
Synopsis
Synopsis
0
CEP/COS
0
KDMF
0
NDC API
0
VMF
0
FDA Orange Book
0
Canada
0
Australia
0
South Africa
DRUG PRODUCT COMPOSITIONS
0
US Patents
0
US Exclusivities
0
Health Canada Patents
US Medicaid
NA
Annual Reports
NA
Regulatory FDF Prices
NA
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
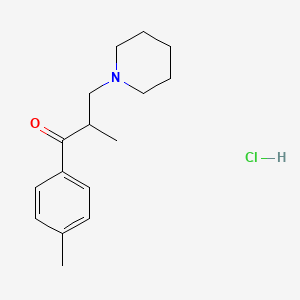

1. Hydrochloride, Tolperisone
2. Midocalm
3. Mydeton
4. Mydocalm
5. Tolperisone
1. 3644-61-9
2. Tolperisone Hcl
3. Midocalm
4. Muscalm
5. Tolperisone (hydrochloride)
6. 2,4'-dimethyl-3-piperidinopropiophenone Hydrochloride
7. 2-methyl-3-piperidino-1-p-tolylpropan-1-one Hydrochloride
8. Tolperisone Hydrochloride [jan]
9. 2-methyl-1-(4-methylphenyl)-3-(piperidin-1-yl)propan-1-one Hydrochloride
10. N-553
11. 2-methyl-1-(4-methylphenyl)-3-(1-piperidinyl)-1-propanone Hydrochloride
12. 3644-61-9 (hcl)
13. 8z075k2tig
14. 1-piperidino-2-methyl-3-(p-tolyl)-3-propanone Hydrochloride
15. Ncgc00094929-01
16. Dsstox_cid_25868
17. Dsstox_rid_81187
18. Dsstox_gsid_45868
19. Arantoick
20. Atmosgen
21. Besnoline
22. Isocalm
23. Kineorl
24. Metosomin
25. Abbsa
26. 2-methyl-3-(piperidin-1-yl)-1-p-tolylpropan-1-one Hydrochloride
27. 2-methyl-1-(4-methylphenyl)-3-piperidin-1-ylpropan-1-one;hydrochloride
28. Cas-3644-61-9
29. Tolperisonehydrochloride
30. Einecs 222-876-5
31. Av 650
32. Unii-8z075k2tig
33. 1-propanone, 2-methyl-1-(4-methylphenyl)-3-(1-piperidinyl)-, Hydrochloride
34. Muscalm (tn)
35. Mfcd00058211
36. Biocalm
37. Minacalm
38. Naismeritin
39. Tolfree
40. Tolisartine
41. Tolpidol
42. Ncgc00182078-02
43. Mls004773941
44. Schembl872770
45. Spectrum1501194
46. 2,4'-dimethyl-3-piperidino-propiophenone Hydrochloride
47. Chembl1395150
48. Dtxsid2045868
49. Chebi:32244
50. Propiophenone, 2,4'-dimethyl-3-piperidino-, Hydrochloride
51. Tolperisone Hydrochloride (jp17)
52. Hms1921p09
53. Pharmakon1600-01501194
54. Hy-b1139
55. Tox21_111360
56. Tox21_113139
57. Ccg-40311
58. Nsc757872
59. S4200
60. Tolperisone Hydrochloride [mi]
61. 2-methyl-1-(4-methylphenyl)-3-(1-piperidyl)propan-1-one Hydrochloride
62. Akos005167032
63. Tox21_111360_1
64. Ac-4685
65. Cs-4744
66. Ncgc00094929-02
67. Ncgc00178060-03
68. Tolperisone Hydrochloride [mart.]
69. As-12472
70. Bp-10588
71. Smr003500666
72. Tolperisone Hydrochloride [who-dd]
73. Ft-0603642
74. Ft-0675274
75. Sw219278-1
76. T1319
77. A18694
78. D01507
79. H12066
80. 644t619
81. Sr-01000872774
82. Tolperisone Hydrochloride, >=98% (hplc), Solid
83. Sr-01000872774-1
84. W-106614
85. Q27271220
86. F9995-4198
87. 2-methyl-3-(piperidin-1-yl)-1-(p-tolyl)propan-1-one Hydrochloride
88. 1-propanone, 2-methyl-1-(4-methylphenyl)-3-(1-piperidinyl)-, Hydrochloride (1:1)
| Molecular Weight | 281.82 g/mol |
|---|---|
| Molecular Formula | C16H24ClNO |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 2 |
| Rotatable Bond Count | 4 |
| Exact Mass | 281.1546421 g/mol |
| Monoisotopic Mass | 281.1546421 g/mol |
| Topological Polar Surface Area | 20.3 Ų |
| Heavy Atom Count | 19 |
| Formal Charge | 0 |
| Complexity | 262 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 1 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 2 |
Muscle Relaxants, Central
A heterogeneous group of drugs used to produce muscle relaxation, excepting the neuromuscular blocking agents. They have their primary clinical and therapeutic uses in the treatment of muscle spasm and immobility associated with strains, sprains, and injuries of the back and, to a lesser degree, injuries to the neck. They have been used also for the treatment of a variety of clinical conditions that have in common only the presence of skeletal muscle hyperactivity, for example, the muscle spasms that can occur in MULTIPLE SCLEROSIS. (From Smith and Reynard, Textbook of Pharmacology, 1991, p358) (See all compounds classified as Muscle Relaxants, Central.)
 Transo-Pharm GmbH works globally to supply Active Pharmaceutical Ingredients adhering to the highest quality & GMP standards.
Transo-Pharm GmbH works globally to supply Active Pharmaceutical Ingredients adhering to the highest quality & GMP standards.

