
Synopsis
Synopsis
0
VMF
0
US Patents
0
US Exclusivities
0
Health Canada Patents
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
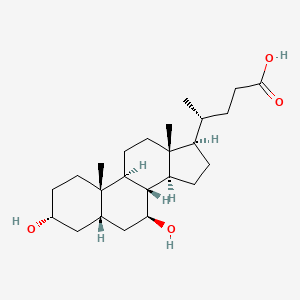

1. 3 Alpha,7 Beta Dihydroxy 5 Beta Cholan 24 Oic Acid
2. 3 Alpha,7 Beta-dihydroxy-5 Beta-cholan-24-oic Acid
3. Acid, Deoxyursocholic
4. Acid, Ursacholic
5. Acid, Ursodeoxycholic
6. Cholit-ursan
7. Cholofalk
8. Delursan
9. Deoxyursocholic Acid
10. Destolit
11. Sodium Ursodeoxycholate
12. Urdox
13. Ursacholic Acid
14. Urso
15. Urso Heumann
16. Ursobilane
17. Ursochol
18. Ursodeoxycholate, Sodium
19. Ursodiol
20. Ursofalk
21. Ursogal
22. Ursolite
23. Ursolvan
1. Ursodiol
2. 128-13-2
3. Actigall
4. Ursodeoxycholate
5. Udca
6. Ursofalk
7. Ursolvan
8. Urso
9. Delursan
10. Ursodesoxycholic Acid
11. Urso Forte
12. Destolit
13. Ursochol
14. Cholit-ursan
15. Litursol
16. Solutrat
17. Ursobilin
18. Ursodamor
19. Arsacol
20. Deursil
21. Lyeton
22. Ursacol
23. Urso Ds
24. Udcs
25. 3alpha,7beta-dihydroxy-5beta-cholan-24-oic Acid
26. Ursosan
27. Urso 250
28. 3-alpha,7-beta-dioxycholanic Acid
29. Ursocholic Acid, Deoxy-
30. 3-alpha,7-beta-dihydroxycholanic Acid
31. Ursonorm
32. Nsc 683769
33. (3alpha,5beta,7beta)-3,7-dihydroxycholan-24-oic Acid
34. 3-alpha,7-beta-dihydroxy-5-beta-cholanoic Acid
35. Ursodeoxycholicacid
36. (4r)-4-[(3r,5s,7s,8r,9s,10s,13r,14s,17r)-3,7-dihydroxy-10,13-dimethyl-2,3,4,5,6,7,8,9,11,12,14,15,16,17-tetradecahydro-1h-cyclopenta[a]phenanthren-17-yl]pentanoic Acid
37. Nsc 657950
38. Brn 3219888
39. 3,7-dihydroxycholan-24-oic Acid
40. Peptarom
41. Ursodeoxycholic Acid [inn]
42. Chembl1551
43. Mls000028461
44. Chebi:9907
45. 724l30y2qr
46. Nsc-683769
47. Ursodiol [usan]
48. Antigall
49. Smr000058403
50. Urosiol
51. Cholan-24-oic Acid, 3,7-dihydroxy-, (3.alpha.,5.beta.,7.beta.)-
52. 7-beta-hydroxylithocholic Acid
53. Ursodexycholic Acid
54. Acido Ursodeossicolico [italian]
55. (r)-4-((3r,5s,7s,8r,9s,10s,13r,14s,17r)-3,7-dihydroxy-10,13-dimethylhexadecahydro-1h-cyclopenta[a]phenanthren-17-yl)pentanoic Acid
56. Acido Ursodeoxicolico
57. Acido Ursodeossicolico
58. Acido Ursodeoxicolico [inn-spanish]
59. Acidum Ursodeoxycholicum [inn-latin]
60. Acide Ursodesoxycholique
61. Acide Ursodesoxycholique [inn-french]
62. Acidum Ursodeoxycholicum
63. Actigall (tn)
64. Ursodiol (usp)
65. (4r)-4-[(1s,2s,5r,7s,9s,10r,11s,14r,15r)-5,9-dihydroxy-2,15-dimethyltetracyclo[8.7.0.0^{2,7}.0^{11,15}]heptadecan-14-yl]pentanoic Acid
66. Ccris 5502
67. Urso (tn)
68. Sr-01000737091
69. Ursodiol [usan:usp]
70. Einecs 204-879-3
71. Mfcd00003680
72. Ursodexycholate
73. Paptarom
74. Udiliv
75. Desol
76. Urdes
77. Unii-724l30y2qr
78. Urosdesoxycholate
79. 17-beta-(1-methyl-3-carboxypropyl)etiocholane-3-alpha,7-beta-diol
80. Ursodeoxycholoc Acid
81. Urosdesoxycholic Acid
82. 5beta-cholanic Acid-3alpha,7beta-diol
83. Ursodiol [inci]
84. Cholan-24-oic Acid, 3,7-dihydroxy-, (3alpha,5beta,7beta)-
85. 3alpha,7beta-dihydroxy-5beta-cholanic Acid
86. Ursodiol [mi]
87. Ursodiol (actigal Urso)
88. Ursodiol [vandf]
89. Prestwick0_000958
90. Prestwick1_000958
91. Prestwick2_000958
92. Prestwick3_000958
93. U0030
94. 7a-hydroxylithocholic Acid
