
Synopsis
Synopsis
0
CEP/COS
0
JDMF
0
EU WC
0
KDMF
0
NDC API
0
VMF
0
FDA Orange Book
0
Australia
0
South Africa
DRUG PRODUCT COMPOSITIONS
0
US Patents
0
US Exclusivities
0
Health Canada Patents
US Medicaid
NA
Annual Reports
NA
Regulatory FDF Prices
NA
0
FDF
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
0
News #PharmaBuzz
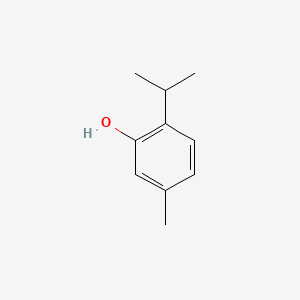

1. Apiguard
1. 89-83-8
2. 2-isopropyl-5-methylphenol
3. Thyme Camphor
4. 5-methyl-2-isopropylphenol
5. 6-isopropyl-m-cresol
6. 5-methyl-2-(1-methylethyl)phenol
7. Thymic Acid
8. 3-p-cymenol
9. Phenol, 5-methyl-2-(1-methylethyl)-
10. P-cymen-3-ol
11. 3-hydroxy-p-cymene
12. 6-isopropyl-3-methylphenol
13. Isopropyl Cresol
14. 5-methyl-2-(propan-2-yl)phenol
15. M-thymol
16. Apiguard
17. 5-methyl-2-propan-2-ylphenol
18. 5-methyl-2-isopropyl-1-phenol
19. 3-methyl-6-isopropylphenol
20. 1-hydroxy-5-methyl-2-isopropylbenzene
21. P-cymene, 3-hydroxy-
22. M-cresol, 6-isopropyl-
23. 1-methyl-3-hydroxy-4-isopropylbenzene
24. 2-hydroxy-1-isopropyl-4-methylbenzene
25. 6-isopropyl-p-cresol
26. 2-isopropyl-5-methyl-phenol
27. Fema No. 3066
28. Phenol, 2-isopropyl-5-methyl-
29. 3-hydroxy-1-methyl-4-isopropylbenzene
30. Thymolum
31. Nsc 11215
32. Nsc-11215
33. Nsc-47821
34. Nsc-49142
35. Chembl29411
36. Chebi:27607
37. 3j50xa376e
38. Ncgc00159373-02
39. Ncgc00159373-04
40. Thymol (natural)
41. Dsstox_cid_14972
42. Dsstox_rid_79231
43. Dsstox_gsid_34972
44. Cymophenol, Alpha-
45. Wln: Qr C1 Fy1&1
46. Caswell No. 856a
47. Fema Number 3066
48. Cas-89-83-8
49. Ccris 7299
50. Hsdb 866
51. Thymol [jan:nf]
52. Einecs 201-944-8
53. Epa Pesticide Chemical Code 080402
54. Thymate
55. Unii-3j50xa376e
56. Ai3-00708
57. Thymol, Puriss.
58. Thymol & Propolis
59. 5-methyl-2-(1-methylethyl)-phenol
60. Mfcd00002309
61. Thymol (tn)
62. Thymol,(s)
63. Thymol Crystal Puriss
64. Thymol, Fcc, Fg
65. Thymolum [hpus]
66. Thymol (jp17/nf)
67. Thymol [vandf]
68. Thymol [fhfi]
69. Thymol [hsdb]
70. Thymol [inci]
71. Thymol [fcc]
72. Thymol [jan]
73. Thymol [usp-rs]
74. Thymol [who-dd]
75. Thymol [ii]
76. Thymol [mi]
77. Thymol [mart.]
78. Ec 201-944-8
79. Thymol, Analytical Standard
80. Thymol, >=98.5%
81. Schembl22165
82. Mls001074692
83. Bidd:er0658
84. Thymol [ep Monograph]
85. Gtpl2499
86. Dtxsid6034972
87. 5-methyl-2-propan-2-yl-phenol
88. 1e06
89. Hms2267p15
90. Zinc967597
91. Hy-n6810
92. Nsc11215
93. Nsc47821
94. Nsc49142
95. Tox21_111613
96. Tox21_300358
97. Bbl011604
98. Bdbm50240432
99. S5157
100. Stk397445
101. Thymol, Tested According To Ph.eur.
102. Akos000119786
103. Tox21_111613_1
104. Ccg-266209
105. Db02513
106. Ks-5170
107. Lmpr0102090029
108. Mb00129
109. Thymol, Saj First Grade, >=98.0%
110. 1-methyl-3-hydroxy-4-isopropyl Benzene
111. Ncgc00159373-03
112. Ncgc00159373-05
113. Ncgc00254459-01
114. Ac-34742
115. Smr000471893
116. Db-002030
117. Cs-0008421
118. Ft-0612711
119. M0410
120. C09908
121. D01039
122. Thymol, Primary Pharmaceutical Reference Standard
123. A845314
124. A861043
125. Ae-562/43461428
126. Q408883
127. Sr-01000763796
128. Sr-01000763796-2
129. W-100357
130. Thymol, Standard For Quantitative Nmr, Tracecert(r)
131. Z57127464
132. Benzene,2-hydroxy,1-isopropyl,4-methyl Thymol
133. F0001-2201
134. Thymol, European Pharmacopoeia (ep) Reference Standard
135. Thymol, United States Pharmacopeia (usp) Reference Standard
136. Thymol, Meets Analytical Specification Of Ph. Eur., Bp, Nf, 99-101%
137. Thymol, Pharmaceutical Secondary Standard; Certified Reference Material
| Molecular Weight | 150.22 g/mol |
|---|---|
| Molecular Formula | C10H14O |
