Synopsis
Synopsis
0
API Suppliers
0
USDMF
0
CEP/COS
0
JDMF
0
EU WC
0
KDMF
0
NDC API
0
VMF
0
Listed Suppliers
0
EDQM
0
USP
0
JP
0
Others
0
FDF Dossiers
0
FDA Orange Book
0
Europe
0
Canada
0
Australia
0
South Africa
0
Listed Dossiers
DRUG PRODUCT COMPOSITIONS
0
US Patents
0
US Exclusivities
0
Health Canada Patents
US Medicaid
NA
Annual Reports
NA
Regulatory FDF Prices
NA
0
API
0
FDF
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
0
News #PharmaBuzz
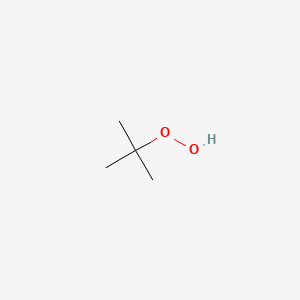

1. Hydroperoxide, T-butyl
2. Hydroperoxide, Tert-butyl
3. T Butyl Hydroperoxide
4. T Butylhydroperoxide
5. T-butyl Hydroperoxide
6. T-butylhydroperoxide
7. Tert Butyl Hydroperoxide
8. Tert Butylhydroperoxide
9. Tert-butylhydroperoxide
10. Tertiary Butylhydroperoxide
11. Tertiary-butylhydroperoxide
1. 75-91-2
2. Tbhp
3. T-butyl Hydroperoxide
4. Tert-butylhydroperoxide
5. 2-hydroperoxy-2-methylpropane
6. Perbutyl H
7. T-butylhydroperoxide
8. 1,1-dimethylethyl Hydroperoxide
9. Cadox Tbh
10. Hydroperoxide, 1,1-dimethylethyl
11. Tert-butyl Hydrogen Peroxide
12. Terc. Butylhydroperoxid
13. Hydroperoxyde De Butyle Tertiaire
14. Hydroperoxide, Tert-butyl
15. Slimicide De-488
16. Tertiary Butyl Hydroperoxide
17. Trigonox A-75
18. Trigonox A-w70
19. Tbhp-70
20. 1,1-dimethylethylhydroperoxide
21. Nsc 672
22. T-buooh
23. Tertiary-butyl Hydroperoxide
24. Tert-butyl-hydroperoxide
25. Dimethylethyl Hydroperoxide
26. T-hydro
27. Perbutyl H 69t
28. Luperox Tbh 70x
29. Trigonox A-w 70
30. Tert-butyl Hydroperoxide Solution
31. Chebi:64090
32. Nsc-672
33. 955vyl842b
34. Tert-butyl Hydroperoxide (70% Solution In Water)
35. Caswell No. 130bb
36. Trigonox A-75 [czech]
37. Tbooh
38. Terc.butylhydroperoxid [czech]
39. Ccris 5892
40. Hsdb 837
41. Terc.butylhydroperoxid
42. Terc. Butylhydroperoxid [czech]
43. Tert Butylhydroperoxide
44. Einecs 200-915-7
45. De 488
46. De-488
47. Brn 1098280
48. Hydroperoxyde De Butyle Tertiaire [french]
49. Unii-955vyl842b
50. Ai3-50541
51. Kayabutyl H
52. Tbuooh
53. Tert-buooh
54. Hydroperoxide, 1,1-dimethylethyl-
55. Perbutyl H 69
56. Perbutyl H 80
57. T-butyl-hydroperoxide
58. Terbutyl Hydroperoxide
59. Tert-butyhydroperoxide
60. Tert-c4h9ooh
61. T-butyl Hydrogenperoxide
62. T-butyl-hydrogenperoxide
63. Tert.-butylhydroperoxide
64. Tert.butyl Hydroperoxide
65. Tertiarybutylhydroperoxide
66. Tertbutylhydrogen Peroxide
67. T-butyl Hydrogen Peroxide
68. Tert.-butyl Hydroperoxide
69. Kayabutyl H 70
70. Dsstox_cid_4693
71. Tert-butylhydrogen Peroxide
72. Ec 200-915-7
73. Dsstox_rid_78866
74. Dsstox_gsid_31209
75. Tertiary Butyl Hydro Peroxide
76. Hydroperoxide,1-dimethylethyl
77. Trigonox A-80 (salt/mix)
78. Un 2093 (salt/mix)
79. Un 2094 (salt/mix)
80. Usp -800 (salt/mix)
81. Chembl348399
82. Nsc672
83. Dtxsid9024693
84. Tert-butyl Hydroperoxide (8ci)