95. Ursodiol [usp-rs]
96. Ec 204-879-3
97. U-9000
98. Schembl27200
99. Bspbio_000956
100. 4-10-00-01604 (beilstein Handbook Reference)
101. 7bet.-hydroxylithocholic Acid
102. Cid_31401
103. Mls001066373
104. Mls002548885
105. Spbio_003105
106. Ursodeoxycholic Acid, >=99%
107. Ursodiol [orange Book]
108. Bpbio1_001052
109. Gtpl7104
110. 7.beta.-hydroxylithocholic Acid
111. Ursodiol (ursodeoxycholic Acid)
112. 5-beta-cholan-24-oic Acid, 3-alpha,7-beta-dihydroxy-
113. Dtxsid6023731
114. Ursodiol [usp Monograph]
115. Bdbm53721
116. (3alpha,5beta,7beta,8xi)-3,7-dihydroxycholan-24-oic Acid
117. Hms1570p18
118. Hms2097p18
119. Hms2233l14
120. Hms3259a13
121. Hms3714p18
122. Ursodeoxycholic Acid (jp17/inn)
123. Ursodeoxycholic Acid [jan]
124. 3alpha,7beta-dihydroxycholanic Acid
125. Act02676
126. Cholan-24-oic Acid, 3,7-dihydroxy-, (3-alpha,5-beta,7-beta)-
127. Zinc3914809
128. 3a,7b-dihydroxy-5b-cholan-24-oate
129. Lmst04010033
130. S1643
131. Ursodeoxycholic Acid [mart.]
132. Ursodeoxycholic Acid [who-dd]
133. 5a-cholan-24-oic Acid-3a,7a-diol
134. Akos015955898
135. Ccg-220958
136. Cs-1932
137. Db01586
138. Ks-5243
139. Nc00487
140. Smp2_000012
141. 3.alpha.,7.beta.-dihydroxycholanic Acid
142. 3a,7a-dihydroxy-5a-holan-24-oic Acid
143. 3a,7b-dihydroxy-5b-cholan-24-oic Acid
144. Ncgc00179363-01
145. Ncgc00179363-12
146. Ac-18919
147. Cas#128-13-2
148. Cpd000058403
149. Hy-13771
150. Nci60_028904
151. Ursodeoxycholic Acid [ep Impurity]
152. Ursodeoxycholic Acid [ep Monograph]
153. Ab00513977
154. (3a,5b,7b)-3,7-dihydroxy-cholan-24-oate
155. 3alpha, 7beta-dihydroxy-5beta-cholanoic Acid
156. 5bet.-cholan-24-oic Acid-3alp.,7bet.-diol
157. (3a,5b,7b)-3,7-dihydroxycholan-24-oic Acid
158. C07880
159. D00734
160. ((3a,5b,7b)-3,7-dihydroxycholan-24-oic Acid
161. (3a,5b,7b)-3,7-dihydroxy-cholan-24-oic Acid
162. Ab00513977-09
163. Ab00513977_10
164. 3alp.,7bet.-dihydroxy-5bet.-cholan-24-oic Acid
165. A905413
166. Q241374
167. 3.alpha.,7.beta.-dihydroxy-5.beta.-cholanic Acid
168. J-005566
169. J-650210
170. Sr-01000737091-3
171. Sr-01000737091-4
172. Brd-k15697815-001-16-2
173. 3.alpha.,7.beta.-dihydroxy-5.beta.-cholan-24-oic Acid
174. Z2786051717
175. 3.alpha.,7.beta.-dihydroxy-5b-cholan-24-oic Acid
176. 5.beta.-cholan-24-oic Acid, 3.alpha.,7.beta.-dihydroxy-
177. Ursodiol, United States Pharmacopeia (usp) Reference Standard
178. Ursodeoxycholic Acid, British Pharmacopoeia (bp) Reference Standard
179. Ursodeoxycholic Acid, European Pharmacopoeia (ep) Reference Standard
180. 17.beta.-(1-methyl-3-carboxypropyl)etiocholane-3.alpha.,7.beta.-diol
181. 3,7-dihydroxycholan-24-oic Acid-, (3.alpha.,5.beta.,7.beta.)- #
182. Ursodeoxycholic Acid, 500 Mug/ml In Methanol, Certified Reference Material
183. Ursodiol, Pharmaceutical Secondary Standard; Certified Reference Material
184. Ursodeoxycholic Acid For System Suitability, European Pharmacopoeia (ep) Reference Standard
185. (4r)-4-[(1s,2s,5r,7s,9s,10r,11s,14r,15r)-5,9-dihydroxy-2,15-dimethyltetracyclo[8.7.0.02,1.011,1]heptadecan-14-yl]pentanoic Acid