| XLogP3 | 3.3 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 1 |
| Rotatable Bond Count | 1 |
| Exact Mass | 150.104465066 g/mol |
| Monoisotopic Mass | 150.104465066 g/mol |
| Topological Polar Surface Area | 20.2 Ų |
| Heavy Atom Count | 11 |
| Formal Charge | 0 |
| Complexity | 120 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
Anti-Infective Agents; Anti-Infective Agents, Local; Antifungal Agents
National Library of Medicine's Medical Subject Headings online file (MeSH, 2014)
EXPL THER Thymol, a naturally occurring monocyclic phenolic compound derived from Thymus vulgaris (Lamiaceae), has been reported to exhibit anti-inflammatory property in vivo and vitro. However, the mechanism of thymol is not clear. The aim of the present study was to investigate the effects of thymol on allergic inflammation in OVA-induced mice asthma and explore its mechanism. The model of mouse asthma was established by the induction of OVA. Thymol was orally administered at a dose of 4, 8, and 16 mg/kg body weight 1hr before OVA challenge. At 24h after the last challenge, mice were sacrificed, and the data were collected by various experimental methods. The results revealed that pretreatment with thymol reduced the level of OVA-specific IgE, inhibited recruitment of inflammatory cells into airway, and decreased the levels of IL-4, IL-5, and IL-13 in BALF. Moreover, the pathologic changes of lung tissues were obviously ameliorated and goblet cell hyperplasia was effectively inhibited by the pretreatment of thymol. In addition, thymol reduced the development of airway hyperresponsiveness and blocked the activation of NF-kappaB pathway. All data suggested that thymol ameliorated airway inflammation in OVA-induced mouse asthma, possibly through inhibiting NF-kappaB activation. These findings indicated that thymol may be used as an alternative agent for treating allergic asthma.
PMID:24785965 Zhou E et al; Fitoterapia. 2014 Jul;96:131-7. doi: 10.1016/j.fitote.2014.04.016
EXPL THER Obesity has become a worldwide health problem. Most of the synthetic anti-obesity drugs have failed to manage the obesity due to either ineffectiveness or adverse effect. The research of prominent chemical constituents from herbal for the management of obesity has greatly increased. The main objective of the present study was intended to examine the effects of thymol in high-fat diet (HFD)-induced obesity in murine model. Male Wistar rats were fed HFD for 6 weeks to induce obesity. Thymol (14 mg/kg) administered orally twice a day to HFD-fed rats for 4 weeks. Alteration in body weight gain, visceral fat-pads weight and serum biochemical markers were assessed. At the end of study, rats fed with HFD exhibited significantly (p< 0.001) enhanced body weight gain, visceral pad weight, lipids, alanine aminotransferase (ALT), aspartate aminotransaminase (AST), lactate dehydrogenase (LDH), blood urea nitrogen (BUN), glucose, insulin and leptin levels compared with rats fed with normal diets. Thymol treatment showed significantly (p< 0.001) decreased body weight gain, visceral fat-pad weights, lipids, ALT, AST, LDH, BUN, glucose, insulin, and leptin levels in HFD-induced obese rats. Furthermore, thymol treatment showed significantly decreased serum lipid peroxidation and increased antioxidant levels in HFD-induced obese rats. Thymol prevents HFD-induced obesity in murine model through several mechanisms including attenuation of visceral fat accumulation, lipid lowering action, improvement of insulin and leptin sensitivity and enhanced antioxidant potential.