Date of Issue : 2022-02-22
Valid Till : 2026-12-31
Written Confirmation Number : TFDA-0002291
Address of the Firm :

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 25466
Submission : 2011-10-31
Status : Active
Type : II


GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 5590
Submission : 1984-10-24
Status : Inactive
Type : II




Date of Issue : 2025-08-01
Valid Till : 2028-07-02
Written Confirmation Number : WC-0112
Address of the Firm :


Date of Issue : 2025-11-11
Valid Till : 2028-08-22
Written Confirmation Number : WC-0532
Address of the Firm :




 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 5590
Submission : 1984-10-24
Status : Inactive
Type : II

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 40265
Submission : 2025-10-24
Status : Active
Type : II

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 25466
Submission : 2011-10-31
Status : Active
Type : II

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]Japanese Pharmacopoeia Torperisone Hydrochloride
Registration Number : 217MF10876
Registrant's Address : 3-19-12 Kami-Ochiai, Shinjuku-ku, Tokyo
Initial Date of Registration : 2005-11-07
Latest Date of Registration : 2006-07-03

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results] Transo-Pharm GmbH works globally to supply Active Pharmaceutical Ingredients adhering to the highest quality & GMP standards.
Transo-Pharm GmbH works globally to supply Active Pharmaceutical Ingredients adhering to the highest quality & GMP standards.
Date of Issue : 2022-02-22
Valid Till : 2026-12-31
Written Confirmation Number : TFDA-0002291
Address of the Firm : No. 168, Kai Yuan Rd, Hsin-Ying, Tainan City 73055, Taiwan
Date of Issue : 2025-07-22
Valid Till : 2028-07-07
Written Confirmation Number : WC-0104
Address of the Firm : 124, 125 and 126, SIPCOT Industrial Complex, Hosur -635126, Tamil Nadu, India

Date of Issue : 2025-08-01
Valid Till : 2028-07-02
Written Confirmation Number : WC-0112
Address of the Firm : Plot No. 69/A-2, GIDC Industrial Estate, Vapi, Dist

Date of Issue : 2025-11-11
Valid Till : 2028-08-22
Written Confirmation Number : WC-0532
Address of the Firm : N-213/3, M.I.D.C. Tarapur, MIDC Tarapur Boisar, District: Thane-Zone4, Maharasht...

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]Market Place

REF. STANDARDS & IMPURITIES

ANALYTICAL

ABOUT THIS PAGE
11
PharmaCompass offers a list of Tolperisone API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Tolperisone manufacturer or Tolperisone supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Tolperisone manufacturer or Tolperisone supplier.
A Tolperisone manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Tolperisone, including repackagers and relabelers. The FDA regulates Tolperisone manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Tolperisone API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
click here to find a list of Tolperisone manufacturers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PhamaCompass.
A Tolperisone supplier is an individual or a company that provides Tolperisone active pharmaceutical ingredient (API) or Tolperisone finished formulations upon request. The Tolperisone suppliers may include Tolperisone API manufacturers, exporters, distributors and traders.
click here to find a list of Tolperisone suppliers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PharmaCompass.
A Tolperisone DMF (Drug Master File) is a document detailing the whole manufacturing process of Tolperisone active pharmaceutical ingredient (API) in detail. Different forms of Tolperisone DMFs exist exist since differing nations have different regulations, such as Tolperisone USDMF, ASMF (EDMF), JDMF, CDMF, etc.
A Tolperisone DMF submitted to regulatory agencies in the US is known as a USDMF. Tolperisone USDMF includes data on Tolperisone's chemical properties, information on the facilities and procedures used, and details about packaging and storage. The Tolperisone USDMF is kept confidential to protect the manufacturer’s intellectual property.
click here to find a list of Tolperisone suppliers with USDMF on PharmaCompass.
The Pharmaceuticals and Medical Devices Agency (PMDA) established the Japan Drug Master File (JDMF), also known as the Master File (MF), to permit Japanese and foreign manufacturers of drug substances, intermediates, excipients, raw materials, and packaging materials (‘Products’) to voluntarily register confidential information about the production and management of their products in Japan.
The Tolperisone Drug Master File in Japan (Tolperisone JDMF) empowers Tolperisone API manufacturers to present comprehensive information (e.g., production methods, data, etc.) to the review authority, i.e., PMDA (Pharmaceuticals & Medical Devices Agency).
PMDA reviews the Tolperisone JDMF during the approval evaluation for pharmaceutical products. At the time of Tolperisone JDMF registration, PMDA checks if the format is accurate, if the necessary items have been included (application), and if data has been attached.
click here to find a list of Tolperisone suppliers with JDMF on PharmaCompass.
A Tolperisone written confirmation (Tolperisone WC) is an official document issued by a regulatory agency to a Tolperisone manufacturer, verifying that the manufacturing facility of a Tolperisone active pharmaceutical ingredient (API) adheres to the Good Manufacturing Practices (GMP) regulations of the importing country. When exporting Tolperisone APIs or Tolperisone finished pharmaceutical products to another nation, regulatory agencies frequently require a Tolperisone WC (written confirmation) as part of the regulatory process.
click here to find a list of Tolperisone suppliers with Written Confirmation (WC) on PharmaCompass.
Tolperisone Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Tolperisone GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Tolperisone GMP manufacturer or Tolperisone GMP API supplier for your needs.
A Tolperisone CoA (Certificate of Analysis) is a formal document that attests to Tolperisone's compliance with Tolperisone specifications and serves as a tool for batch-level quality control.
Tolperisone CoA mostly includes findings from lab analyses of a specific batch. For each Tolperisone CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Tolperisone may be tested according to a variety of international standards, such as European Pharmacopoeia (Tolperisone EP), Tolperisone JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Tolperisone USP).

