
Synopsis
Synopsis
0
CEP/COS
0
EU WC
0
KDMF
0
NDC API
0
VMF
0
FDA Orange Book
0
Canada
0
Australia
0
South Africa
DRUG PRODUCT COMPOSITIONS
0
US Patents
0
US Exclusivities
0
Health Canada Patents
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
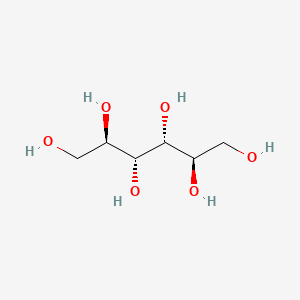

1. (l)-mannitol
2. Osmitrol
3. Osmofundin
1. D-mannitol
2. 69-65-8
3. Mannite
4. Osmitrol
5. Manna Sugar
6. 87-78-5
7. Cordycepic Acid
8. Osmofundin
9. Resectisol
10. D-(-)-mannitol
11. Mannit
12. Osmosal
13. Mannazucker
14. Mannidex
15. Mannigen
16. Mannistol
17. Diosmol
18. Invenex
19. Isotol
20. Mannitol, D-
21. Marine Crystal
22. Maniton-s
23. Mannogem 2080
24. (2r,3r,4r,5r)-hexane-1,2,3,4,5,6-hexol
25. (2r,3r,4r,5r)-hexane-1,2,3,4,5,6-hexaol
26. Bronchitol
27. Mannitol (van)
28. Aridol
29. Sdm No. 35
30. Hexahydroxyhexane
31. Dl-mannitol
32. Nci-c50362
33. Mannitol (usp)
34. Mannitol [usp]
35. Brn 1721898
36. Chebi:16899
37. Mfcd00064287
38. 3owl53l36a
39. Ins No.421
40. 133-43-7
41. E-421
42. Mannitol-d
43. Ins-421
44. Mannitol 5%
45. Nsc-407017
46. Mannitol [usan]
47. 1,2,3,4,5,6-hexanehexol
48. Mannitol 10%
49. Mannitol 15%
50. Mannitol 20%
51. Ncgc00164246-01
52. E421
53. Mannidex 16700
54. Dsstox_cid_3235
55. Osmitrol 5% In Water
56. Dsstox_rid_76936
57. Osmitrol 10% In Water
58. Osmitrol 15% In Water
59. Osmitrol 20% In Water
60. Dsstox_gsid_23235
61. 287100-73-6
62. Mtl
63. Mannitol 25%
64. Resectisol In Plastic Container
65. Mannitol 5% In Plastic Container
66. Mannitol 10% In Plastic Container
67. Mannitol 15% In Plastic Container
68. Mannitol 20% In Plastic Container
69. Cas-69-65-8
70. Osmitrol (tn)
71. Smr000857324
72. Ccris 369
73. Hsdb 714
74. Sr-01000838849
75. Nsc 9256
76. Einecs 200-711-8
77. Einecs 201-770-2
78. Osmitrol 5% In Water In Plastic Container
79. Nsc 407017
80. Osmitrol 10% In Water In Plastic Container
81. Osmitrol 15% In Water In Plastic Container
82. Osmitrol 20% In Water In Plastic Container
83. Unii-3owl53l36a
84. Cordycepate
85. Mannitolum
86. Manitol
87. D-mitobronitol
88. Mannitol 10% W/ Dextrose 5% In Distilled Water
89. Ai3-19511
90. Maniton S
91. Mannit P
92. D-mannit
93. D-?mannitol
94. Bronchitol (tn)
95. (d)-mannitol
96. Mannitol 25
97. Mannitol 35
98. Mannitol 60
99. D(-)mannitol
100. Mannitol,(s)
101. Pearlitol 25 C
102. Pearlitol 50 C
103. Mannitol M300
104. Pearlitol 160 C
105. Mannitol 200
106. Mannitol 300
107. Pearlitol 200 Sd
108. Pearlitol 300 Dc
109. Mannitol 2080
110. D-mannitol (osmitrol)
111. D-mannitol (jp17)
112. Mannitol [hsdb]
113. Mannitol [inci]
114. Mannitol [fcc]
115. Mannitol 15% W/ Dextrose 5% In Sodium Chloride 0.45%
116. Mannitol 5% W/ Dextrose 5% In Sodium Chloride 0.12%
117. Crystalline Mannitol Fine
118. Mannitol [ii]
119. Mannitol [mi]
120. D-[2-13c]glucitol
121. Mannitol [vandf]
122. D-mannitol, Acs Reagent
123. D-mannitol, >=98%
124. M0044
125. D-mannitol [jan]