186. (4r)-4-[(3r,5s,7s,8r,9s,10s,13r,14s,17r)-10,13-dimethyl-3,7-bis(oxidanyl)-2,3,4,5,6,7,8,9,11,12,14,15,16,17-tetradecahydro-1h-cyclopenta[a]phenanthren-17-yl]pentanoic Acid
187. (4r)-4-[(3r,5s,7s,8r,9s,10s,13r,14s,17r)-3,7-dihydroxy-10,13-dimethyl-2,3,4,5,6,7,8,9,11,12,14,15,16,17-tetradecahydro-1h-cyclopenta[a]phenanthren-17-yl]valeric Acid
188. 108609-27-4
| Molecular Weight | 392.6 g/mol |
|---|---|
| Molecular Formula | C24H40O4 |
| XLogP3 | 4.9 |
| Hydrogen Bond Donor Count | 3 |
| Hydrogen Bond Acceptor Count | 4 |
| Rotatable Bond Count | 4 |
| Exact Mass | 392.29265975 g/mol |
| Monoisotopic Mass | 392.29265975 g/mol |
| Topological Polar Surface Area | 77.8 Ų |
| Heavy Atom Count | 28 |
| Formal Charge | 0 |
| Complexity | 605 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 10 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
| 1 of 8 | |
|---|---|
| Drug Name | Actigall |
| Drug Label | Actigall is a bile acid available as 300 mg capsules suitable for oral administration.Actigall is ursodiol, USP (ursodeoxycholic acid), a naturally occurring bile acid found in small quantities in normal human bile and in the biles of certain other m... |
| Active Ingredient | Ursodiol |
| Dosage Form | Capsule |
| Route | Oral |
| Strength | 300mg |
| Market Status | Prescription |
| Company | Watson Pharms |
| 2 of 8 | |
|---|---|
| Drug Name | Urso 250 |
| PubMed Health | Ursodiol (By mouth) |
| Drug Classes | Gastrointestinal Agent |
| Drug Label | URSO 250 (ursodiol, 250 mg) is available as a film-coated tablet for oral administration. URSO Forte (ursodiol, 500 mg) is available as a scored film-coated tablet for oral administration. Ursodiol (ursodeoxycholic acid, UDCA) is a naturally occurrin... |
| Active Ingredient | Ursodiol |
| Dosage Form | Tablet |
| Route | Oral |
| Strength | 250mg |
| Market Status | Prescription |
| Company | Forest Labs |
| 3 of 8 | |
|---|---|
| Drug Name | Urso forte |
| PubMed Health | Ursodiol (By mouth) |
| Drug Classes | Gastrointestinal Agent |
| Drug Label | URSO 250 (ursodiol, 250 mg) is available as a film-coated tablet for oral administration. URSO Forte (ursodiol, 500 mg) is available as a scored film-coated tablet for oral administration. Ursodiol (ursodeoxycholic acid, UDCA) is a naturally occurrin... |
| Active Ingredient | Ursodiol |
| Dosage Form | Tablet |
| Route | Oral |
| Strength | 500mg |
| Market Status | Prescription |
| Company | Forest Labs |
| 4 of 8 | |
|---|---|
| Drug Name | Ursodiol |
| PubMed Health | Ursodiol (By mouth) |
| Drug Classes | Gastrointestinal Agent |
| Drug Label | Ursodiol Capsules, USP are a bile acid available as 300 mg capsules suitable for oral administration.Ursodiol, USP (ursodeoxycholic acid) is a naturally occurring bile acid found in small quantities in normal human bile and in the biles of certain ot... |
| Active Ingredient | Ursodiol |
| Dosage Form | Tablet; Capsule |
| Route | Oral |
| Strength | 250mg; 500mg; 300mg |
| Market Status | Prescription |
| Company | Corepharma; Epic Pharma; Par Pharm; Glenmark Generics; Teva Pharms; Lannett; Mylan; Impax Labs |
| 5 of 8 | |
|---|---|
| Drug Name | Actigall |