85. Tert-butyl Hydroperoxide, >90% With Water [forbidden]
86. Wln: Qox1&1&1
87. Tert-butyl-hydroperoxide Solution
88. 2-methyl-prop-2-yl-hydroperoxide
89. Zinc8585869
90. Tox21_200838
91. Aztec T-butyl Hydroperoxide-70, Aq
92. Mfcd00002130
93. Tert-butyl Hydroperoxide [ii]
94. Tert-butyl Hydroperoxide [mi]
95. Tert-butyl Hydroperoxide Solution, Cp
96. Akos000121070
97. Tert-butyl Hydroperoxide [hsdb]
98. Ncgc00090725-01
99. Ncgc00090725-02
100. Ncgc00090725-03
101. Ncgc00258392-01
102. Tert-butyl Hydroperoxide Aqueous Solution
103. Hydroperoxide, 1,1-dimethylethyl (9ci)
104. Tert-butyl Hydroperoxide (70% In Water)
105. Tert-butyl Hydroperoxide, >90% With Water
106. B3153
107. Ft-0657109
108. Tert-butyl Hydroperoxide, 70% Solution In Water
109. Q286326
110. J-509597
111. Tert-butyl Hydroperoxide Solution, ~5.5 M In Decane
112. F1905-8242
113. Tert-butyl Hydroperoxide Solution (tbhp), 70% In H2o
114. Tert-butyl Hydroperoxide Solution, 5.0-6.0 M In Decane
115. Tert-butyl Hydroperoxide Solution, 5.0-6.0 M In Nonane
116. Luperox(r) Tbh70x, Tert-butyl Hydroperoxide Solution, 70 Wt. % In H2o
117. Tert-butyl Hydroperoxide Solution, ~80% In Di-tert-butyl Peroxide/water 3:2
118. Tert-butyl Hydroperoxide Solution, Packed In Fep Bottles, ~5.5 M In Decane (over Molecular Sieve 4??)
119. Tert-butyl Hydroperoxide Solution, Packed In Fep Bottles, ~5.5 M In Nonane (over Molecular Sieve 4 ??)
| Molecular Weight | 90.12 g/mol |
|---|---|
| Molecular Formula | C4H10O2 |
| XLogP3 | 0.6 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 2 |
| Rotatable Bond Count | 1 |
| Exact Mass | 90.068079557 g/mol |
| Monoisotopic Mass | 90.068079557 g/mol |
| Topological Polar Surface Area | 29.5 Ų |
| Heavy Atom Count | 6 |
| Formal Charge | 0 |
| Complexity | 35.3 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
t-Butyl hydroperoxide and cumene hydroperoxide, both known to be substrates for glutathione peroxidase, were used to oxidize erythrocyte GSH. Addition of concentrations of hydroperoxides equimolar with respect to GSH in the erythrocytes or whole blood quantitatively oxidizes GSH in the erythrocytes with a half-time of 4.5 s at 37 C and about three times as long at 4 C. In the presence of glucose, normal erythrocytes regenerate all the GSH in about 25 min. However, glucose 6-phosphate dehydrogenase-deficient erythrocytes failed to regenerate GSH. Treatment of erythrocytes with hydroperoxides does not affect erythrocyte survival in rabbits. Oxidation of erythrocyte GSH with equimolar concentrations of hydroperoxides does not lead to formation of mixed disulfides of hemoglobin and GSH. The hydroperoxides do not affect erythrocyte glycolytic and hexose monophosphate-shunt-pathway enzymes. Previous studies on transport of GSSG from erythrocytes were confirmed by using t-butyl hydroperoxide to oxidize erythrocyte GSH.