126. Mannitol [mart.]
127. Schembl919
128. Bmse000099
129. Chembl689
130. Epitope Id:114705
131. Mannitol [usp-rs]
132. Mannitol [who-dd]
133. Isomalt Impurity, Mannitol-
134. Crystalline Mannitol Standard
135. D-mannitol [who-ip]
136. 4-01-00-02841 (beilstein Handbook Reference)
137. Ed1d1e61-fefb-430a-afdc-d1f4a957fc3d
138. Mls001335977
139. Mls001335978
140. D-mannitol, Biochemical Grade
141. Crystalline Mannitol Extra-fine
142. Mannitol [ep Impurity]
143. Mannitol [orange Book]
144. D-mannitol, Ar, >=99%
145. D-mannitol, Lr, >=99%
146. Mannitol [ep Monograph]
147. Dtxsid1023235
148. Mannitol [usp Monograph]
149. Dtxsid30858955
150. Mannitolum [who-ip Latin]
151. Hms2230n11
152. Amy33410
153. Hy-n0378
154. Zinc2041302
155. Tox21_112092
156. Tox21_201487
157. Tox21_300483
158. Potassiumnonafluoro-t-butoxide
159. Akos006280947
160. D-mannitol, Plant Cell Culture Tested
161. Tox21_112092_1
162. Bcp9000575
163. Ccg-266445
164. D-mannit 1000 Microg/ml In Methanol
165. D-mannitol, Bioxtra, >=98% (gc)
166. Db00742
167. D-mannitol, Nist(r) Srm(r) 920
168. Isomalt Impurity B [ep Impurity]
169. Ncgc00164246-03
170. Ncgc00164246-04
171. Ncgc00164246-05
172. Ncgc00254277-01
173. Ncgc00259038-01
174. 85085-15-0
175. Ac-12776
176. Ac-14054
177. As-30501
178. D-mannitol, Tested According To Ph.eur.
179. D-mannitol, P.a., 96.0-101.5%
180. D-mannitol, Saj First Grade, >=99.0%
181. Sorbitol-mannitol Component Mannitol
182. B2090
183. D-mannitol, Saj Special Grade, >=99.0%
184. E 421
185. S2381
186. Sw220287-1
187. Mannitol Component Of Sorbitol-mannitol
188. C00392
189. D00062
190. Ab00443917_06
191. 065m361
192. Isomalt Impurity, Mannitol- [usp Impurity]
193. Q407646
194. Wurcs=2.0/1,1,0/[h1122h]/1/
195. Q-101039
196. Sr-01000838849-3
197. Sr-01000838849-4
198. D-mannitol, Acs Reagent, For Microbiology, >=99.0%
199. Lactitol Monohydrate Impurity C [ep Impurity]
200. D-mannitol, Meets Ep, Fcc, Usp Testing Specifications
201. Mannitol, European Pharmacopoeia (ep) Reference Standard
202. D-mannitol, Bioultra, >=99.0% (sum Of Enantiomers, Hplc)
203. Mannitol, United States Pharmacopeia (usp) Reference Standard
204. D-mannitol, >=99.9999% (metals Basis), For Boron Determination
205. Mannitol, Pharmaceutical Secondary Standard; Certified Reference Material
| Molecular Weight | 182.17 g/mol |
|---|---|
| Molecular Formula | C6H14O6 |
| XLogP3 | -3.1 |
| Hydrogen Bond Donor Count | 6 |
| Hydrogen Bond Acceptor Count | 6 |
| Rotatable Bond Count | 5 |
| Exact Mass | 182.07903816 g/mol |
| Monoisotopic Mass | 182.07903816 g/mol |
| Topological Polar Surface Area | 121 Ų |
| Heavy Atom Count | 12 |
| Formal Charge | 0 |
| Complexity | 105 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 4 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
| 1 of 4 | |
|---|---|
| Drug Name | Aridol |
| PubMed Health | Mannitol |