| Drug Label | Actigall is a bile acid available as 300 mg capsules suitable for oral administration.Actigall is ursodiol, USP (ursodeoxycholic acid), a naturally occurring bile acid found in small quantities in normal human bile and in the biles of certain other m... |
| Active Ingredient | Ursodiol |
| Dosage Form | Capsule |
| Route | Oral |
| Strength | 300mg |
| Market Status | Prescription |
| Company | Watson Pharms |
| 6 of 8 | |
|---|---|
| Drug Name | Urso 250 |
| PubMed Health | Ursodiol (By mouth) |
| Drug Classes | Gastrointestinal Agent |
| Drug Label | URSO 250 (ursodiol, 250 mg) is available as a film-coated tablet for oral administration. URSO Forte (ursodiol, 500 mg) is available as a scored film-coated tablet for oral administration. Ursodiol (ursodeoxycholic acid, UDCA) is a naturally occurrin... |
| Active Ingredient | Ursodiol |
| Dosage Form | Tablet |
| Route | Oral |
| Strength | 250mg |
| Market Status | Prescription |
| Company | Forest Labs |
| 7 of 8 | |
|---|---|
| Drug Name | Urso forte |
| PubMed Health | Ursodiol (By mouth) |
| Drug Classes | Gastrointestinal Agent |
| Drug Label | URSO 250 (ursodiol, 250 mg) is available as a film-coated tablet for oral administration. URSO Forte (ursodiol, 500 mg) is available as a scored film-coated tablet for oral administration. Ursodiol (ursodeoxycholic acid, UDCA) is a naturally occurrin... |
| Active Ingredient | Ursodiol |
| Dosage Form | Tablet |
| Route | Oral |
| Strength | 500mg |
| Market Status | Prescription |
| Company | Forest Labs |
| 8 of 8 | |
|---|---|
| Drug Name | Ursodiol |
| PubMed Health | Ursodiol (By mouth) |
| Drug Classes | Gastrointestinal Agent |
| Drug Label | Ursodiol Capsules, USP are a bile acid available as 300 mg capsules suitable for oral administration.Ursodiol, USP (ursodeoxycholic acid) is a naturally occurring bile acid found in small quantities in normal human bile and in the biles of certain ot... |
| Active Ingredient | Ursodiol |
| Dosage Form | Tablet; Capsule |
| Route | Oral |
| Strength | 250mg; 500mg; 300mg |
| Market Status | Prescription |
| Company | Corepharma; Epic Pharma; Par Pharm; Glenmark Generics; Teva Pharms; Lannett; Mylan; Impax Labs |
The drug decreases the absorption of cholesterol and is used to dissolve (cholesterol) gallstones in patients as an alternative to a surgical procedure to remove the gallstones.
FDA Label
Ursodiol (commonly known as ursodeoxycholic acid) is a product of metabolism of bacteria in the intestine. It is considered a secondary bile acid. The other type of bile acid, primary bile acids, are produced hepatically and subsequently stored in the gallbladder. When primary bile acids are secreted into the large intestine, they can be broken down into secondary bile acids by bacteria present in the intestine. Both types of bile acids assist in the metabolism of dietary fat. Ursodeoxycholic acid regulates cholesterol levels by slowing the rate at which the intestine is able to absorb cholesterol and also acts to break down micelles, which contain cholesterol. Because of this property, ursodeoxycholic acid is used to treat gall stones non-surgically.
