
Synopsis
Synopsis
0
USDMF
0
CEP/COS
0
JDMF
0
EU WC
0
KDMF
0
NDC API
0
VMF
0
EDQM
0
USP
0
JP
0
Others
0
FDA Orange Book
0
Canada
0
Australia
0
South Africa
0
Listed Dossiers
DRUG PRODUCT COMPOSITIONS
0
US Patents
0
US Exclusivities
0
Health Canada Patents
US Medicaid
NA
Annual Reports
NA
Regulatory FDF Prices
NA
0
FDF
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
0
News #PharmaBuzz

1. 10-undecenoic Acid
2. Mycodermine
1. 10-undecenoic Acid
2. Undec-10-enoic Acid
3. 112-38-9
4. Undecenoic Acid
5. 10-hendecenoic Acid
6. Desenex
7. Renselin
8. Declid
9. Sevinon
10. 9-undecylenic Acid
11. 10-undecylenic Acid
12. Undecyl-10-enic Acid
13. Undecylenate
14. Undecen-10-acid-1
15. Undecylenic Acids
16. Fema No. 3247
17. Undecylenic Acid [jan]
18. Kyselina Undecylenova
19. 10-henedecenoic Acid
20. 10-hendecenoic
21. Desenex, Solution
22. Nsc 2013
23. N-undecylenic Acid
24. Omega-undecenoic Acid
25. Cruex (tn)
26. Omega-hendecenoic Acid
27. Chebi:35045
28. Nsc-2013
29. 1333-28-4
30. 10-undecenoic Acid, 98%
31. Undecylenic Acid (jan/usp)
32. K3d86kj24n
33. 10-undecenoic Acid, Homopolymer
34. Nsc2013
35. Undesine
36. C11:1n-1
37. 96451-28-4
38. Ncgc00159425-02
39. Ncgc00159425-04
40. Desenex Solution
41. Dsstox_cid_15001
42. Dsstox_rid_79235
43. Dsstox_gsid_35001
44. Caswell No. 901
45. Undecenoicacid
46. Undecenoic Acid, Omega-
47. Hendecenoic Acid, Omega-
48. Cas-112-38-9
49. Kyselina Undecylenova [czech]
50. Einecs 203-965-8
51. Mfcd00004442
52. Epa Pesticide Chemical Code 085501
53. Brn 1762631
54. Unii-k3d86kj24n
55. Kyselina 9-decen-1-karboxylova [czech]
56. Undecylenic Acid [usp:jan]
57. Undecelinic Acid
58. Undecylenic-acid
59. Ai3-02065
60. Kyselina 9-decen-1-karboxylova
61. Undecylenenic Acid
62. 10-undecensaeure
63. 10-undecenic Acid
64. Einecs 215-583-9
65. Undecen-10-saeure
66. N-undecylenic Acid (10-1)
67. Zinc Undecylenate (undecylenic Acid)
68. 10-undeceneoic Acid
69. Acide 10-undecylique
70. Acido 10-undecenoico
71. Acide 10-undecanoique
72. Undecylenic Acid, Usan
73. Ec 203-965-8
74. Nciopen2_002642
75. Undecylenate [vandf]
76. Wln: Qv9u1
77. Schembl17827
78. 4-02-00-01612 (beilstein Handbook Reference)
79. Undecylenic Acid [ii]
80. Undecylenic Acid [mi]
81. Undecylenic Acid [inci]
82. Chembl1276010
83. Dtxsid8035001
84. Undecenoic Acid [mart.]
85. Undecylenic Acid [vandf]
86. Fema 3247
87. Undecylenic Acid, >=95%, Fg
88. Undecylenic Acid, >=96%, Fg
89. Undecylenic Acid [usp-rs]
90. Undecylenic Acid [who-dd]
91. Hms2093l21
92. Pharmakon1600-01505468
93. 10-undecenoic Acid [fhfi]
94. Hy-b0914
95. Zinc1577120
96. Tox21_111657
97. Tox21_300383
98. Bbl027462
99. Bdbm50248304
100. Lmfa01030036
101. Nsc759153
102. S9452
103. Stk801548
104. Akos009031595
105. Tox21_111657_1
106. Ccg-213461
107. Db11117
108. Nsc-759153
109. Pb48616
110. Undecylenic Acid [ep Monograph]
111. 10-undecenoic Acid, Analytical Standard
112. Undecylenic Acid [usp Monograph]
113. Ncgc00159425-03
114. Ncgc00159425-05
115. Ncgc00159425-09
116. Ncgc00254296-01
117. Undecylenic Acid, Natural, >=97%, Fg
118. Nci60_001670
119. Sbi-0206806.p001
120. Sbi-0206806.p002
121. Ft-0607203
122. U0007
123. 10-undecenoic Acid, Purum, >=97.0% (gc)
124. D02159
125. P19683
126. Ab00630892_02
127. Q420346
128. Sr-05000002050
129. 10-undecenoic Acid, Vetec(tm) Reagent Grade, 98%
130. Sr-05000002050-1
131. W-200840
132. F0001-0258
133. Undecylenic Acid, United States Pharmacopeia (usp) Reference Standard
134. 10-undecenoic Acid, Puriss., Meets Analytical Specification Of Ph. Eur., Bp, Usp, 98-100.5%
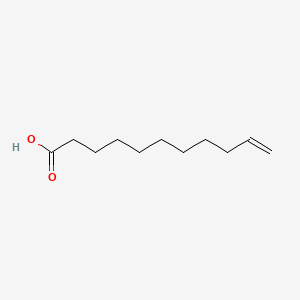
| Molecular Weight | 184.27 g/mol |
|---|---|
| Molecular Formula | C11H20O2 |
| XLogP3 | 3.9 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 2 |
| Rotatable Bond Count | 9 |
| Exact Mass | 184.146329876 g/mol |
| Monoisotopic Mass | 184.146329876 g/mol |
| Topological Polar Surface Area | 37.3 Ų |
| Heavy Atom Count | 13 |
| Formal Charge | 0 |
| Complexity | 141 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
Indicated for the treatment of fungal infections as a salt form. No therapeutic indications on its own.
Zinc undecylendate acts as a fungistatic agent but fungicidal activity may be observed with chronic exposure in high concentrations. It is effective against _Candida albicans_. It is proposed that undecylenic acid exerts antimicrobial actions via interacting with nonspecific components in the cell membrane.
D - Dermatologicals
D01 - Antifungals for dermatological use
D01A - Antifungals for topical use
D01AE - Other antifungals for topical use
D01AE04 - Undecylenic acid
Absorption
Undecylenic acid may be absorbed through the skin [MSDS].
Route of Elimination
No information regarding route of elimination.
Volume of Distribution
No information regarding volume of distribution.
Clearance
No information regarding clearance.
No information regarding metabolism.
No information regarding half-life.
Undecylenic acid demonstrated effectiveness against _Candida albicans_, which is an opportunistic pathogenic yeast with two cellular morphologies: the round yeast form and the filamentous form with elongated hyphae. Hyphae formation is associated with active infections and virulence. A study proposed that undecylenic acid inhibits biofilm formation of _Candida albicans_ with optimal concentration above 3 mM and disrupts hyphal growth, which is the morphological transition from yeast to filamentous phase, at concentration above 4 mM. Under the drug treatment, hyphal formation related genes, like HWP1, were significantly reduced in transcriptional level leading to poor biofilm formation. Both biofilm and hyphae formation are critical virulence factors for the initiation of skin infection and late development of disseminated infection. Undecylenic acid may also inhibit enzyme involved in lipid metabolism and abolish germ tube formation by carrying protons across the plasma membrane, thus altering cytoplasmic pH.
Related Excipient Companies