| Drug Classes | Cardiovascular Agent, Diagnostic Agent, Bronchial, Diagnostic Agent, Kidney Function |
| Active Ingredient | Mannitol |
| Dosage Form | Powder |
| Route | inhalation |
| Strength | 10mg; 40mg; 20mg |
| Market Status | Prescription |
| Company | Pharmaxis |
| 2 of 4 | |
|---|---|
| Drug Name | Resectisol in plastic container |
| Active Ingredient | Mannitol |
| Dosage Form | Solution |
| Route | Irrigation |
| Strength | 5gm/100ml |
| Market Status | Prescription |
| Company | B Braun |
| 3 of 4 | |
|---|---|
| Drug Name | Aridol |
| PubMed Health | Mannitol |
| Drug Classes | Cardiovascular Agent, Diagnostic Agent, Bronchial, Diagnostic Agent, Kidney Function |
| Active Ingredient | Mannitol |
| Dosage Form | Powder |
| Route | inhalation |
| Strength | 10mg; 40mg; 20mg |
| Market Status | Prescription |
| Company | Pharmaxis |
| 4 of 4 | |
|---|---|
| Drug Name | Resectisol in plastic container |
| Active Ingredient | Mannitol |
| Dosage Form | Solution |
| Route | Irrigation |
| Strength | 5gm/100ml |
| Market Status | Prescription |
| Company | B Braun |
Diuretics, Osmotic
National Library of Medicine's Medical Subject Headings online file (MeSH, 1999)
MEDICATION (VET): INTRAAORTIC PRETREATMENT WITH 10% MANNITOL SOLN PRIOR TO INTRAAORTIC CONTRAST ANGIOGRAPHY PROCEDURES GIVES RENAL PROTECTION & REDUCES INCIDENCE OF PARAPLEGIA & AZOTEMIA IN DOGS.
Rossoff, I.S. Handbook of Veterinary Drugs. New York: Springer Publishing Company, 1974., p. 329
MEDICATION (VET): IN DOGS AS OSMOTIC DIURETIC CAUSING CELLULAR DEHYDRATION, TO REDUCE INTRAOCULAR PRESSURE IN GLAUCOMA, & TO REDUCE CEREBRAL EDEMA FOLLOWING SURGERY OR INJURY.
Osol, A. and J.E. Hoover, et al. (eds.). Remington's Pharmaceutical Sciences. 15th ed. Easton, Pennsylvania: Mack Publishing Co., 1975., p. 863
DOSE FOR REDUCTION OF INTRACRANIAL PRESSURE & BRAIN MASS PRIOR TO NEUROSURGERY, OR FOR REDUCTION OF INTRAOCULAR TENSION...OF CONGESTIVE GLAUCOMA OR FOR OPHTHALMIC SURGERY, IS 1.5 TO 2 G/KG, GIVEN AS 15 OR 20% SOLN OVER PERIOD OF 30 TO 60 MIN.
Gilman, A. G., L. S. Goodman, and A. Gilman. (eds.). Goodman and Gilman's The Pharmacological Basis of Therapeutics. 6th ed. New York: Macmillan Publishing Co., Inc. 1980., p. 896
For more Therapeutic Uses (Complete) data for D-MANNITOL (15 total), please visit the HSDB record page.
IN EDEMATOUS STATES ASSOC WITH DIMINISHED CARDIAC RESERVE, ADMIN OF MANNITOL INTRODUCES A RISK THAT MAY FAR OUTWEIGH ANY THERAPEUTIC BENEFIT.
Goodman, L.S., and A. Gilman. (eds.) The Pharmacological Basis of Therapeutics. 5th ed. New York: Macmillan Publishing Co., Inc., 1975., p. 822
CONTRAINDICATIONS TO ADMIN OF MANNITOL INCL RENAL DISEASE...ANURIA, MARKED PULMONARY CONGESTION OR EDEMA, MARKED DEHYDRATION, & INTRACRANIAL HEMORRHAGE... MANNITOL SHOULD BE TERMINATED IF PATIENTS DEVELOPS...PROGRESSIVE RENAL DYSFUNCTION, HEART FAILURE, OR PULMONARY CONGESTION.