Cholagogues and Choleretics
Gastrointestinal agents that stimulate the flow of bile into the duodenum (cholagogues) or stimulate the production of bile by the liver (choleretic). (See all compounds classified as Cholagogues and Choleretics.)
A05AA02
S76 | LUXPHARMA | Pharmaceuticals Marketed in Luxembourg | Pharmaceuticals marketed in Luxembourg, as published by d'Gesondheetskeess (CNS, la caisse nationale de sante, www.cns.lu), mapped by name to structures using CompTox by R. Singh et al. (in prep.). List downloaded from https://cns.public.lu/en/legislations/textes-coordonnes/liste-med-comm.html. Dataset DOI:10.5281/zenodo.4587355
A - Alimentary tract and metabolism
A05 - Bile and liver therapy
A05A - Bile therapy
A05AA - Bile acids and derivatives
A05AA02 - Ursodeoxycholic acid
Route of Elimination
Only small quantities of ursodiol appear in the systemic circulation and very small amounts are excreted into urine. Eighty percent of lithocholic acid formed in the small bowel is excreted in the feces, but the 20% that is absorbed is sulfated at the 3-hydroxyl group in the liver to relatively insoluble lithocholyl conjugates which are excreted into bile and lost in feces.
Ursodeoxycholic acid reduces elevated liver enzyme levels by facilitating bile flow through the liver and protecting liver cells. The main mechanism if anticholelithic. Although the exact process of ursodiol's anticholelithic action is not completely understood, it is thought that the drug is concentrated in bile and decreases biliary cholesterol by suppressing hepatic synthesis and secretion of cholesterol and by inhibiting its intestinal absorption. The reduced cholesterol saturation permits the gradual solubilization of cholesterol from gallstones, resulting in their eventual dissolution.
DRUG PRODUCT COMPOSITIONS

Related Excipient Companies

Excipients by Applications
Global Sales Information

Market Place

Reply
01 Apr 2026
Reply
26 Feb 2026
Reply
31 Oct 2025
Reply
17 Oct 2025
Reply
04 Aug 2025
Reply
20 May 2025
Reply
26 Apr 2025
Reply
27 Mar 2025
Reply
12 Sep 2024
Reply
05 Aug 2024
 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
REF. STANDARDS & IMPURITIES

ANALYTICAL

ABOUT THIS PAGE
69
PharmaCompass offers a list of Ursodeoxycholic Acid API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Ursodeoxycholic Acid manufacturer or Ursodeoxycholic Acid supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Ursodeoxycholic Acid manufacturer or Ursodeoxycholic Acid supplier.
A UDCA manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of UDCA, including repackagers and relabelers. The FDA regulates UDCA manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. UDCA API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
click here to find a list of UDCA manufacturers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PhamaCompass.
A UDCA supplier is an individual or a company that provides UDCA active pharmaceutical ingredient (API) or UDCA finished formulations upon request. The UDCA suppliers may include UDCA API manufacturers, exporters, distributors and traders.
click here to find a list of UDCA suppliers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PharmaCompass.
A UDCA DMF (Drug Master File) is a document detailing the whole manufacturing process of UDCA active pharmaceutical ingredient (API) in detail. Different forms of UDCA DMFs exist exist since differing nations have different regulations, such as UDCA USDMF, ASMF (EDMF), JDMF, CDMF, etc.
A UDCA DMF submitted to regulatory agencies in the US is known as a USDMF. UDCA USDMF includes data on UDCA's chemical properties, information on the facilities and procedures used, and details about packaging and storage. The UDCA USDMF is kept confidential to protect the manufacturer’s intellectual property.
click here to find a list of UDCA suppliers with USDMF on PharmaCompass.
The Pharmaceuticals and Medical Devices Agency (PMDA) established the Japan Drug Master File (JDMF), also known as the Master File (MF), to permit Japanese and foreign manufacturers of drug substances, intermediates, excipients, raw materials, and packaging materials (‘Products’) to voluntarily register confidential information about the production and management of their products in Japan.