PMID:4447610 Full text: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC1166284 Srivastava SK et al; Biochem J 139 (2): 289 (1974)
Metabolic activation of peroxides and hydroperoxides to free radicals is associated with the tumor promoting activity of these compounds. tert-Butyl hydroperoxide (t-BOOH) metabolism has been extensively studied as a model of peroxide biotransformation. In vivo studies are limited, and the hemoglobin-thiyl radical was the only species thus far identified in the blood of treated rats. Here we further examine t-BOOH metabolism in vivo with regard to free radical and DNA adduct production. Spin-trapping experiments with phenyl-N-tert-butylnitrone (PBN) led to the detection of electron paramagnetic resonance (EPR) signals in the blood, bile, and organic extracts of the liver and stomach of rats treated with t-BOOH. Analysis of these signals demonstrated that t-BOOH metabolism in vivo produces alkyl radicals, detected in the bile and organic extracts of liver and stomach, in addition to the previously identified hemoglobin-thiyl radical. To characterize the produced alkyl radicals, experiments were performed with (13)C-labeled t-BOOH and two spin traps, PBN and alpha-(4-pyridyl-1-oxide)-N-tert-butylnitrone (POBN). The latter was used because the EPR signals obtained with PBN were too weak to be unambiguous. Nevertheless, the EPR signals present in the bile of animals treated with (13)C-labeled t-BOOH and PBN or POBN were consistent with adducts of (13)C-labeled methyl radical and an unidentified alkyl radical. The latter is probably derived from lipids oxidized by the metabolically produced primary radicals, methyl and its precursor, tert-butoxyl. The presence of 8-methylguanine and 7-methylguanine in hydrolysates of DNA from liver and stomach of rats treated with t-BOOH was also examined. 8-Methylguanine, a typical product of methyl radical attack on DNA, was detectable in both the liver and stomach of treated rats. The results may be relevant to the understanding of the genotoxic properties of other peroxides, particularly of cumene hydroperoxide.
PMID:11080055 Hix S et al; Chem Res Toxicol 13 (10): 1056-64 (2000)
A short-term exposure of PC12 cells to tert-butylhydroperoxide, followed by recovery in fresh culture medium, causes cell death and the extent of this response progressively increases during the 120 min of post-treatment incubation. Morphological and biochemical analyses of these cells revealed that the mode of cell death was necrosis. Cell killing induced by the hydroperoxide seems to be in part mediated by peroxynitrite because the lethal response was markedly and similarly reduced by the nitric oxide synthase inhibitor N omega-nitro-L-arginine methylester and by scavengers of nitric oxide or peroxynitrite. This peroxynitrite-dependent mechanism of cytotoxicity was blunted by antioxidants and inhibitors of mitochondrial permeability transition and the onset of cell death was preceded by mitochondrial depolarization and loss of cellular ATP. We conclude that tert-butylhydroperoxide promotes peroxynitrite-dependent PC12 cell necrosis causally linked to peroxidation of membrane lipids and mitochondrial permeability transition.
PMID:11536321 Palomba L et al; J Neurosci Res 65 (5): 387-95 (2001)
ABOUT THIS PAGE
73
PharmaCompass offers a list of Tert-Butyl Hydroperoxide API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Tert-Butyl Hydroperoxide manufacturer or Tert-Butyl Hydroperoxide supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Tert-Butyl Hydroperoxide manufacturer or Tert-Butyl Hydroperoxide supplier.
A tert Butyl Hydroperoxide manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of tert Butyl Hydroperoxide, including repackagers and relabelers. The FDA regulates tert Butyl Hydroperoxide manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. tert Butyl Hydroperoxide API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
A tert Butyl Hydroperoxide supplier is an individual or a company that provides tert Butyl Hydroperoxide active pharmaceutical ingredient (API) or tert Butyl Hydroperoxide finished formulations upon request. The tert Butyl Hydroperoxide suppliers may include tert Butyl Hydroperoxide API manufacturers, exporters, distributors and traders.
tert Butyl Hydroperoxide Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of tert Butyl Hydroperoxide GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right tert Butyl Hydroperoxide GMP manufacturer or tert Butyl Hydroperoxide GMP API supplier for your needs.
A tert Butyl Hydroperoxide CoA (Certificate of Analysis) is a formal document that attests to tert Butyl Hydroperoxide's compliance with tert Butyl Hydroperoxide specifications and serves as a tool for batch-level quality control.
tert Butyl Hydroperoxide CoA mostly includes findings from lab analyses of a specific batch. For each tert Butyl Hydroperoxide CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
tert Butyl Hydroperoxide may be tested according to a variety of international standards, such as European Pharmacopoeia (tert Butyl Hydroperoxide EP), tert Butyl Hydroperoxide JP (Japanese Pharmacopeia) and the US Pharmacopoeia (tert Butyl Hydroperoxide USP).