Gilman, A. G., L. S. Goodman, and A. Gilman. (eds.). Goodman and Gilman's The Pharmacological Basis of Therapeutics. 6th ed. New York: Macmillan Publishing Co., Inc. 1980., p. 896
ITS SAFE USE DURING PREGNANCY & IN CHILDREN UNDER 12 YR OF AGE HAS NOT BEEN ESTABLISHED.
Osol, A. (ed.). Remington's Pharmaceutical Sciences. 16th ed. Easton, Pennsylvania: Mack Publishing Co., 1980., p. 875
FACTITIOUS HYPOPHOSPHATEMIA WAS OBSERVED IN A PATIENT RECEIVING LARGE AMT OF IV MANNITOL. CONCN AS LOW AS 25 MMOL/L INHIBITED PHOSPHORUS MEASUREMENT BY DUPONT ACA ENDPOINT METHOD; A KINETIC METHOD WAS UNAFFECTED. MECHANISM OF MANNITOL INTERFERENCE WAS BINDING TO MOLYBDATE IN REACTION, DECR RATE OF COLOR DEVELOPMENT & ENDPOINT MEASUREMENT.
PMID:6793267 DONHOWE JM ET AL; CLIN CHEM 27 (10): 1765 (1981)
For more Drug Warnings (Complete) data for D-MANNITOL (9 total), please visit the HSDB record page.
Used for the promotion of diuresis before irreversible renal failure becomes established, the reduction of intracranial pressure, the treatment of cerebral edema, and the promotion of urinary excretion of toxic substances. Mannitol is also indicated as add-on maintenance therapy for improving pulmonary function in cystic fibrosis patients aged 18 and over who have passed the BRONCHITOL tolerance test (BTT). It is recommended that patients take an orally inhaled short-acting bronchodilator 5-15 minutes prior to every inhaled mannitol dose.
FDA Label
Bronchitol is indicated for the treatment of cystic fibrosis (CF) in adults aged 18 years and above as an add-on therapy to best standard of care.
Cystic fibrosis with pulmonary disease
Bowel cleansing prior to clinical procedures
Chemically, mannitol is an alcohol and a sugar, or a polyol; it is similar to xylitol or sorbitol. However, mannitol has a tendency to lose a hydrogen ion in aqueous solutions, which causes the solution to become acidic. For this reason, it is not uncommon to add a substance to adjust its pH, such as sodium bicarbonate. Mannitol is commonly used to increase urine production (diuretic). It is also used to treat or prevent medical conditions that are caused by an increase in body fluids/water (e.g., cerebral edema, glaucoma, kidney failure). Mannitol is frequently given along with other diuretics (e.g., furosemide, chlorothiazide) and/or IV fluid replacement. Inhaled mannitol has the possibility to cause bronchospasm and hemoptysis; the occurrence of either should lead to discontinuation of inhaled mannitol.
Sweetening Agents
Substances that sweeten food, beverages, medications, etc., such as sugar, saccharine or other low-calorie synthetic products. (From Random House Unabridged Dictionary, 2d ed) (See all compounds classified as Sweetening Agents.)
Diuretics, Osmotic
Compounds that increase urine volume by increasing the amount of osmotically active solute in the urine. Osmotic diuretics also increase the osmolarity of plasma. (See all compounds classified as Diuretics, Osmotic.)