The UDCA Drug Master File in Japan (UDCA JDMF) empowers UDCA API manufacturers to present comprehensive information (e.g., production methods, data, etc.) to the review authority, i.e., PMDA (Pharmaceuticals & Medical Devices Agency).
PMDA reviews the UDCA JDMF during the approval evaluation for pharmaceutical products. At the time of UDCA JDMF registration, PMDA checks if the format is accurate, if the necessary items have been included (application), and if data has been attached.
click here to find a list of UDCA suppliers with JDMF on PharmaCompass.
In Korea, the Ministry of Food and Drug Safety (MFDS) is in charge of regulating pharmaceutical products and services.
Pharmaceutical companies submit a UDCA Drug Master File in Korea (UDCA KDMF) to the MFDS, which includes comprehensive information about the production, processing, facilities, materials, packaging, and testing of UDCA. The MFDS reviews the UDCA KDMF as part of the drug registration process and uses the information provided in the UDCA KDMF to evaluate the safety and efficacy of the drug.
After submitting a UDCA KDMF to the MFDS, the registered manufacturer can provide importers or distributors with the registration number without revealing confidential information to Korean business partners. Applicants seeking to register their UDCA API can apply through the Korea Drug Master File (KDMF).
click here to find a list of UDCA suppliers with KDMF on PharmaCompass.
A UDCA CEP of the European Pharmacopoeia monograph is often referred to as a UDCA Certificate of Suitability (COS). The purpose of a UDCA CEP is to show that the European Pharmacopoeia monograph adequately controls the purity of UDCA EP produced by a given manufacturer. Suppliers of raw materials can prove the suitability of UDCA to their clients by showing that a UDCA CEP has been issued for it. The manufacturer submits a UDCA CEP (COS) as part of the market authorization procedure, and it takes on the role of a UDCA CEP holder for the record. Additionally, the data presented in the UDCA CEP (COS) is managed confidentially and offers a centralized system acknowledged by numerous nations, exactly like the UDCA DMF.
A UDCA CEP (COS) is recognised by all 36 nations that make up the European Pharmacopoeia Convention. UDCA CEPs may be accepted in nations that are not members of the Ph. Eur. at the discretion of the authorities there.
click here to find a list of UDCA suppliers with CEP (COS) on PharmaCompass.
A UDCA written confirmation (UDCA WC) is an official document issued by a regulatory agency to a UDCA manufacturer, verifying that the manufacturing facility of a UDCA active pharmaceutical ingredient (API) adheres to the Good Manufacturing Practices (GMP) regulations of the importing country. When exporting UDCA APIs or UDCA finished pharmaceutical products to another nation, regulatory agencies frequently require a UDCA WC (written confirmation) as part of the regulatory process.
click here to find a list of UDCA suppliers with Written Confirmation (WC) on PharmaCompass.
National Drug Code is a comprehensive database maintained by the FDA that contains information on all drugs marketed in the US. This directory includes information about finished drug products, unfinished drug products, and compounded drug products, including those containing UDCA as an active pharmaceutical ingredient (API).
The FDA updates the NDC directory daily. The NDC numbers for UDCA API and other APIs are published in this directory by the FDA.
The NDC unfinished drugs database includes product listing information submitted for all unfinished drugs, such as active pharmaceutical ingredients (APIs), drugs intended for further processing and bulk drug substances for compounding.
Pharmaceutical companies that manufacture UDCA as an active pharmaceutical ingredient (API) must furnish the FDA with an updated record of all drugs that they produce, prepare, propagate, compound, or process for commercial distribution in the US at their facilities.
The NDC directory also contains data on finished compounded human drug products that contain UDCA and are produced by outsourcing facilities. While these outsourcing facilities are not mandated to assign a UDCA NDC to their finished compounded human drug products, they may choose to do so.
click here to find a list of UDCA suppliers with NDC on PharmaCompass.
UDCA Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of UDCA GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right UDCA GMP manufacturer or UDCA GMP API supplier for your needs.
A UDCA CoA (Certificate of Analysis) is a formal document that attests to UDCA's compliance with UDCA specifications and serves as a tool for batch-level quality control.
UDCA CoA mostly includes findings from lab analyses of a specific batch. For each UDCA CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
UDCA may be tested according to a variety of international standards, such as European Pharmacopoeia (UDCA EP), UDCA JP (Japanese Pharmacopeia) and the US Pharmacopoeia (UDCA USP).