Excipients by Applications
Market Place

ABOUT THIS PAGE
63
PharmaCompass offers a list of Undecylenic Acid API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Undecylenic Acid manufacturer or Undecylenic Acid supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Undecylenic Acid manufacturer or Undecylenic Acid supplier.
A Undecylenic Acid manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Undecylenic Acid, including repackagers and relabelers. The FDA regulates Undecylenic Acid manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Undecylenic Acid API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
click here to find a list of Undecylenic Acid manufacturers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PhamaCompass.
A Undecylenic Acid supplier is an individual or a company that provides Undecylenic Acid active pharmaceutical ingredient (API) or Undecylenic Acid finished formulations upon request. The Undecylenic Acid suppliers may include Undecylenic Acid API manufacturers, exporters, distributors and traders.
click here to find a list of Undecylenic Acid suppliers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PharmaCompass.
Undecylenic Acid Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Undecylenic Acid GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Undecylenic Acid GMP manufacturer or Undecylenic Acid GMP API supplier for your needs.
A Undecylenic Acid CoA (Certificate of Analysis) is a formal document that attests to Undecylenic Acid's compliance with Undecylenic Acid specifications and serves as a tool for batch-level quality control.
Undecylenic Acid CoA mostly includes findings from lab analyses of a specific batch. For each Undecylenic Acid CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Undecylenic Acid may be tested according to a variety of international standards, such as European Pharmacopoeia (Undecylenic Acid EP), Undecylenic Acid JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Undecylenic Acid USP).