R05CB16
B05BC01
S76 | LUXPHARMA | Pharmaceuticals Marketed in Luxembourg | Pharmaceuticals marketed in Luxembourg, as published by d'Gesondheetskeess (CNS, la caisse nationale de sante, www.cns.lu), mapped by name to structures using CompTox by R. Singh et al. (in prep.). List downloaded from https://cns.public.lu/en/legislations/textes-coordonnes/liste-med-comm.html. Dataset DOI:10.5281/zenodo.4587355
A - Alimentary tract and metabolism
A06 - Drugs for constipation
A06A - Drugs for constipation
A06AD - Osmotically acting laxatives
A06AD16 - Mannitol
B - Blood and blood forming organs
B05 - Blood substitutes and perfusion solutions
B05B - I.v. solutions
B05BC - Solutions producing osmotic diuresis
B05BC01 - Mannitol
B - Blood and blood forming organs
B05 - Blood substitutes and perfusion solutions
B05C - Irrigating solutions
B05CX - Other irrigating solutions
B05CX04 - Mannitol
R - Respiratory system
R05 - Cough and cold preparations
R05C - Expectorants, excl. combinations with cough suppressants
R05CB - Mucolytics
R05CB16 - Mannitol
V - Various
V04 - Diagnostic agents
V04C - Other diagnostic agents
V04CX - Other diagnostic agents
V04CX04 - Mannitol
Absorption
Approximately 7% of ingested mannitol is absorbed during gastrointestinal perfusion in uremic patients. Inhalation of 635 mg of mannitol powder yields a plasma Cmax of 13.71 g/mL in 1.5 hours (Tmax) and a mean systemic AUC of 73.15 g\*h/mL.
Route of Elimination
Mannitol is primarily excreted unchanged in the urine. Following oral inhalation of 635 mg of mannitol in healthy volunteers, 55% of the total dose was recovered unchanged in the urine; following oral or intravenous administration of 500 mg, the corresponding values were 54 and 87%, respectively.
Volume of Distribution
Mannitol administered intravenously has a volume of distribution of 34.3 L.
Clearance
Intravenous administration of mannitol yields a total clearance of 5.1 L/hr and renal clearance of 4.4 L/hr.
MANNITOL IS GENERALLY REGARDED AS BEING UNABSORBED FROM GI TRACT. HOWEVER, RECENT WORK CONTRADICTS THIS BELIEF, FOR 18% OF ORAL DOSE OF D-(14)C MANNITOL WAS RECOVERED UNCHANGED IN 48-HR URINE OF HUMAN SUBJECTS & UP TO 19% AS CO2 IN EXPIRED AIR IN 12 HR. 32% PRESENT IN FECES IN 48 HR...UNABSORBED MATERIAL.
The Chemical Society. Foreign Compound Metabolism in Mammals. Volume 2: A Review of the Literature Published Between 1970 and 1971. London: The Chemical Society, 1972., p. 160
SUBSTANCES /MANNITOL/ HAVING APPARENT VOL OF DISTRIBUTION CORRESPONDING TO TOTAL EXTRACELLUR WATER, WHICH IS ABOUT 20% BODY WT...PENETRATE CAPILLARY MEMBRANES BUT DO NOT PENETRATE CELLULAR MEMBRANES.
LaDu, B.N., H.G. Mandel, and E.L. Way. Fundamentals of Drug Metabolism and Disposition. Baltimore: Williams and Wilkins, 1971., p. 52
MANNITOL UNDERGOES VERY LITTLE REABSORPTION, & FOR MANY PRACTICAL PURPOSES TUBULE MAY BE CONSIDERED TO BE IMPERMEABLE TO IT. ...OSMOTIC DIURETICS, WHICH, BY DEFINITION, ARE POORLY REABSORBED BY RENAL TUBULES, ARE ALSO NOT ABSORBED FROM GI TRACT. ...THESE AGENTS MUST BE ADMIN PARENTERALLY...TO ACHIEVE EFFECTIVE PLASMA CONCN.
Goodman, L.S., and A. Gilman. (eds.) The Pharmacological Basis of Therapeutics. 5th ed. New York: Macmillan Publishing Co., Inc., 1975., p. 821
Mannitol is metabolized only slightly, if at all, to glycogen in the liver.
...POLYHYDRIC SUGAR ALC...MANNITOL (C6H14O6)...LARGELY EXCRETED UNCHANGED IN URINE.
Parke, D. V. The Biochemistry of Foreign Compounds. Oxford: Pergamon Press, 1968., p. 140
MANNITOL OCCURS IN LARGE AMT IN SPORES OF ASPERGILLUS ORYZAE, WHERE IT IS RAPIDLY METABOLIZED IN EARLY STAGES OF GERMINATION. IT IS CONVERTED TO FRUCTOSE BY D-MANNITOL DEHYDROGENASE...
