (alpha-TOS) the role of the Bcl-2 family proteins in the mechanism of malignant cell apoptosis has been determined. Exposure of several different cancer cell lines to alpha-TOS increased expression of the Noxa protein, but none of the other proteins of the Bcl-2 family, an event that was independent of the cellular p53 status. alpha-TOS caused a profound conformational change in the pro-apoptotic protein, Bak, involving oligomerization in all cell types, and this also applied to the Bax protein, but only in non-small cell lung cancer cells. Immunoprecipitation studies indicated that alpha-TOS activates the two BH1-3 proteins, Bak or Bax, to form high molecular weight complexes in the mitochondria. RNAi knockdown revealed that Noxa and Bak are required for alpha-TOS-induced apoptosis, and the role of Bak was confirmed using Bak- and/or Bax-deficient cells. We conclude that the major events induced by alpha-TOS in cancer cells downstream of ROS production leading to mitochondrial apoptosis involve the Noxa-Bak axis. It is proposed that this represents a common mechanism for mitochondrial destabilization activated by a variety of mitocans that induce accumulation of ROS in the early phases of apoptosis. /alpha-Tocopheryl succinate/
PMID:20217235 Prochazka L et al; Apoptosis. 15 (7): 782-94 (2010)

API/FDF Prices: Book a Demo to explore the features and consider upgrading later
API Imports and Exports
| Importing Country | Total Quantity (KGS) |
Average Price (USD/KGS) |
Number of Transactions |
|---|
Upgrade, download data, analyse, strategize, subscribe with us
DRUG PRODUCT COMPOSITIONS

Related Excipient Companies

Excipients by Applications
Global Sales Information

Market Place

Patents & EXCLUSIVITIES
REF. STANDARDS & IMPURITIES

ANALYTICAL

ABOUT THIS PAGE
26
PharmaCompass offers a list of Vitamin E API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, Price,and more, enabling you to easily find the right Vitamin E manufacturer or Vitamin E supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Vitamin E manufacturer or Vitamin E supplier.
PharmaCompass also assists you with knowing the Vitamin E API Price utilized in the formulation of products. Vitamin E API Price is not always fixed or binding as the Vitamin E Price is obtained through a variety of data sources. The Vitamin E Price can also vary due to multiple factors, including market conditions, regulatory modifications, or negotiated pricing deals.
A Vitamin E Nicotinate manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Vitamin E Nicotinate, including repackagers and relabelers. The FDA regulates Vitamin E Nicotinate manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Vitamin E Nicotinate API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
click here to find a list of Vitamin E Nicotinate manufacturers with USDMF, JDMF, KDMF, CEP, GMP, COA and API Price related information on PhamaCompass.
A Vitamin E Nicotinate supplier is an individual or a company that provides Vitamin E Nicotinate active pharmaceutical ingredient (API) or Vitamin E Nicotinate finished formulations upon request. The Vitamin E Nicotinate suppliers may include Vitamin E Nicotinate API manufacturers, exporters, distributors and traders.
click here to find a list of Vitamin E Nicotinate suppliers with USDMF, JDMF, KDMF, CEP, GMP, COA and API Price related information on PharmaCompass.
A Vitamin E Nicotinate DMF (Drug Master File) is a document detailing the whole manufacturing process of Vitamin E Nicotinate active pharmaceutical ingredient (API) in detail. Different forms of Vitamin E Nicotinate DMFs exist exist since differing nations have different regulations, such as Vitamin E Nicotinate USDMF, ASMF (EDMF), JDMF, CDMF, etc.
A Vitamin E Nicotinate DMF submitted to regulatory agencies in the US is known as a USDMF. Vitamin E Nicotinate USDMF includes data on Vitamin E Nicotinate's chemical properties, information on the facilities and procedures used, and details about packaging and storage. The Vitamin E Nicotinate USDMF is kept confidential to protect the manufacturer’s intellectual property.
click here to find a list of Vitamin E Nicotinate suppliers with USDMF on PharmaCompass.
The Pharmaceuticals and Medical Devices Agency (PMDA) established the Japan Drug Master File (JDMF), also known as the Master File (MF), to permit Japanese and foreign manufacturers of drug substances, intermediates, excipients, raw materials, and packaging materials (‘Products’) to voluntarily register confidential information about the production and management of their products in Japan.
The Vitamin E Nicotinate Drug Master File in Japan (Vitamin E Nicotinate JDMF) empowers Vitamin E Nicotinate API manufacturers to present comprehensive information (e.g., production methods, data, etc.) to the review authority, i.e., PMDA (Pharmaceuticals & Medical Devices Agency).
PMDA reviews the Vitamin E Nicotinate JDMF during the approval evaluation for pharmaceutical products. At the time of Vitamin E Nicotinate JDMF registration, PMDA checks if the format is accurate, if the necessary items have been included (application), and if data has been attached.
click here to find a list of Vitamin E Nicotinate suppliers with JDMF on PharmaCompass.
A Vitamin E Nicotinate CEP of the European Pharmacopoeia monograph is often referred to as a Vitamin E Nicotinate Certificate of Suitability (COS). The purpose of a Vitamin E Nicotinate CEP is to show that the European Pharmacopoeia monograph adequately controls the purity of Vitamin E Nicotinate EP produced by a given manufacturer. Suppliers of raw materials can prove the suitability of Vitamin E Nicotinate to their clients by showing that a Vitamin E Nicotinate CEP has been issued for it. The manufacturer submits a Vitamin E Nicotinate CEP (COS) as part of the market authorization procedure, and it takes on the role of a Vitamin E Nicotinate CEP holder for the record. Additionally, the data presented in the Vitamin E Nicotinate CEP (COS) is managed confidentially and offers a centralized system acknowledged by numerous nations, exactly like the Vitamin E Nicotinate DMF.
A Vitamin E Nicotinate CEP (COS) is recognised by all 36 nations that make up the European Pharmacopoeia Convention. Vitamin E Nicotinate CEPs may be accepted in nations that are not members of the Ph. Eur. at the discretion of the authorities there.
click here to find a list of Vitamin E Nicotinate suppliers with CEP (COS) on PharmaCompass.
Vitamin E Nicotinate Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Vitamin E Nicotinate GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, API price, and more, enabling you to easily find the right Vitamin E Nicotinate GMP manufacturer or Vitamin E Nicotinate GMP API supplier for your needs.
A Vitamin E Nicotinate CoA (Certificate of Analysis) is a formal document that attests to Vitamin E Nicotinate's compliance with Vitamin E Nicotinate specifications and serves as a tool for batch-level quality control.
Vitamin E Nicotinate CoA mostly includes findings from lab analyses of a specific batch. For each Vitamin E Nicotinate CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Vitamin E Nicotinate may be tested according to a variety of international standards, such as European Pharmacopoeia (Vitamin E Nicotinate EP), Vitamin E Nicotinate JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Vitamin E Nicotinate USP).