PMID:24175857 Haque MR et al; Toxicol Mech Methods. 2014 Feb;24(2):116-23. doi: 10.3109/15376516.2013.861888. Epub 2013 Dec 5
EXPL THER Mast cells play a critical role in inflammatory skin diseases through releasing proinflammatory mediators; however, few therapies directly target these cells. In 1878, the use of topical thymol, a now recognized potent agonist for transient receptor potential channels, was first described to treat eczema and psoriasis. /The objective was/ to determine the mechanisms through which thymol can alter skin inflammation. METHODS: /This study/ examined the effect of topical thymol on IgE-dependent responses using a mast cell-dependent passive cutaneous anaphylaxis (PCA) model, as well as in vitro-cultured mast cells. Thymol dose-dependently inhibited PCA when administered topically 24 hours before antigen challenge but provoked an ear-swelling response directly on application. This direct effect was associated with local mast cell degranulation and was absent in histamine-deficient mice. However, unlike with PCA responses, there was no late-phase swelling. In vitro thymol directly triggered calcium flux in mast cells through transient receptor potential channel activation, along with degranulation and cytokine transcription. However, no cytokine protein was produced. Instead, thymol induced a significant increase in apoptotic cell death that was seen both in vitro and in vivo. /The authors/ propose that the efficacy of thymol in reducing IgE-dependent responses is through promotion of activation-induced apoptotic cell death of mast cells and that this likely explains the clinical benefits observed in early clinical reports.
PMID:24486068 Full text: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4040322 Wechsler JB et al; J Allergy Clin Immunol. 133 (6): 1735-43 (2014)
For more Therapeutic Uses (Complete) data for THYMOL (8 total), please visit the HSDB record page.
Antifungal Agents
Substances that destroy fungi by suppressing their ability to grow or reproduce. They differ from FUNGICIDES, INDUSTRIAL because they defend against fungi present in human or animal tissues. (See all compounds classified as Antifungal Agents.)
Anti-Infective Agents
Substances that prevent infectious agents or organisms from spreading or kill infectious agents in order to prevent the spread of infection. (See all compounds classified as Anti-Infective Agents.)
Anti-Infective Agents, Local
Substances used on humans and other animals that destroy harmful microorganisms or inhibit their activity. They are distinguished from DISINFECTANTS, which are used on inanimate objects. (See all compounds classified as Anti-Infective Agents, Local.)
Aromatic herbs as feed additives in animal production are encountering growing interest, but data on the fate of the aromatic compounds from the plant in the animal body are very scarce. In the present study, thyme (Thymus vulgaris) herb consisting of leaves and flowers without stems was used as an ingredient in the diet for broilers. The herb was fed for 35 days to five groups of broilers (0, 0.1, 0.2, 0.3, and 1% w/w in the diet). Animal performance and the concentrations of the main essential oil component from thyme, thymol, were measured in gut contents, plasma and liver and muscle tissues using solid phase microextraction and gas chromatography/mass spectrometry. There were no differences between the groups in feed intake, daily weight gain, feed conversion and slaughter weight. Thymol was detected in gut contents, plasma and liver and muscle tissues. Increased intestinal thymol concentrations were found in the group with 1% thyme compared with the other groups (P<0.05). In liver and muscle tissues the thymol levels were close to the limit of quantification. The data do not indicate a positive effect of thyme on animal performance. With high dietary levels of thyme herb, thymol concentrations increased in gut contents and plasma but were very low in edible tissues such as liver and flesh.
PMID:24862829 Haselmeyer A et al; J Sci Food Agric. 2014 May 24. doi: 10.1002/jsfa.6758.