Bergmeyer, H.W. (ed.). Methods of Enzymatic Analysis. 2nd English ed. New York City: Academic Press, 1974., p. 1271
...FATE OF MANNITOL IN ANIMAL BODY (MONKEYS, RABBITS, RATS, DOGS, ETC) AFTER ABSORPTION FROM DIGESTIVE TRACT INCL LIMITED CONVERSION TO GLYCOGEN IN LIVER & ELIMINATION OF BALANCE UNCHANGED IN URINE.
Rossoff, I.S. Handbook of Veterinary Drugs. New York: Springer Publishing Company, 1974., p. 329
Mannitol has an elimination half-life of 4.7 hours following oral administration; the mean terminal elimination half-life is similar regardless of administration route (oral, inhalation, and intravenous.
Mannitol is an osmotic diuretic that is metabolically inert in humans and occurs naturally, as a sugar or sugar alcohol, in fruits and vegetables. Mannitol elevates blood plasma osmolality, resulting in enhanced flow of water from tissues, including the brain and cerebrospinal fluid, into interstitial fluid and plasma. As a result, cerebral edema, elevated intracranial pressure, and cerebrospinal fluid volume and pressure may be reduced. As a diurectic mannitol induces diuresis because it is not reabsorbed in the renal tubule, thereby increasing the osmolality of the glomerular filtrate, facilitating excretion of water, and inhibiting the renal tubular reabsorption of sodium, chloride, and other solutes. Mannitol promotes the urinary excretion of toxic materials and protects against nephrotoxicity by preventing the concentration of toxic substances in the tubular fluid. As an Antiglaucoma agent mannitol levates blood plasma osmolarity, resulting in enhanced flow of water from the eye into plasma and a consequent reduction in intraocular pressure. As a renal function diagnostic aid mannitol is freely filtered by the glomeruli with less than 10% tubular reabsorption. Therefore, its urinary excretion rate may serve as a measurement of glomerular filtration rate (GFR). The exact mechanism of action of inhaled mannitol in the symptomatic maintenance treatment of cystic fibrosis remains unclear. It is hypothesized that mannitol produces an osmotic gradient across the airway epithelium that draws fluid into the extracellular space and alters the properties of the airway surface mucus layer, allowing easier mucociliary clearance.
MANNITOL IS.../USED/ IN PROPHYLAXIS OF ACUTE RENAL FAILURE. IT IS USED FOR THIS PURPOSE IN CONDITIONS AS DIVERSE AS CARDIOVASCULAR OPERATIONS, SEVERE TRAUMATIC INJURY, OPERATIONS IN THE PRESENCE OF SEVERE JAUNDICE, AND MGMNT OF HEMOLYTIC TRANSFUSION REACTIONS. IN EACH OF THESE CONDITIONS, A PRECIPITOUS FALL IN THE FLOW OF URINE MAY BE ANTICIPATED EITHER AS THE RESULT OF AN ACUTELY REDUCED FILTRATION RATE OR FROM ACUTE CHANGES IN TUBULAR PERMEABILITY. THE LATTER MAY BE CONSEQUENCE OF THE PRESENCE OF NOXIOUS AGENT WITHIN THE TUBULAR FLUID IN EXCESSIVELY HIGH CONCN, IN SOME INSTANCES SUFFICIENT TO RESULT IN ACTUAL PRECIPITATION. IN THESE SITUATIONS, MANNITOL EXERTS OSMOTIC EFFECT WITHIN THE TUBULAR FLUID, INHIBITS WATER REABSORPTION, & MAINTAINS THE RATE OF URINE FLOW. ...CONCN OF TOXIC AGENT WITHIN TUBULAR FLUID DOES NOT REACH EXCESSIVELY HIGH LEVELS THAT OTHERWISE WOULD HAVE BEEN ACHIEVED BY MORE COMPLETE REABSORPTION OF WATER.