Thymol is readily absorbed from the gastrointestinal tract following oral administration. It is essentially excreted in the urine within the first 24 hours after absorption.
Toxikologische Bewertung. Heidelberg, Berufsgenossenschaft der chemischen Industrie 259 38 p (2000)
Only small amounts of the absorbed substance undergo urinary excretion as hydroxylated compounds. Thymol is predominantly excreted unchanged and in the form of its glucuronide and sulfate conjugates.
Toxikologische Bewertung. Heidelberg, Berufsgenossenschaft der chemischen Industrie 259 38 p (2000)
Substituted monophenols, thymol ... which occur in essential oils of plants, particularly thyme, are ... conjugated with glucuronic acid & sulfate.
Parke, D. V. The Biochemistry of Foreign Compounds. Oxford: Pergamon Press, 1968., p. 147
Thymol has known human metabolites that include Thymoquinol, p-Cymen-8-en-3-ol, p-Cymene-2,3-diol, thymol O-glucuronide, and thymol sulfate.
S73 | METXBIODB | Metabolite Reaction Database from BioTransformer | DOI:10.5281/zenodo.4056560
The potent role of thymol, a natural compound, in modulation of macrophage activity was evaluated by determining all the sequential steps involved during phagocytosis. We found a significant increase in the proliferation of splenocytes in the presence of thymol and it proved to be a good mitogen. Uptake capacity of macrophages was enhanced due to increased membrane fluidity after treatment with thymol and it also increases lysosomal activity of macrophages. Data of superoxide anion generation revealed the involvement of thymol in the generation of respiratory burst as it potentiated this property of macrophages at a concentration of 150 uM. In the case of TNF-a, IL-1beta and PGE(2) a decreased level of secretion was observed 154 pg/mL, 736.1 pg/mL, and 151 pg/mL respectively when compared with lipopolysaccharide treated cells, where the level of these cytokines was significantly high. We also determined the anti-complementary activity of thymol which showed to be more effective than rosmarinic acid. Thus, the results obtained from the study suggest the potential role of thymol as a natural immunostimulatory drug which can be used in the treatment of various immunological disorders.
PMID:24316253 Chauhan AK et al; Int Immunopharmacol. 2014 Feb;18(2):340-6. doi: 10.1016/j.intimp.2013.11.025. Epub 2013 Dec 5.
Related Excipient Companies

Excipients by Applications
Market Place

REF. STANDARDS & IMPURITIES

ABOUT THIS PAGE
49
PharmaCompass offers a list of Thymol API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Thymol manufacturer or Thymol supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Thymol manufacturer or Thymol supplier.
A Thymol manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Thymol, including repackagers and relabelers. The FDA regulates Thymol manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Thymol API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
click here to find a list of Thymol manufacturers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PhamaCompass.
A Thymol supplier is an individual or a company that provides Thymol active pharmaceutical ingredient (API) or Thymol finished formulations upon request. The Thymol suppliers may include Thymol API manufacturers, exporters, distributors and traders.
click here to find a list of Thymol suppliers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PharmaCompass.
A Thymol DMF (Drug Master File) is a document detailing the whole manufacturing process of Thymol active pharmaceutical ingredient (API) in detail. Different forms of Thymol DMFs exist exist since differing nations have different regulations, such as Thymol USDMF, ASMF (EDMF), JDMF, CDMF, etc.
A Thymol DMF submitted to regulatory agencies in the US is known as a USDMF. Thymol USDMF includes data on Thymol's chemical properties, information on the facilities and procedures used, and details about packaging and storage. The Thymol USDMF is kept confidential to protect the manufacturer’s intellectual property.
click here to find a list of Thymol suppliers with USDMF on PharmaCompass.
Thymol Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Thymol GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Thymol GMP manufacturer or Thymol GMP API supplier for your needs.
A Thymol CoA (Certificate of Analysis) is a formal document that attests to Thymol's compliance with Thymol specifications and serves as a tool for batch-level quality control.
Thymol CoA mostly includes findings from lab analyses of a specific batch. For each Thymol CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Thymol may be tested according to a variety of international standards, such as European Pharmacopoeia (Thymol EP), Thymol JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Thymol USP).