Gilman, A. G., L. S. Goodman, and A. Gilman. (eds.). Goodman and Gilman's The Pharmacological Basis of Therapeutics. 6th ed. New York: Macmillan Publishing Co., Inc. 1980., p. 895
...EVEN THOUGH /GLOMERULAR/ FILTRATION RATE IS REDUCED, MANNITOL IS STILL FILTERED @ GLOMERULUS. THE TUBULAR IMPERMEABILITY TO MANNITOL IS NOT ALTERED BY ACUTE RENAL ISCHEMIA OF SHORT DURATION. HENCE, THE MANNITOL THAT IS FILTERED IS ALSO EXCRETED IN THE VOIDED URINE. UNREABSORBED SOLUTE LIMITS BACK DIFFUSION OF WATER. ...URINE VOL CAN BE MAINTAINED EVEN IN PRESENCE OF DECR GLOMERULAR FILTRATION.
Gilman, A. G., L. S. Goodman, and A. Gilman. (eds.). Goodman and Gilman's The Pharmacological Basis of Therapeutics. 6th ed. New York: Macmillan Publishing Co., Inc. 1980., p. 894
Global Sales Information

Market Place

REF. STANDARDS & IMPURITIES

ABOUT THIS PAGE
96
PharmaCompass offers a list of Mannitol API API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Mannitol API manufacturer or Mannitol API supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Mannitol API manufacturer or Mannitol API supplier.
A Tox21_300483 manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Tox21_300483, including repackagers and relabelers. The FDA regulates Tox21_300483 manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Tox21_300483 API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
click here to find a list of Tox21_300483 manufacturers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PhamaCompass.
A Tox21_300483 supplier is an individual or a company that provides Tox21_300483 active pharmaceutical ingredient (API) or Tox21_300483 finished formulations upon request. The Tox21_300483 suppliers may include Tox21_300483 API manufacturers, exporters, distributors and traders.
click here to find a list of Tox21_300483 suppliers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PharmaCompass.
A Tox21_300483 DMF (Drug Master File) is a document detailing the whole manufacturing process of Tox21_300483 active pharmaceutical ingredient (API) in detail. Different forms of Tox21_300483 DMFs exist exist since differing nations have different regulations, such as Tox21_300483 USDMF, ASMF (EDMF), JDMF, CDMF, etc.
A Tox21_300483 DMF submitted to regulatory agencies in the US is known as a USDMF. Tox21_300483 USDMF includes data on Tox21_300483's chemical properties, information on the facilities and procedures used, and details about packaging and storage. The Tox21_300483 USDMF is kept confidential to protect the manufacturer’s intellectual property.
click here to find a list of Tox21_300483 suppliers with USDMF on PharmaCompass.
The Pharmaceuticals and Medical Devices Agency (PMDA) established the Japan Drug Master File (JDMF), also known as the Master File (MF), to permit Japanese and foreign manufacturers of drug substances, intermediates, excipients, raw materials, and packaging materials (‘Products’) to voluntarily register confidential information about the production and management of their products in Japan.
The Tox21_300483 Drug Master File in Japan (Tox21_300483 JDMF) empowers Tox21_300483 API manufacturers to present comprehensive information (e.g., production methods, data, etc.) to the review authority, i.e., PMDA (Pharmaceuticals & Medical Devices Agency).
PMDA reviews the Tox21_300483 JDMF during the approval evaluation for pharmaceutical products. At the time of Tox21_300483 JDMF registration, PMDA checks if the format is accurate, if the necessary items have been included (application), and if data has been attached.
click here to find a list of Tox21_300483 suppliers with JDMF on PharmaCompass.
Tox21_300483 Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Tox21_300483 GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Tox21_300483 GMP manufacturer or Tox21_300483 GMP API supplier for your needs.
A Tox21_300483 CoA (Certificate of Analysis) is a formal document that attests to Tox21_300483's compliance with Tox21_300483 specifications and serves as a tool for batch-level quality control.
Tox21_300483 CoA mostly includes findings from lab analyses of a specific batch. For each Tox21_300483 CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Tox21_300483 may be tested according to a variety of international standards, such as European Pharmacopoeia (Tox21_300483 EP), Tox21_300483 JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Tox21_300483 USP).

